The benefits and challenges of family genetic testing in rare genetic diseases—lessons from Fabry disease
Abstract
Background
Family genetic testing of patients newly diagnosed with a rare genetic disease can improve early diagnosis of family members, allowing patients to receive disease-specific therapies when available. Fabry disease, an X-linked lysosomal storage disorder caused by pathogenic variants in GLA, can lead to end-stage renal disease, cardiac arrhythmias, and stroke. Diagnostic delays are common due to the rarity of the disease and non-specificity of early symptoms. Newborn screening and screening of at-risk populations, (e.g., patients with hypertrophic cardiomyopathy or undiagnosed nephropathies) can identify individuals with Fabry disease. Subsequent cascade genotyping of family members may disclose a greater number of affected individuals, often at younger age than they would have been diagnosed otherwise.
Methods
We conducted a literature search to identify all published data on family genetic testing for Fabry disease, and discussed these data, experts’ own experiences with family genetic testing, and the barriers to this type of screening that are present in their respective countries.
Results
There are potential barriers that make implementation of family genetic testing challenging in some countries. These include associated costs and low awareness of its importance, and cultural and societal issues. Regionally, there are barriers associated with population educational levels, national geography and infrastructures, and a lack of medical geneticists.
Conclusion
In this review, the worldwide experience of an international group of experts of Fabry disease highlights the issues faced in the family genetic testing of patients affected with rare genetic diseases.
1 INTRODUCTION
Patients with rare genetic diseases often experience substantial delays in diagnosis, which can negatively impact the management and postpone treatment. Family genetic testing or cascade genotyping of all newly diagnosed individuals can accelerate the diagnosis of rare genetic diseases, and allow more patients to be identified at a younger age. Fabry disease (OMIM #301500) is a rare, progressive, X-linked lysosomal storage disorder caused by pathogenic variants in the GLA gene (HUGO Gene Nomenclature Committee ID: 4296; Gene Entrez: 2717; NCBI reference sequence: NM_000169.2) that encodes α-galactosidase (α-GAL, Enzyme Commission number: EC 3.2.1.22; UniProt ID: P06280), resulting in the absence or substantially decreased activity of the enzyme (Desnick et al., 2003; Eng et al., 1993; Germain, 2010; Germain & Poenaru, 1999). A deficiency in α-GAL activity leads to the progressive accumulation of glycosphingolipids, mainly globotriaosylceramide (GL-3 or Gb3) and its deacylated form globotriaosylsphingosine (lyso-GL-3 or lysoGb3; Auray-Blais et al., 2015; Pettazzoni et al., 2017), in virtually all cells throughout the body; this accumulation can then cause a range of symptoms including neuropathic pain (Politei et al., 2016), gastrointestinal disturbances (Hopkin et al., 2008), angiokeratoma, fatigue (Germain, Fouilhoux, et al., 2019), impaired sweating, heat/cold intolerance, respiratory involvement (Magage et al., 2007), and/or arterial remodeling (Boutouyrie et al., 2002). Over time, this results in end organ damage such as renal and/or cardiac failure, arrhythmia, and early stroke (Hagège et al., 2019; Kolodny et al., 2015; Linhart et al., 2020; Namdar, 2016; Ortiz et al., 2010; Rost et al., 2016). Besides classic Fabry disease, several pathogenic variants have been reported to be associated with a later-onset, predominantly cardiac phenotype of Fabry disease, including p.F113L (Oliveira et al., 2020), p.N215S (Germain et al., 2018), p.M296I, p.R301Q (Germain, 2001), p.G328R, and IVS4+919G>A (Hsu et al., 2016; Ishii et al., 2002).
Fabry disease is a rare condition and its early symptoms are non-specific (Germain, 2010; Ortiz et al., 2018). This contributes to diagnostic delays, sometimes longer than a decade (Desnick et al., 2003; Germain, 2010; Mehta et al., 2010; Reisin et al., 2017; Wilcox et al., 2008). To improve the diagnosis of Fabry patients, newborn screening initiatives have been carried out in some countries (e.g., Italy, Japan, and Taiwan; Burlina et al., 2018; Chinen et al., 2017; Liao et al., 2018; Spada et al., 2006), and screening studies have been conducted in at-risk populations (e.g., hypertrophic cardiomyopathy, hemodialysis patients; Hagège et al., 2011; Nakao et al., 1995, 2003; Sachdev et al., 2002). Newborn screening studies have reported that up to 0.03%–0.08% of screened newborns may have Fabry disease (Chinen et al., 2017; Liao et al., 2018; Spada et al., 2006). Diagnosing newborns before symptom onset, which often occurs in childhood but can be delayed to adulthood in some patients, would allow for close monitoring and potential treatment, before irreversible severe disease develops. A large analysis of more than 60 GLA screening studies of hemodialysis, cardiac, and stroke populations has revealed pathogenic GLA variants in, respectively, 0.21% and 0.15% of screened male and female hemodialysis patients, 0.94% and 0.90% of screened male and female cardiac patients, and 0.13% and 0.14% of screened male and female stroke patients (Doheny et al., 2018).
Although valuable tools to improve diagnosis and identify undiagnosed patients, screening studies for Fabry disease, may reveal individuals with GLA variants of yet unknown significance or non-pathogenic (likely) benign variants (Doheny et al., 2018; Germain, Fouilhoux, et al., 2019; Germain & Jurca-Simina, 2018; Germain et al., 2020; van der Tol et al., 2014; Varela et al., 2020). Reanalysis of the GLA variants identified in 63 previously published screening studies demonstrated that 47.9% of males and 74.1% of females had non-pathogenic variants (Doheny et al., 2018). This highlights the need to be aware of likely benign and benign GLA variants (Germain, Fouilhoux, et al., 2019; Germain et al., 2020; Richards et al., 2015) when interpreting the findings of screening studies and determining the prevalence of Fabry disease based on screening reports. Current Fabry disease management recommendations state that patients who are identified as having a GLA variant of unknown significance require further diagnostic testing to determine clinical, histological, or biochemical evidence of Fabry disease in order to confirm the pathogenicity of the variant before initiating any disease-specific treatment (Germain, Fouilhoux, et al., 2019). A fully confirmed diagnosis of Fabry disease, for example, is required before enzyme replacement therapy (ERT) or chaperone therapy can be initiated. Moreover, specific therapy for Fabry disease should not be started in individuals bearing a non-pathogenic GLA variant (Germain, Fouilhoux, et al., 2019).
In this context, the combination of a detailed pedigree analysis and cascade genetic testing of at-risk family members (family screening) of newly diagnosed patients represents a powerful tool to increase the number of patients identified, improve early diagnosis, and shed light on the pathogenicity of novel GLA variants identified through various screening initiatives. One large study reported that analysis of the pedigrees of 74 propositi diagnosed with Fabry disease identified, on average, 5 affected individuals related to each index case (Laney & Fernhoff, 2008), whereas another study found 15 affected relatives per index patient among 31 families (Rozenfeld et al., 2019). One Brazilian study described family screening for three index cases, and, for one of those cases, 18 affected relatives were identified (Silva et al., 2016). This has prompted the most recent international consensus management and treatment recommendations for adults and children with Fabry disease to highlight the importance of clinical and genetic screening of at-risk family members of newly diagnosed patients (Germain, Fouilhoux, et al., 2019; Hopkin et al., 2016; Ortiz et al., 2018).
Although family genetic testing of newly diagnosed patients appears the most effective strategy to identify undiagnosed Fabry patients, it is not universally implemented. As a result, the International Fabry Family Screening Advisory Board, consisting of 19 Fabry experts from 15 countries and 4 continents, conducted a literature search to identify all published data on family screening for Fabry disease, and discussed these data, their own experiences with family genetic testing for Fabry disease, and the barriers to this type of screening that are present in their respective countries. For most countries, the experiences are based on the opinion of one physician and his/her personal network, and are therefore not necessarily reflective of the country as a whole. Nevertheless, this unique review is one of the first initiatives to describe the global practical experiences of family genetic testing, and will provide valuable insights to medical geneticists aiming to improve the diagnosis of rare diseases worldwide.
1.1 Insights from the literature
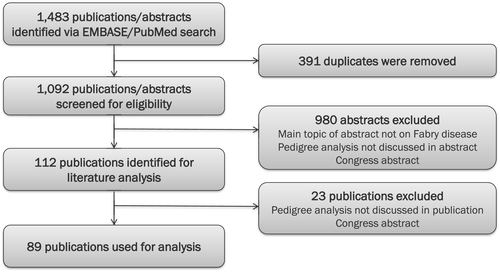
In March 2018, a comprehensive literature review was conducted to identify published data on family genetic testing in Fabry disease. The primary objective of the review was to gain insight into the role of pedigree drawing and family genetic testing in the diagnosis of Fabry disease. The EMBASE and NCBI PubMed databases were used to search for papers published in English from 2001 (i.e., when ERT became available) up to March 2018. This search was repeated in June 2020 to identify additional papers. A total of 1,483 publications were identified; after the removal of duplicates, abstracts of 1,092 publications were screened for eligibility (Figure 1). Of these, 980 publications were excluded because they either were not on Fabry disease or did not discuss or report on family genetic testing or pedigree analysis of individuals identified with a GLA variant. Congress abstracts were also excluded from the analysis. Overall, 112 publications were included in the literature analysis. Following a review of the full papers, 23 more papers were excluded based on the eligibility criteria. This left a total of 89 publications for the final review (Appendix 1). The publications analyzed consisted of 49 case reports, 36 clinical studies, 3 review papers, and 1 consensus report published in 65 different journals.
A total of 365 probands with Fabry disease were identified in 82 of the 89 publications (7 studies did not indicate a proband). Of the 365 probands, 1,744 affected family members were identified through family genetic testing, which is equivalent to an average of 4.8 additional affected family members per proband. Although 65% of the probands identified in these papers were male, nearly two-thirds (65%) of the affected relatives identified through genetic testing were female. This literature review suggests that family genetic testing used in combination with newborn screening or screening of at-risk populations, such as patients on renal replacement therapy or those with hypertrophic cardiomyopathy (HCM), can lead to the identification of several additional family members with Fabry disease (Table 1), with one screening study among 150 patients with HCM reporting an additional 99 relatives diagnosed with the disease from 21 probands (Azevedo et al., 2020). Variable prevalence of Fabry disease was found using newborn screening or screening of at-risk populations, with additional affected relatives disclosed using family genetic testing after identification of index cases (Adalsteinsdottir et al., 2017; Azevedo et al., 2020; Barman et al., 2020; Chinen et al., 2017; De Brabander et al., 2013; Feriozzi et al., 2007; Hagège et al., 2011; Liao et al., 2018; Lin et al., 2018; Lv et al., 2009; Maron et al., 2018; Merta et al., 2007; Okur et al., 2013; Russo et al., 2018; Silva et al., 2016; Spada et al., 2006; Terryn et al., 2008; Turkmen et al., 2016; Veloso et al., 2018). Family screening for rare genetic diseases can be highly effective, but the logistics and economics of its implementation are complex. Moreover, there are numerous geographic, environmental, societal, and cultural barriers that limit the adoption of pedigree analysis and family genetic screening throughout the different regions of the world.
| Reference | Country of study/proband | Proband diagnosis—type of screening | Size of screening population, n | Age/gender of proband(s) at diagnosis | Family members identified through family analysis, n |
|---|---|---|---|---|---|
| Adalsteinsdottir et al., (2017) | Iceland | At-risk population screening—HCM | Nationwide screening among HCM patients, size not specified |
8 Probands; Family A: 3 males, aged 50, 54, and 61 years; 2 females Family B: 2 males, aged 59 and 61 years; 1 female |
8 Probands +33 relatives; Family A: 16 relatives (8 females) Family B: 25 relatives (15 females) |
| Azevedo et al., (2020) | Portugal | At-risk population screening—HCM | 150 | 21 Probands (8 females) | 21 Probands +99 relatives (65 females) |
| Barman et al., (2020) | Turkey | At-risk population screening—HCM | 190 | 2 Probands (both females, aged 55 and 58 years) |
Proband 1 (55 years old) +1 relative (1 female) Proband 2 (58 years old) +2 relatives (1 female) |
| Chinen et al., (2017) | Japan | NBS | 2,443 | 1 Proband (male, aged 11 years) | Proband +2 relatives (2 females) |
| De Brabander et al., (2013) | Belgium | At-risk population screening—stroke | 1,000 | 5 Probands (all females) |
5 Probands +18 relatives |
| Feriozzi et al., (2007) | Italy | At-risk population screening—HD | Not stated | 1 Proband (male, aged 26 years) | Proband +2 relatives (1 female) |
| Hagège et al., (2011) | France | At-risk population screening—HCM | 392 (278 males) | 4 Probands (all males, aged 41, 49, 59, and 59 years) | 4 Probands +8 relatives (6 females) |
| Liao et al., (2018) | Taiwan | NBS | 792,247 | 2 Probands (1 male, 1 female; newborn) |
Family 1: female proband +3 relatives (1 female) Family 2: male proband +7 relatives (4 females) |
| Lin et al., (2018) | Taiwan | At-risk population screening—CKD | 1,012 | 6 Probands (all males) | 4 Probands +3 relatives (2 females) |
| Lv et al., (2009) | China | At-risk population screening—HD | 1,662 | 2 Probands (both males, aged 33 and 50 years) |
Proband 1 (33 years old) +6 relatives (3 females) Proband 2 (50 years old) +6 relatives (5 females) |
| Maron et al., (2018) | US | At-risk population screening—HCM | 585 | 2 Probands (1 male, aged 53 years; 1 female, aged 69 years) |
Proband male +11 relatives (7 females) Proband female +16 relatives (13 females) |
| Merta et al., (2007) | Czech Republic | At-risk population screening—HD | 3,370 (54.9% female) | 5 Probands (4 males, 1 female; aged 26, 45, 64, 67, and 54, respectively) | Several additional relatives with undiagnosed FD were identified from pedigree analysis |
| Okur et al., (2013) | Turkey | At-risk population screening—HD | 1,136 | 2 Probands (both males, aged 48 and 52 years) |
Proband 1 (48 years old) +1 relative (1 female) Proband 2 (52 years old) +5 relatives (4 females) |
| Russo et al., (2018) | Italy | At-risk population screening—HD | Not stated | 1 Proband (male, aged 44 years) | 1 Proband +3 relatives (3 females) |
| Silva et al., (2016) | Brazil | At-risk population screening—HD | 2,583 | 3 Probands (all males, aged 44, 61, and 73 years) |
Proband 1 (44 years old) +5 relatives Proband 2 (61 years old) +18 relatives Proband 3 (73 years old) +2 relatives |
| Spada et al., (2006) | Italy | NBS | 37,104 | 12 Probands (all males) | 12 Probands +29 relatives (24 females) |
| Terryn et al., (2008) | Belgium | At-risk population screening—HD | 922 (742 females) | 2 Probands (both females, aged 80 and 84 years) |
Proband 1 (80 years old) +5 relatives (3 females) Proband 2 (84 years old) +4 relatives (2 females) |
| Turkmen et al., (2016) | Turkey | At-risk population screening—CKD | 313 | 3 Probands (all males) | 3 Probands +8 relatives (4 females) |
| Veloso et al., (2018) | Brazil | At-risk population screening—HD | 108 | 1 Proband (male, aged 43 years) | Proband +11 relatives (8 females) |
- Abbreviations: CKD, chronic kidney disease; FD, Fabry disease; HCM, hypertrophic cardiomyopathy; HD, hemodialysis; NBS, newborn screening.
2 PRACTICAL IMPLEMENTATION OF A PEDIGREE ANALYSIS IN FABRY DISEASE
2.1 Constructing a family pedigree
When a new patient is diagnosed with a rare genetic disease such as Fabry disease, clinicians/geneticists should draw a detailed family pedigree with a standard or a clearly defined format to show male and female patients, deceased patients, patients who have had clinical assessment, and patients who have been genetically tested; genotypes should be noted on the pedigree (Germain & Jurca-Simina, 2018). Depending on the nature of the GLA variant and the pattern of X-chromosome inactivation (XCI), heterozygous females will either be (rarely) asymptomatic, mildly symptomatic, or develop Fabry-related tissue and organ damage. It is, therefore, vital that all potentially affected females in the proband's family should undergo genetic testing after informed consent, and that a full medical history be obtained.
A detailed, fully annotated Fabry family pedigree may appear cluttered and initially confusing to the untrained reader; however, once the symbols and notations are understood, an up-to-date family pedigree provides an ideal way to keep the family medical history organized and easily accessible. Displaying a family pedigree in this format allows physicians to easily visualize the inheritance patterns or symptom clustering that may make interpretation of clinical phenotypes easier. It is also important that physicians and genetic counselors are aware of, and fully compliant with, new data privacy regulations when obtaining and storing a patient's genetic pedigree information. Ideally, family genetic pedigrees should be created and saved digitally, since digital pedigrees are easier to access, adapt, and update over time. Several online software tools for pedigree drawing are currently available (e.g., Cyrillic: http://www.apbenson.com/why-use-cyrillic/; Genial Pedigree Draw: http://pedigreedraw.com/; Invitae: https://www.invitae.com/en/familyhistory/; Phenotips: https://phenotips.org/; Progeny: http://www.progenygenetics.com/). While useful, some of these software tools are quite complicated. There is a need for new, user-friendly, and simpler pedigree drawing software tools.
2.2 Example of a family pedigree in Fabry disease
Figure 2 illustrates a “typical” example of a three-generation family pedigree in Fabry disease. In this example, a 27-year-old male (index case, III-1) without any medical history presented with a stroke. Fabry disease was hypothesized and subsequently diagnosed by the identification of a pathogenic variant p.Leu243Phe (c.729G>C, rs397515874) in GLA. Initial family screening showed the expected heterozygosity in his mother (II-2, panel B), therefore, prompting cascade genotyping of GLA in his brother (III-3, already affected with end-stage renal disease in the absence of diagnosis), which detected the presence of the pathogenic variant in GLA, which was also found in his daughter (IV-1, panel C). Both mother's parents of the index case were already deceased (I-1 and I-2, panel B). Despite extensive discussion, a second branch of the family was brought to medical attention only after a few years (panel D). Further pedigree drawing and cascade genotyping then identified five additional affected individuals, highlighting the need for repeated careful investigation of family members (panels E, F). ERT was initiated in individual III-6 at 30 years of age. After 19 years of specific therapies, this male patient has normal cardiac and kidney function, in contrast to family members who have been initiated much later on FD-specific therapy.
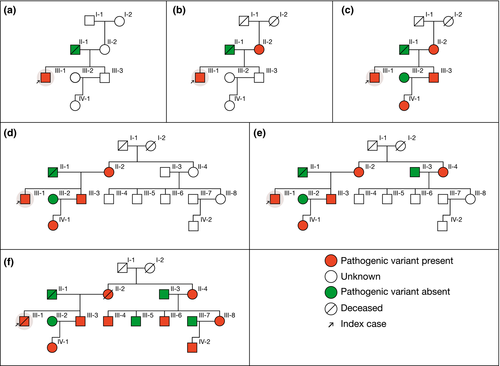
2.3 Interpreting family pedigree information
There are several key issues to be aware of when interpreting genetic and clinical information in a family pedigree. As mentioned previously, the clinical phenotype in heterozygous females for a GLA variant depends partly on the type of GLA pathogenic variant (mild missense vs. others) and partly on the pattern of XCI. This inactivation makes it difficult to predict the clinical presentation and pattern of disease progression in female patients affected by Fabry disease. XCI is the process that determines the pattern of inactivation of the two X chromosomes in cells throughout the female body and (often) occurs randomly in most females. A proportion of females, however, exhibits what is known as “skewed” XCI, in which one of the X chromosomes is preferentially silenced relative to the other. The direction of XCI skewing (i.e., in favor of the X chromosome bearing the wild-type or the one bearing the mutated GLA allele) in female patients who are heterozygous for the GLA pathogenic variant has been shown to impact the clinical phenotype, disease severity, and disease progression in females with classic Fabry disease (Echevarria et al., 2016; Germain & Jurca-Simina, 2018). Therefore, females with the same GLA pathogenic variant can manifest different symptoms or develop symptoms of different severity and at varying ages, complicating the interpretation of the family clinical history and emphasizing the importance of genotyping all potentially at-risk female relatives, irrespective of whether symptoms are present (Germain, Arad, et al., 2019).
Another point to be aware of when collecting family pedigree information is the potential for false paternity information to be omitted, either intentionally or unintentionally. This possibility should be borne in mind and allow for cautious discussions with patients. In addition, consanguinity and the custom of marrying only within the limits of the close local community (endogamy) may be prevalent in some countries for cultural reasons. The impact of these issues should be considered, even in an X-linked genetic disease, where this may result in the occurrence of homozygous females. Finally, although very unusual, Fabry disease can unexpectedly recur within a family due to germline mosaicism (i.e., when a genetic variant occurs during the development of the germinal cell line in a non-affected parent; Dobrovolný et al., 2005; Germain & Jurca-Simina, 2018). In contrast, Fabry disease can also appear spontaneously in a family with no previous history of the disease because of rare (~5%–10% of cases) de novo variants (Gal et al., 2017).
3 BARRIERS TO FAMILY SCREENING/PEDIGREE ANALYSIS
There are many potential barriers that may negatively impact family genetic testing for Fabry disease and limit its use (Table 2). These barriers vary from region to region and between countries according to screening and treatment costs, local regulations, and cultural issues.
| Type of barriers |
|---|
| Cost |
|
| Cultural/societal issues |
|
| Logistical issues |
|
|
Communication
|
3.1 Cost of family screening and genetic testing
The cost of genetic testing for Fabry disease may be a barrier to implementing family screening in many countries; however, and although the cost of genetic testing is significant, any screening cost must always be considered relative to the cost of treating potentially avoidable complications of Fabry disease, such as renal failure or acute stroke. Another important financial barrier is the cost of funding geneticists/counselors to conduct and support genetic testing for Fabry disease, particularly in developing nations with a wide geographical area and low population density. In some countries, patient organizations may be able to assist with either the cost or the logistics of accessing available family screening. In India, which has no national policy for the provision of ERT in Fabry disease, patients can be diagnosed through charitable access programs.
3.2 Cultural and societal issues
There are many cultural and societal issues that affect family genetic testing for a rare disease. Some geographical regions have cultures that favor consanguineous marriages. For example, the custom of consanguineous marriage within Muslim communities is embedded in many Middle Eastern cultures (e.g., in Oman) and is favored by local populations despite the known risk of increasing the prevalence of genetic diseases (particularly autosomal recessive diseases such as spinal muscular atrophy, cystic fibrosis, phenylketonuria), recessively inherited congenital birth defects, and intellectual deficiency (Rajab et al., 2013). Since Fabry disease has an X-linked inheritance pattern, consanguinity likely increases the prevalence of homozygous Fabry disease females, who are anticipated to exhibit more severe clinical phenotypes with, however, a negligible impact in the overall prevalence of Fabry disease females, mostly consisting of heterozygotes. This phenomenon has been reported in some remote areas of China. Because of possible stigmatization, it is necessary to emphasize data privacy when communicating with patients and their family members, and to carefully choose methods of communication for maximum discretion. The complex family structures that exist in some countries, such as Mexico, where men may have two or three different families, also complicate family screening and pedigree analysis. Extended, fragmented family structures are prevalent in many countries in both the developed and developing regions of the world; family members may be widely separated geographically and not in contact with each other, thus making it difficult to trace family members of a newly diagnosed patient with Fabry disease. In some countries (e.g., Mexico), high levels of internal migration and regional or local violence/conflicts can make it hard to trace family members. Moreover, certain countries (e.g., Saudi Arabia) have highly conservative, patriarchal societies in which husbands/fathers prevent female family members from having genetic testing; this deeply rooted cultural and religious issue can be very difficult to overcome. In some developing nations, such as in India, there is considerable stigma associated with assigning to a family a rare genetic disease diagnosis for which there is limited access to expensive treatment; patients often prefer to live in the hope that they are not affected by any disease (including Fabry disease) rather than seek genetic testing. In South Korea, although ERT for Fabry disease has been fully funded by the government since 2003, the disease is still underdiagnosed. The authors of a recent nationwide survey attributed this in part to the refusal of family genetic testing due to fear of stigmatization (Choi et al., 2017). Religious issues may also present barriers to family analysis and genetic testing for inherited diseases, as is the case in some communities in Mexico, and in many nations with conservative or strongly religious populations.
3.3 Logistical issues
The geographic spread of populations over large distances, including very remote and hard-to-reach areas, can pose logistical challenges for implementing family screening programs for Fabry disease in certain regions/countries (e.g., Russia, China, and Colombia). Families may migrate over large distances, making interfamilial communication difficult. In addition, many developing nations have a weak national communications infrastructure and a poor transport network, which compounds the problems of diagnosis when there are few/no regional genetic testing services—patients usually need to travel long distances to the capital city to receive their specialist's diagnosis and care. In wealthier countries, dried blood spot (DBS) samples for genetic analysis can be easily transported by courier to the genetic testing laboratory. In Russia, for example, a funded courier service exists that allows healthcare professionals in any city to call a courier, and the DBS will reach the nearest genetics laboratory the next day, free of charge to the patient and physician. However, national regulations in Russia need to be put in place to allow genetic testing, and reagents and diagnostic test equipment need to be officially registered with the government.
3.4 Patient education/communication
In many developing countries, low educational levels and low per capita income may be important barriers to diagnosis, family genetic testing, and treatment access for patients with genetic diseases. Many patients do not have formal jobs, social security numbers, and/or health insurance and, therefore, cannot access treatment that is available through government health services. Low educational levels, particularly in rural areas, may present challenges for physicians when explaining the importance of diagnosing a rare genetic disease and when communicating with family members regarding the importance of family screening. Communication difficulties are often further complicated by religious issues that are often raised as reasons not to carry out genetic testing. Strategies to help physicians communicate with new patients, and to help patients communicate with their families are, therefore, important. Genetic counselors/medical geneticists are well placed to help when available, but are a scarce resource. In many cases, newly diagnosed patients may have a low level of understanding of the disease with which they have just been diagnosed. A simple leaflet or booklet to provide an overview of the key facts about Fabry disease and a diagram to illustrate the inheritance pattern would be invaluable (Figure 2). Having a standard set of appropriate questions may also be helpful to guide patients when they discuss their diagnosis with family members and attempt to identify other affected relatives. Social media may play a role in finding distant relatives and re-establishing contact. Education on the importance of genetic testing for rare genetic diseases may also be needed for general practice physicians, depending on the region/country, to overcome cultural barriers to testing, particularly in female relatives of patients.
4 HOW CAN THE IMPLEMENTATION OF FAMILY SCREENING/PEDIGREE ANALYSIS IN FABRY DISEASE BE IMPROVED?
4.1 Patients as advocates: the Argentinian experience
In Argentina, between 600 and 700 patients have been diagnosed with Fabry disease. The population is spread over a wide geographical area, and communication with patients is challenging due to poor mobile phone network coverage and internet connectivity. Patients also fear the consequences of telling their families that they have a genetically inherited disease. Patient associations are vital cornerstones of communication with the Argentinian Fabry population, and there are two national Fabry disease patient associations that convene annually at one large patient meeting. Two genetic counselors are also available to support patients diagnosed with Fabry disease in Argentina. Patients are an essential part of the family screening process for Fabry disease in Argentina, and are utilized both as advocates within their family for genetic testing and as a source of new information—each year, a questionnaire is completed by patients at the annual meeting to try and identify new cases within their families. The Argentinian experience highlights the vital role of patients in family screening: well-informed patients (i.e., the so-called “expert patients”) who understand their disease and its inheritance are motivated to find new family members who may be helped by earlier diagnosis and treatment.
4.2 Inter-speciality communication: key to improving family genetic testing—The Brazilian hemodialysis screening program
The kidneys are one of the main organ systems affected by Fabry disease, and nephrologists often play a key role in the screening for Fabry disease. Current guidelines for the management of chronic kidney disease (CKD) recommend screening for Fabry disease in male patients younger than 50 years of age who have CKD of unknown etiology, and in females with unexplained CKD, irrespective of age, who present with other potential manifestations of Fabry disease (Schiffmann et al., 2017). Moreover, as patients with end-stage renal disease need to attend dialysis centers three times/week for hemodialysis, nephrologists are well placed to discuss screening for Fabry disease in this at-risk population.
Two Brazilian screening studies for Fabry disease in male hemodialysis patients have been published that included subsequent family genetic analysis (Silva et al., 2016; Veloso et al., 2018). The first study reported the screening of 2,583 patients in 23 dialysis centers in Bahia, Brazil (Silva et al., 2016); 3 patients with Fabry disease were identified in the at-risk hemodialysis population (prevalence 0.12%), one of whom initially had a novel missense genetic variant of unknown significance (GVUS). Subsequent family analysis resulted in a confirmed diagnosis of Fabry disease in 5 relatives of the first index case, 18 relatives of the second index case, and 2 relatives of the third index case. The family tree analysis also helped to confirm the pathogenicity of the novel missense variant (p.C52F) by identifying a male relative with this variant who expressed the classic Fabry phenotype (Silva et al., 2016). The second publication reported a smaller screening study from a single dialysis center in Goiás, Brazil (Veloso et al., 2018). From a population of 108 male hemodialysis patients, 1 patient with Fabry disease was identified (prevalence 0.9%). This patient had a novel missense variant (p.G35V) and the classic Fabry phenotype. The patient had been receiving dialysis for 8 years, but had not previously been suspected of having Fabry disease. Family analysis identified a further nine affected family members. In this study, family tree analysis allowed two male relatives (nephews) and four female relatives (two daughters and two nieces) to be diagnosed at an earlier age than the index patient. These studies highlight the importance of family genetic testing as a tool to identify patients with Fabry disease at an earlier age, when the benefit of therapy may be greater, and to characterize the pathogenicity of a novel variant.
Multidisciplinary coordination is the recommended approach to optimize the management of Fabry disease (Ortiz et al., 2018). However, when specialist's referral for multidisciplinary care is not available or feasible, as is often the case in Brazil and other developing nations, nephrologists are well placed to help medical geneticists in coordinating the evaluation and treatment of patients with Fabry disease at all stages (i.e., from an initial diagnosis, through treatment and long-term follow-up).
4.3 Collaboration with the pharmaceutical industry: Colombian Fabry disease expert
A positive example of industry collaboration in the diagnosis of Fabry disease is seen in Colombia, which has a population of over 48 million inhabitants and a population density of 42 inhabitants per km2. Because of the population geography, patients with rare genetic diseases need to travel to major cities for diagnosis and treatment. Diagnosis of Fabry disease is difficult in Colombia; there are few medical geneticists in the country, and only one referral laboratory in the capital city, Bogota. To assist with family screening, a dedicated, industry-sponsored physician is available who can travel to patients and/or their relatives to assist with diagnosis and genetic testing. The role of this physician is to assist with diagnosis only—this healthcare provider is not allowed to decide on which treatment to use; treatment decisions must be made by a second, independent physician. Currently, there are 20 cases of index patients with Fabry disease in Colombia, and 18 families have agreed to have the help of the dedicated physician to find extra affected family members through family screening.
4.4 Role of the medical geneticist in family screening: insights from Mexico
Current recommendations state that all newly diagnosed patients with Fabry disease should be referred for genetic counseling to develop a family pedigree and receive advice on family screening (Germain, 2010; Germain, Fouilhoux, et al., 2019; Ortiz et al., 2018). However, in many countries, genetic counselors are not available, or are not legally recognized as healthcare professionals, and geneticists are expected to fulfill this role with training support provided by industry. In Mexico, for example, Fabry disease is significantly underdiagnosed and there are no genetic counselors. To improve the diagnosis of the disease in Mexico, a family tree program was initiated in 2015. As part of this program, medical geneticists received appropriate Fabry disease-specific training. To date, more than 10 geneticists have been included in the program and patients from 17 different states in Mexico are involved. Regular meetings are held to discuss the field experience of each geneticist in the program, to assess barriers/issues faced, and to create strategies to optimize the program. Forty families have participated in the family tree program to date, which has resulted in 93 new patients being identified since 2015, and 91% of all new cases of Fabry disease in Mexico have been diagnosed through the program (M. Kramis, personal communication). In addition to the lack of genetic counselors, there are other barriers to the implementation of family screening in Mexico: diverse and complex family structures, a possible low educational level within some subsets of the population, and a widely dispersed rural population. There is also a low awareness and minimal knowledge of Fabry disease among many Mexican physicians, and a reluctance to study, diagnose, and treat it. Additionally, there is a high prevalence of chronic degenerative diseases in Mexico, which can contribute to a high potential for misdiagnosis of Fabry disease. The medical geneticist in Mexico, therefore, has not only a role in the diagnosis of new patients with rare genetic diseases such as Fabry disease, but also a responsibility to promote knowledge and awareness of rare diseases overall. The Mexican family tree program highlights how structured collaboration between geneticists and the pharmaceutical industry can improve both diagnosis of the disease and communication with/between patients and their families.
5 IMPORTANCE OF FAMILY GENETIC TESTING FOR EARLIER DIAGNOSIS LEADING TO POTENTIAL EARLIER TREATMENT
Early diagnosis of a treatable rare genetic disease is important, as starting treatment early can improve outcomes. For example, in patients with the attenuated form of mucopolysaccharidosis type I, early diagnosis and initiation of ERT can substantially modify the development of symptoms and improve long-term outcomes (Gabrielli et al., 2016). ERT can also be used to treat patients with Fabry disease (Eng et al., 2001; Germain, Elliott, et al., 2019; Schiffmann et al., 2001) and—a more recent development—chaperone therapy (Germain & Fan, 2009; Germain et al., 2016) can be given to a subset of patients when both amenability and pathogenicity of their GLA variants have been thoroughly studied and confirmed. Early initiation of treatment leads to better outcomes compared with treatment started later, once patients have more extensive and irreversible organ damage (Germain et al., 2015; Germain, Elliott, et al., 2019; Ortiz et al., 2016; Wanner et al., 2018; Warnock et al., 2012).
6 CONCLUSIONS
Family genetic testing of index patients identified through symptom presentation or as part of a screening program can greatly increase the number of patients diagnosed with rare genetic diseases and facilitate earlier diagnosis, the latter being particularly important for treatable genetic disorders. This review of the published scientific literature on family genetic testing for Fabry disease supports the value of thorough pedigree drawing and analysis for all newly diagnosed patients and genetic testing of all potentially affected individuals in their family tree. Indeed, this review shows that detailed pedigree analysis of a newly diagnosed patient can identify on average 4.8 additional affected relatives. A similar number has also been reported in a U.S. study (Laney & Fernhoff, 2008), however, it may likely vary depending on family size. Family size depends on cultural and ethnic backgrounds, although the literature search failed to identify other numbers in different ethnic backgrounds. Family genetic testing can also identify affected individuals at a younger age than would occur through clinical presentation, thus facilitating early treatment. The combination of screening protocols with pedigree drawing and cascade genotyping is also important, as screening programs detect a significant percentage of non-pathogenic GLA variants (Ferreira et al., 2015) or variants of unknown significance (Doheny et al., 2018; Germain, Fouilhoux, et al., 2019; Germain & Jurca-Simina, 2018; van der Tol et al., 2014). In this context, familial segregation studies of novel GLA variants (Germain et al., 1996) can provide information on the likely pathogenicity of a GLA variant, and avoid costs associated with over-diagnosing patients with benign GLA variants (Germain, Fouilhoux, et al., 2019).
Currently, the use of family screening in Fabry disease is suboptimal due to many regional and national barriers, including the cost of establishing and running a family screening program, cultural and societal issues such as patriarchal societies, complex family structures, regional issues related to population geography, the educational level of the population, and the lack of trained medical geneticists. Initiatives that may help to improve the use of family genetic testing in Fabry disease could include programs focused on educating physicians via presentations at international congresses or as part of online seminars/webinars with live questions and answers (this may be especially important during the COVID-19 pandemic; Laney et al., 2020), design of simpler software tools to help draw digital family trees, and creation of patient educational booklets or online tools to (a) assist communication with patients and (b) facilitate communication between patients and their family members, helping them to explain the need for family screening and overcome the stigma of a rare disease diagnosis.
ACKNOWLEDGMENTS
The advisory board meeting was organized and funded by Sanofi Genzyme. The attendees recommended that the information that was shared and discussed would be useful to inform healthcare professionals and suggested preparation of a publication. The authors received editorial/writing support in the preparation of this manuscript from Tom Rouwette, PhD, of Excerpta Medica, funded by Sanofi Genzyme, but no payment for writing this publication. The authors are responsible for the content of this manuscript and the decision to submit the manuscript for publication.
CONFLICTS OF INTEREST
D.P. Germain has received honoraria and consulting fees from Amicus Therapeutics, Sanofi Genzyme, and Takeda. S. Moiseev has received travel grants and/or fees for participation in advisory boards from Sanofi Genzyme and Takeda. G. Altarescu has received speaker honoraria from Amicus Therapeutics, Sanofi Genzyme, and Takeda. F.C. Barreto has received speaker honoraria from Sanofi Genzyme. I. Maksimova is an employee of Sanofi Genzyme. M. Kramis has received honoraria as scientific advisor and speaker from Sanofi Genzyme. S. Nampoothiri has received honoraria for travel and attending advisory boards from Sanofi Genzyme. D-M. Niu has received research funding from Sanofi Genzyme and Takeda. J. Politei has received honoraria from Amicus Therapeutics, Sanofi Genzyme, and Takeda and consulting fees from Sanofi Genzyme and Takeda. S. Kutsev has received honoraria from Sanofi Genzyme. F. Suárez-Obando, F. Al Ismaili, F. Haddoum, L-S. Ro, and N. Chen have received honoraria for travel and advisory board attendance from Sanofi Genzyme. H. Al Khawaja, F. Hadipour, K.N. Nguyen, and D. Vu Chi declare no conflicts of interest.
AUTHOR CONTRIBUTIONS
D.P. Germain, S. Moiseev, F. Suárez-Obando, F. Al Ismaili, F.C. Barreto, F. Haddoum, F. Hadipour, I. Maksimova, M. Kramis, S. Nampoothiri, J. Politei, L-S. Ro, N. Chen, and S. Kutsev attended the advisory board. All authors interpreted the data and reviewed, commented on, and critically revised the manuscript for important intellectual content. D.P. Germain wrote the revised version of the manuscript. All authors approved the final version of the manuscript to be published. All authors are accountable for all aspects of the work, in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All authors had access to the data and accept responsibility for submitting the article for publication.
Open Research
DATA AVAILABILITY STATEMENT
The data that support the findings of this study are available from the corresponding author upon reasonable request.




