Distinct Electron Conductance Regimes in Bacterial Decaheme Cytochromes
Graphical Abstract
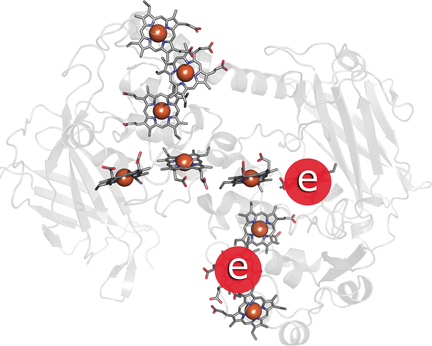
How many electrons? Decaheme cytochromes function as electron conduits for extracellular electron transport in Shewanella oneidensis MR-1. Calculations suggest that redox potentials of the hemes depend on the number of electrons flowing through the system, and that different regimes are operational at different environments or conditions (in vivo, or when the cytochrome is isolated in solution or bound to the electrodes).
Abstract
Shewanella oneidensis MR-1 gains energy by extracellular electron transfer to solid surfaces. They employ c-type cytochromes in two Mtr transmembrane complexes, forming a multiheme wire for electron transport across the cellular outer membrane. We investigated electron- and hole-transfer mechanisms in the external terminal of the two complexes, MtrC and MtrF. Comparison of computed redox potentials with previous voltammetry experiments in distinct environments (isolated and electrode-bound conditions of PFV or in vivo) suggests that these systems function in different regimes depending on the environment. Analysis of redox potential shifts in different regimes indicates strong coupling between the hemes via an interplay between direct Coulomb and indirect interactions through local structural reorganization. The latter results in the screening of Coulomb interactions and explains poor correlation of the strength of the heme-to-heme interactions with the distance between the hemes.