COVID-19-induced thrombotic microangiopathy: A great mimicker of thrombotic thrombocytopenic purpura (TTP)
Abstract
We present a unique case of a 68-year-old male who was diagnosed with COVID-19. His hospital course was complicated by widespread thrombosis, renal failure, and thrombocytopenia. Thrombotic thrombocytopenic purpura was initially suspected, yet plasma exchange and steroids did not improve his disease. Ultimately, he was diagnosed with COVID-19-induced thrombotic microangiopathy.
COVID-19 is a highly pathogenic disease caused by the SARS-CoV-2 virus which primarily results in acute hypoxic respiratory failure. Since its discovery in 2019, COVID-19 has been increasingly associated with a hypercoagulable state, resulting in a high incidence of thrombotic events.1 The pathophysiology of COVID-19-associated hypercoagulability is not well known but may be due to excessive inflammation, platelet activation, endothelial dysfunction, and stasis from the viral disease.2 Thrombotic microangiopathies (TMA) are a spectrum of hematologic findings including microangiopathic hemolytic anemia (MAHA), thrombocytopenia resulting in microvascular thrombosis, and resultant end-organ dysfunction. We present a unique case of COVID-19-induced TMA complicated by widespread venous and arterial thrombosis, which was initially presumed to be a diagnosis of thrombogenic thrombocytopenic purpura (TTP).
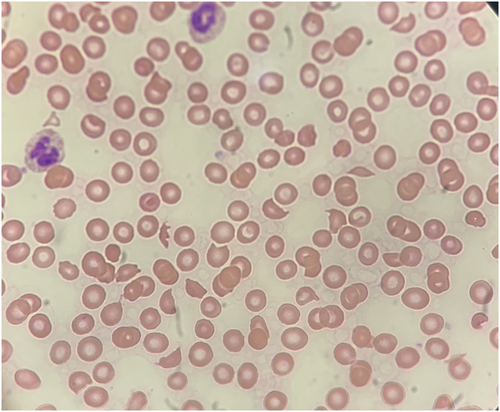
A 68-year-old male patient with past medical history of hypertension, asthma, and allergic rhinitis presented to the emergency room with 9 days of fever, shortness of breath, cough, and altered mental status. He attended a funeral 2 weeks prior and noted that many family members recently tested positive for COVID-19. Upon arrival, the patient was febrile (100.8°F), tachycardic (107 bpm), tachypneic (25 breaths/min), with an oxygen saturation of 70% on room air. He was placed on non-rebreather mask at 15 L of oxygen, after which his oxygen saturation to rose to 95%. The patient was only oriented to person and his physical examination was notable for decreased breath sounds and altered mental status. Nasopharyngeal swab detected the presence of the SARS-CoV-2 virus via reverse transcriptase PCR. The initial complete blood count showed a white blood cell count of 10.1, a hemoglobin of 12.9, and a platelet count of 225. His initial basic metabolic panel was normal. Chest computed tomography (CT) revealed extensive bilateral ground glass opacities with multiple focal areas of consolidation. He was admitted to the medical intensive care unit (MICU) for further monitoring of respiratory status with a low threshold to intubate if he decompensates. The patient was started on dexamethasone, remdesivir, convalescent plasma, ceftriaxone, and azithromycin to treat his COVID-19 infection and possible superimposed bacterial pneumonia. Due to his altered mental status, a head CT was performed, which showed bilateral occipital lobe, right cerebellar lobe, right thalamic, and right corona radiata infarctions concerning for multiple acute ischemic strokes. CT angiogram (CTA) demonstrated a near complete occlusion of the left proximal vertebral artery, also suspicious for an acute to subacute thrombus. Given his COVID-19 diagnosis, elevated D-dimer on arrival, and cerebral thrombosis, a heparin drip was initiated. Upon further questioning, although patient denied vision loss, he was unable to count or track fingers, describe colors, and did not blink to visual threat. Neurology was consulted and diagnosed our patient with Anton–Babinski syndrome, characterized by cortical blindness accompanied by visual anosognosia, defined as the lack insight into vision loss despite clear evidence of blindness. The following day, his platelet count dropped to 73 and his creatinine rose to 5.2. Hemolysis laboratories were significant for undetectable haptoglobin, reticulocytes of 2.27%, normal fibrinogen, and normal PT, PTT, and INR. The patient's D-dimer was 57,438, which remained elevated throughout his hospital course. His abdominal CTA showed hypodensities in the right kidney concerning for infarcts, a thrombus in the right common iliac artery, and splenic infarctions. TTP was the primary diagnosis on our differential given the thrombocytopenia, acute kidney injury, anemia, widespread thrombosis, and hemolytic laboratories. ADAMTS13 levels, HIT antibodies, and anti-phospholipid antibodies were drawn. Hematology was consulted and reviewed a peripheral blood smear which revealed the 4+ schistocytes per high power field (Figure 1). The patient was immediately started on plasma exchange (PLEX) therapy and steroids the same day for suspected diagnosis of TTP. In addition, the heparin drip was stopped, and argatroban was started as empiric therapy for heparin-induced thrombocytopenia (HIT). Interventional radiology performed a common iliac artery angiogram but was unable to retrieve the clot or stent the occlusion. While the ADAMTS13 antibodies were pending, the patient continued to receive PLEX and steroids for 13 total days. His platelet count was monitored daily as an adjunctive marker for response to treatment, yet his platelets did not make a full recovery as expected. By day five, his HIT antibodies came back negative, so argatroban was switched back to heparin. The hypercoagulable workup including protein C, protein S, anti-thrombin III, factor V leiden, prothrombin gene mutation, SPEP, beta-2-glycoprotein antibodies, and cardiolipin antibodies, came back negative. By Day 13, the ADAMTS13 antibodies from day one came back decreased at 43% and his platelet count only increased to 98. At this point, the initial diagnosis of TTP was deemed less likely due to the inadequate platelet rise with mainstay treatment and a low, yet not undetectable, ADAMTS13 level. Thereafter, PLEX was stopped as our index of suspicion for TTP fell. To conclude the case, hematology diagnosed this patient with COVID-19-induced TMA. The patient recovered from a pulmonary standpoint and was eventually discharged to a long-term rehabilitation facility on heparin prophylaxis. He has not regained any meaningful vision back to this date.
TMAs pose both significant diagnostic and therapeutic challenges. They produce a variety of signs, symptoms, and laboratory derangements. TMA treatment requires attention to the microangiopathy itself and its subsequent complications, as well as the underlying disease. This clinical case demonstrates an atypical case of COVID-19-induced TMA, which proved to be a difficult and lengthy diagnosis of exclusion. The intriguing presentation of this patient within the first day included classic COVID-19 symptoms and visual anosognosia. Shortly after admission, his hospital course was complicated by the findings of widespread thromboses, acute kidney failure, and schistocytes on peripheral blood smear. It is important to note that TTP classically results in small vessel thrombi formation, however, large vessel occlusions, while rare, may occur with this disease process as well, as seen in our patient. The majority of thrombi seen in our patient occurred in small vessels, such as in renal and cerebral arterioles, however, our patient also presented with large vessel occlusions in his vertebral and common iliac arteries. Following these findings, the list of differential diagnoses was expansive. In addition to COVID-19, the patient had no other known sites of infection. Anti-phospholipid syndrome (APLS) was considered; however, this was essentially ruled out due to negative APLS antibodies and the high presence of schistocytes on the peripheral blood smear, which are usually not a feature of APLS. HIT was considered less likely given the presence of schistocytes and negative HIT antibodies. Acquired infectious hemolytic uremic syndrome (HUS) was also considered as a potential diagnosis, but lower on our differential due to the absence of recent gastrointestinal symptoms. Complement-mediated atypical HUS was not suspected as complement levels were not suppressed, although it did not exclusively rule out the disease. There were also no causative medications of atypical HUS, and genetic screening and presence of autoantibodies to complement mediators were not available. Disseminated intravascular coagulation (DIC) was also considered a possibility, however, the patient's PT, PTT, and INR were all within normal limits. On initial presentation, TTP was the most likely diagnosis, as the patient exhibited the “classic pentad” of fever, hemolytic anemia, thrombocytopenia, renal impairment, and neurologic manifestations.3 Hence, hematology was consulted for consideration of emergent PLEX.
TTP is a life-threatening TMA characterized by systemic platelet clumping, profound thrombocytopenia, fragmentation of blood cells, and organ ischemia, particularly in the kidneys and the brain. TTP is characterized by a deficiency in ADAMTS13 activity, an enzyme that cleaves von Willebrand factor (vWF) multimers in thrombogenesis. The lack of ADAMTS13 activity causes vWF multimers to accumulate, causing platelet activation, ultimately resulting in systemic microthrombi formation and the “classic pentad” of TTP. When TTP is suspected, it is imperative to obtain baseline ADAMTS13 activity levels and consult hematology, as this is one of the few hematologic emergencies requiring immediate treatment. First line therapy includes immediate plasmapheresis and steroids. Eculizumab may be a potential option for providers in similar predicaments as it is used to treat atypical HUS, however, there is limited data in patients with COVID-19 infection. Left untreated, TTP has been shown to have survival rates less than 10%. Plasmapheresis replaces large volumes of plasma, replenishes activity of ADAMTS13, and removes anti-ADAMTS13 autoantibodies. Further therapies include rituximab, caplacizumab, a medication which blocks vWF platelet binding domains, and even splenectomy.4
This patient's diagnosis and management became complicated as his treatment with plasmapheresis and steroids failed to improve his platelet count. By Day 13 of therapy, his platelet count remained stagnant at 98. Although his ADAMTS13 activity level was low, it was not <10%, making TTP a less likely diagnosis. Of note, as many as 40% of patients that clinically appear to have TTP can have ADAMTS-13 levels above 10%. With this information, our previously certain diagnosis came into question. Although TTP may mimic a secondary-induced TMA, if suspected, it is imperative to not miss this life-threatening condition and start treatment early in the clinical course. As this patient improved with supportive care and treatment of COVID-19, he was diagnosed with COVID-19-induced TMA. Recent emerging anecdotal evidence supports this disease as a unique subset of the TMA syndromes. The clinical, laboratory, and pathological findings confirm a microangiopathic disease process, yet typical TMA treatments thus far are proving to be ineffective.5
AUTHOR CONTRIBUTIONS
Dr. Brian Warnecke, Dr. Vidit Kapoor, and Dr. Paromita Datta were the patient's physicians. Dr. Brian Warnecke collected the clinical and laboratory data and drafted the manuscript. Alexandra Wehbe collected background data. All authors contributed to the critical review of the paper.
ACKNOWLEDGMENTS
None.
CONFLICT OF INTEREST
The authors have no conflicts of interest to disclose.
CONSENT
Written informed consent was obtained from the patient to publish this report in accordance with the journal's patient consent policy.
Open Research
DATA AVAILABILITY STATEMENT
Data available on request from the authors.