Hypoxia-inducible factor 2α drives nonalcoholic fatty liver progression by triggering hepatocyte release of histidine-rich glycoprotein
Potential conflict of interest: Nothing to report.
Supported by the European Union's Horizon 2020 research and innovation programme (634413, for the project Elucidating Pathways of Steatohepatitis, to E.B.); Associazione Italiana per la Ricerca sul Cancro (15274, to M.P.); the CariPLO Foundation (2011-0470, to E.A. and M.P.); and the University of Turin (to E.N. and M.P.). The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Abstract
Mechanisms underlying progression of nonalcoholic fatty liver disease (NAFLD) are still incompletely characterized. Hypoxia and hypoxia-inducible factors (HIFs) have been implicated in the pathogenesis of chronic liver diseases, but the actual role of HIF-2α in the evolution of NAFLD has never been investigated in detail. In this study, we show that HIF-2α is selectively overexpressed in the cytosol and the nuclei of hepatocytes in a very high percentage (>90%) of liver biopsies from a cohort of NAFLD patients at different stages of the disease evolution. Similar features were also observed in mice with steatohepatitis induced by feeding a methionine/choline-deficient diet. Experiments performed in mice carrying hepatocyte-specific deletion of HIF-2α and related control littermates fed either a choline-deficient L-amino acid–defined or a methionine/choline-deficient diet showed that HIF-2α deletion ameliorated the evolution of NAFLD by decreasing parenchymal injury, fatty liver, lobular inflammation, and the development of liver fibrosis. The improvement in NAFLD progression in HIF-2α-deficient mice was related to a selective down-regulation in the hepatocyte production of histidine-rich glycoprotein (HRGP), recently proposed to sustain macrophage M1 polarization. In vitro experiments confirmed that the up-regulation of hepatocyte HRGP expression was hypoxia-dependent and HIF-2α-dependent. Finally, analyses performed on specimens from NAFLD patients indicated that HRGP was overexpressed in all patients showing hepatocyte nuclear staining for HIF-2α and revealed a significant positive correlation between HIF-2α and HRGP liver transcript levels in these patients. Conclusions: These results indicate that hepatocyte HIF-2α activation is a key feature in both human and experimental NAFLD and significantly contributes to the disease progression through the up-regulation of HRGP production. (Hepatology 2018;67:2196-2214).
Abbreviations
-
- ALT
-
- alanine aminotransferase
-
- CCL2
-
- C-C-motif chemokine ligand 2
-
- CD
-
- cluster of differentiation
-
- CDAA
-
- choline-deficient L-amino acid–defined
-
- CLD
-
- chronic liver disease
-
- CSAA
-
- choline-sufficient L-amino acid–defined
-
- hHIF-2α–/–
-
- hepatocyte-specific deletion of HIF-2α
-
- HIF
-
- hypoxia-inducible factor
-
- HRGP
-
- histidine-rich glycoprotein
-
- IHC
-
- immunohistochemistry
-
- IL
-
- interleukin
-
- MCD
-
- methionine/choline-deficient
-
- NAFLD
-
- nonalcoholic fatty liver disease
-
- NASH
-
- nonalcoholic steatohepatitis
-
- α-SMA
-
- α-smooth muscle actin
-
- TNFα
-
- tumour necrosis factor-α
Nonalcoholic fatty liver disease (NAFLD), regarded as the hepatic manifestation of the metabolic syndrome, is becoming the most frequent chronic liver disease (CLD) worldwide, with a prevalence of up to 20%-30% in the general population and even higher among obese individuals and/or patients affected by type II diabetes mellitus.1-6 NAFLD can evolve (20%-30% of patients) into nonalcoholic steatohepatitis (NASH), characterized by hepatocyte injury and lobular inflammation that, in turn, can progress to fibrosis, cirrhosis, and hepatocellular carcinoma; but at present mechanisms underlying NAFLD progression are still incompletely characterized, and no validated therapy is available.1, 4-7
Literature data suggest that hepatic hypoxia can have a role in CLD progression and hepatocellular carcinoma development by sustaining both angiogenesis and fibrogenesis as well as, possibly, inflammatory response and autophagy, with hepatic myofibroblasts being able to both respond to hypoxia and to act in proangiogenic way.8-11 The cellular response to hypoxia is mainly operated by hypoxia-inducible factors (HIFs), evolutionarily conserved heterodimeric transcriptional factors consisting of an oxygen-sensitive α-subunit (HIF-1α or HIF-2α) and a stable, constitutively expressed β-subunit (HIF-1β).12-14 So far, data concerning the role of hypoxia and HIFs in NAFLD are quite limited,10 but morphological evidence indicates that liver hypoxia develops in parallel with NAFLD-mediated steatosis, particularly in perivenous areas, similarly to what has been described for ethanol-induced fatty liver.15, 16 Nonetheless, HIFs can be regulated also by oxygen-independent mechanisms such as mitochondrial dysfunction, reactive oxygen species generation, and endoplasmic reticulum stress.8-14 In their turn, HIF-1α and HIF-2α can modulate the cellular adaptive responses to hypoxia by up-regulating either common or, more often, distinct and nonoverlapping transcriptional programs. For instance, HIF-1α promotes glucose consumption and glycolysis, while lipid storage is mainly regulated by HIF-2α.12-14 These actions on cell metabolism are potentially relevant for the pathogenesis of NAFLD because hypoxic conditions have been reported to stimulate lipid storage and inhibit lipid catabolism through β-oxidation.10, 12, 13, 17 Although both HIF-1α and HIF-2α affect lipid metabolism, experimental studies indicate that HIF-2α activation leads to fatty liver by both up-regulating genes involved in fatty acid synthesis/uptake and lipid storage and down-regulating those involved in fatty acid catabolism.18, 19 HIF-2α activation has also been related to an early increase in the transcription of proinflammatory cytokines and of genes involved in fibrogenesis.19 However, these data have been obtained in mice carrying multiple genetic manipulations in the absence of liver injury18, 19 or in the frame of a short protocol (2 weeks) of ethanol administration using the Lieber-De Carli liquid diet, all conditions that do not reproduce the conditions occurring in NAFLD.19 At present, relevant human data about the role of HIF-2α in fatty liver development and NAFLD progression are lacking. On the other hand, despite the fact that HIF-1α appears to sustain fibrogenesis in the bile duct ligation experimental model of CLD,20 other studies designed to specifically target hepatocyte HIF-1α in experimental models of alcohol-related or NAFLD-related steatosis and progression have led to conflicting results.21-24
In this study, the analysis of liver biopsies from a cohort of NAFLD patients and the induction of experimental NAFLD with two different dietary protocols in mice carrying hepatocyte-specific deletion of HIF-2α provide evidence indicating that HIF-2α plays a critical role in NAFLD progression by up-regulating hepatocyte expression of histidine-rich glycoprotein (HRGP).
Materials and Methods
HUMAN SUBJECTS
The study on NAFLD patients was approved by the ethics committee of the Azienda Ospedaliera Universitaria Città della Salute (Turin, Italy). For this study we analyzed liver biopsies from NAFLD patients (n = 27) ranging from early disease (stage F0-F1) to more advanced conditions of fibrosis (stage F2-F3) or cirrhosis (F4) and referring to the Division of Gastro-Hepatology of the University of Turin. All samples were collected at the time of first diagnosis. Patients were characterized by anthropometric, clinical, and biochemical data; and liver biopsies were evaluated for the severity of steatohepatitis and fibrosis according to Kleiner et al.25 All subjects gave informed consent to the analysis, and the study protocol, which conformed to the ethical guidelines of the 1975 Declaration of Helsinki, was planned according to the guidelines of the local ethics committee. The clinical and biochemical features of the patients are reported in Supporting Table S1. Control human liver tissue (n = 5), as defined for normal histological structures in hematoxylin and eosin sections and the absence of inflammation in the portal tract and parenchyma, was obtained from diagnostic biopsies or from resection samples taken at a distance of more than 5 cm from the border of liver metastasis of colon carcinoma.
ANIMAL EXPERIMENTATION
Mice carrying a hepatocyte-specific deletion of HIF-2α (hHIF-2α–/–) were obtained by breeding HIF-2αfl/fl C57BL/6 mice with mice on the same genetic background expressing Cre-recombinase under the control of the albumin promoter (Alb/Cre+/+ mice; Jackson Laboratories, Bar Harbor, ME). Eight-week-old male hHIF-2α–/– mice and related control sibling littermates not carrying the HIF-2α deletion were fed either a methionine/choline deficient (MCD) diet for 4 or 8 weeks or a choline-deficient L-amino acid–defined (CDAA) diet (Laboratorio Dottori Piccioni, Gessate, Italy) for 12 and 24 weeks.26, 27 Control littermates received the corresponding methionine/choline-sufficient or the choline-sufficient L-amino acid–defined (CSAA) diet. In preliminary experiments 8-week-old normal C57BL/6 mice were fed the MCD diet or the methionine/choline-sufficient control diet for 4 and 8 weeks. The experiments complied with national ethical guidelines for animal experimentation, and the experimental protocols were approved by the Italian Ministry of Health.
BIOCHEMICAL ANALYSES
Plasma alanine aminotransferase (ALT) and liver triglycerides were determined by spectrometric kits supplied by Radim S.p.A. (Pomezia, Italy) and Sigma Diagnostics (Milan, Italy), respectively. Circulating interleukin 12 (IL-12) was evaluated by commercial enzyme-linked immunosorbent assay kits supplied by Peprotech (Milan, Italy).
IMMUNOHISTOCHEMISTRY, SIRIUS RED STAINING, AND HISTOMORPHOMETRIC ANALYSIS
Immunohistochemistry (IHC),28, 29 picro-sirius red staining, and histomorphometric analysis30, 31 were performed on paraffin-embedded human liver biopsies and/or murine liver specimens as reported. More details are in the Supporting Information.
QUANTITATIVE REAL-TIME PCR
RNA extraction, complementary DNA synthesis, and quantitative real-time PCRs were performed on human and murine liver specimens as described.28-30 mRNA levels were measured by quantitative real-time PCR, using the SYBR green method as described.28-30 More details and oligonucleotide sequences of primers used for quantitative real-time PCR are available in the Supporting Information. The murine transcripts for tumour necrosis factor-α (TNFα), cluster of differentiation 11b (CD11b), IL-12p40, C-C-motif chemokine ligand 2 (CCL2), C-X-C-motif chemokine ligand 10, HRGP, and β-actin liver RNA were retrotranscribed with the High Capacity cDNA Reverse Transcription Kit (Applied Biosystems Italia, Monza, Italy). Quantitative real-time PCR for these transcripts was performed in a Techne TC-312 thermacycler (TecneInc, Burlington, NJ) using TaqMan Gene Expression Master Mix and TaqMan Gene Expression probes for indicated murine genes (Applied Biosystems Italia) as described.26, 31 Human sample analysis was performed using SsoFast EvaGreen Supermix (Bio-Rad, Hercules, CA) following the manufacturer's instructions.
INTRAHEPATIC MONONUCLEATED CELL ISOLATION AND FLOW-CYTOMETRIC ANALYSIS
Hepatic mononucleated cells were isolated from the livers of naive and CDAA–fed mice and purified on a density gradient (Lympholyte-M; Cedarlane Laboratoires Ltd., Burlington, Canada) as described.31 Cells were washed with Hank's medium and incubated for 30 min with decomplemented mouse serum to block unspecific immunoglobulin binding. The cells were then stained with fluorochrome-conjugated antibodies for CD45, CD11b, Ly6C (eBiosciences, San Diego, CA), and F4-80 (Invitrogen, Abingdon, UK) and analyzed with a Attune NxT acoustic focusing cytometer (Thermo Fischer Scientific, Waltham, MA) following prior gating for CD45 and the absence of cell aggregates. Intracellular staining for IL-12 was performed using a specific fluorochrome-conjugated antibody (eBiosciences).
IN VITRO EXPERIMENTS AND WESTERN BLOT ANALYSIS
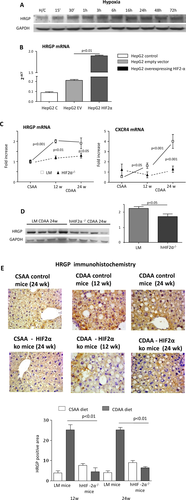
In vitro experiments in this study were performed in normal HepG2 cells (American Type Culture Collection) as well as in cells stably transfected in order to overexpress HIF-2α in either normoxic or hypoxic conditions, as reported.28 Western blot analysis was performed on total cell or tissue lysates as described.28, 30 More details on in vitro studies and western blot analysis are available in the Supporting Information.
DATA ANALYSIS AND STATISTICAL CALCULATIONS
Statistical analyses were performed by SPSS statistical software (SPSS Inc., Chicago, IL) using one-way analysis of variance with Tukey's correction for multiple comparisons or the Kruskal-Wallis test for nonparametric values. Significance was taken at the 5% level. Normality distribution was preliminarily assessed by the Kolmogorov-Smirnov algorithm.
Results
HIF-2α IS OVEREXPRESSED IN HUMAN AND MURINE HEPATOCYTES IN NAFLD-RELATED LIVER SPECIMENS
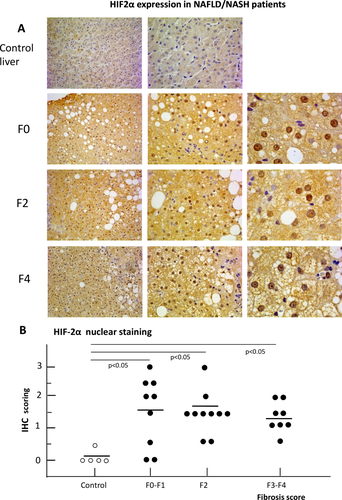
IHC analysis of liver biopsies from a cohort of well-characterized NAFLD patients (n = 27, see Supporting Table S1 for patient characterization) at different stages of the disease evolution showed an evident increase of HIF-2α immune-staining compared to undamaged liver samples (n = 5) from patients undergoing resection for hepatic metastasis of colon carcinoma. In the majority of NAFLD patients (25 out of 27, 92%) HIF-2α positivity was detectable almost exclusively in hepatocytes (Fig. 1A,B) and involved both the cytoplasm and the nuclei, although the number of positive nuclei varied within patients and in different areas of the same biopsy (Fig. 1A). The nuclei of nonparenchymal cells, mainly inflammatory cells or myofibroblast-like cells in fibrotic septa, were essentially negative for HIF-2α. Occasionally, HIF-2α staining was observed in portal/periportal cholangiocytes, particularly in subjects with advanced fibrosis. Semiquantitative assessment of HIF-2α-positive hepatocyte nuclei confirmed a significant increase among NAFLD patients irrespective of the stage of the disease (Fig. 1B). These findings were reproduced in a rodent model of NAFLD-related liver fibrosis based on mice fed the MCD diet up to 8 weeks (Supporting Fig. S1A,B). In fact, while in control mice HIF-2α staining was only detected in the cytoplasm of hepatocytes closely surrounding centrilobular veins (Supporting Fig. S1A), extensive HIF-2α positivity was evident in liver sections from mice receiving the MCD diet (Supporting Fig. S1B). In these animals quantitative real-time PCR analyses confirmed that both HIF-2α as well HIF-1α transcripts were up-regulated in mice with NAFLD, but only those of HIF-2α paralleled the disease progression (Supporting Fig. S1C-F).
HEPATOCYTE-SPECIFIC DELETION OF HIF-2α IMPROVES NAFLD-ASSOCIATED LIVER INJURY, STEATOSIS, INFLAMMATORY RESPONSE, AND FIBROSIS
Based on previous data, we evaluated whether HIF-2α deletion in hepatocytes might modify NAFLD evolution. To this aim, progressive NAFLD was induced in hHIF-2α–/– mice as well as related control littermates by two dietary protocols using the MCD diet, administered for 4 and 8 weeks, and the CDAA diet, administered for 12 and 24 weeks. In preliminary experiments, we first checked for effective HIF-2α depletion in the livers of hHIF-2α–/– mice receiving the MCD diet. Quantitative real-time PCR analysis revealed a very significant down-regulation of liver HIF-2α transcripts in hHIF-2α–/– mice compared to control littermates (Supporting Fig. S2A-C), whereas HIF-1α and vascular-endothelial growth factor transcript levels were not affected. HIF-2α deletion was also confirmed at the protein level by western blotting of total liver proteins (Supporting Fig. S2D).
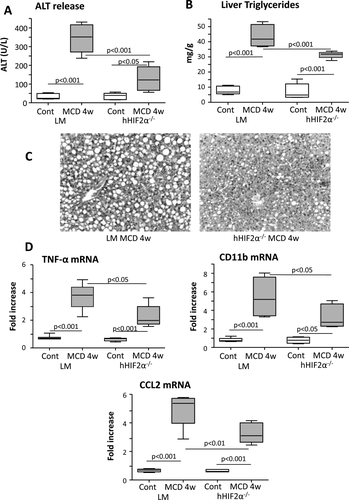
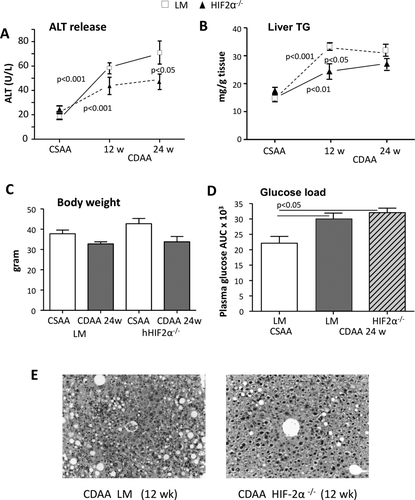
Interestingly, in hHIF-2α–/– mice receiving the MCD diet for 4 weeks the severity of steatohepatitis as evaluated by histology, ALT release, and hepatic triglyceride accumulation was significantly lower than in HIF-2α-sufficient animals (Fig. 2A-C). The extent of lobular inflammation and the hepatic expression of inflammatory markers TNFα, CD11b, and CCL2 were also decreased in hHIF-2α–/– mice receiving the MCD diet (Fig. 2D). On the same line, using CDAA-fed mice we observed that the deletion of hepatocyte HIF-2α (which was confirmed also by means of IHC in these mice versus control littermates; see Supporting Fig. S3) slowed down the time-dependent progression of NAFLD (Fig. 3A,B,E) without affecting the gain in body weight and the development of insulin resistance (Fig. 3C,D). In this experimental model, hHIF-2α–/– mice also showed less liver fibrosis at both 12 and 24 weeks, as evidenced by sirius red staining (Fig. 4A,B). This paralleled a decrease in hepatic transcript for transforming growth factor β1, α-smooth muscle actin (α-SMA), and collagen 1A1 (Fig. 4C), as well as in the number of α-SMA-positive myofibroblasts detected by IHC (Fig. 4D). Remarkably, the protection from fibrosis was also evident in hHIF-2α–/– mice receiving the MCD diet for up to 8 weeks compared to control HIF-2α-sufficient littermates (Supporting Fig. S4A-D).
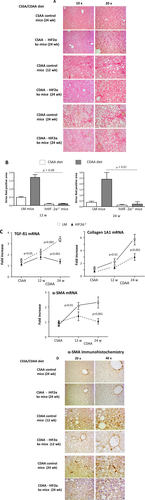
HEPATOCYTE-SPECIFIC DELETION OF HIF-2α PREVENTS THE PROGRESSION OF EXPERIMENTAL NAFLD BY AFFECTING HEPATOCYTE-DEPENDENT RELEASE OF HRGP
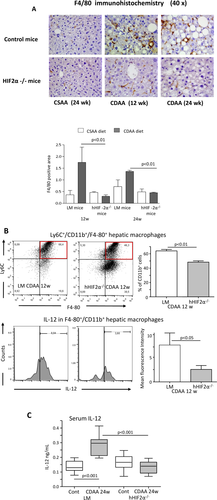
To address the mechanisms by which hepatocyte HIF-2α up-regulation promotes inflammatory mechanisms in NAFLD, we focused our attention on the role of HRGP, a hepatocyte-released mediator (i.e., hepatokine) that has been recently shown to support liver macrophage activation in experimental and clinical NAFLD/NASH.32 To investigate the possible hypoxia-dependent and HIF-2α-dependent modulation of HRGP, we employed HepG2 cells, which are known to rapidly respond to hypoxia with HIF-2α stabilization and the up-regulation of HIF-2α target genes.28, 33 Incubation of HepG2 cells under hypoxic conditions, which is known to rapidly promote the nuclear translocation of HIF-2α,28 was associated with an appreciable increase in HRGP synthesis (Fig. 5A). A very significant up-regulation of HRGP transcription was also observed in HepG2 cells transfected with an HIF-2α-containing vector but not with an empty vector (Fig. 5B), confirming an HIF-2α-dependent modulation of HRGP in liver cells. According to the in vitro data, hepatic HRGP transcript levels increased in a time-dependent manner in control mice fed the CDAA diet in parallel with the up-regulation of the expression of another HIF-2α-dependent gene such as cysteine-X-cysteine receptor 4 (Fig. 5C). Conversely, HRGP up-regulation associated with NAFLD evolution was almost completely prevented at both the mRNA and protein levels in hHIF-2α–/– mice (Fig. 5C,D). The relationships between HIF-2α and HRGP were further confirmed by the evidence that HepG2 stably transfected to overexpress HIF-2α also overexpressed transcript levels of other HIF-2α target genes such as cysteine-X-cysteine receptor 4 and erythropoietin (Supporting Fig. S5A,B) or, as previously reported, SerpinB3.28 Moreover, HRGP transcript and protein levels were down-regulated in HepG2 cells exposed to hypoxic conditions following efficient silencing for HIF-2α (Supporting Fig. S5C,D). IHC performed on liver from mice fed the CDAA diet confirmed that HRGP was selectively expressed by hepatocytes of control mice with steatohepatitis but not in those from hHIF-2α–/– animals (Fig. 5E). Up-regulation of HIF-2α and HRPG was unrelated to choline deficiency and was also observed in mice with NAFLD induced by administration in the drinking water of a high-fat/fructose diet (Supporting Fig. S6A-D). Hepatocyte-derived HRGP has been recently shown to promote liver inflammation by stimulating M1 polarization of hepatic macrophages.32 In our hands, the impaired production of HRPG in mice receiving the CDAA diet decreased the liver infiltration by F4/80-positive macrophages as evidenced by both IHC (Fig. 6A) and flow cytometry (Supporting Fig. S7A). Furthermore, the fraction of inflammatory Ly6Chigh hepatic macrophages and their capacity to produce IL-12 were significantly lower in hHIF-2α–/– mice compared to control littermates (Fig. 6B). Consistently the hepatic expression of M1 cytokines/chemokines TNFα, IL-12p40, CCL2, and C-X-C-motif chemokine ligand 10 (Supporting Fig. S7B); and the serum levels of IL-12 were also significantly reduced in hHIF-2α–/– mice receiving the CDAA diet (Fig. 6C), indicating a possible role of HRGP in the mechanisms by which the stimulation of HIF-2α in parenchymal cells supports proinflammatory responses of hepatic macrophages.
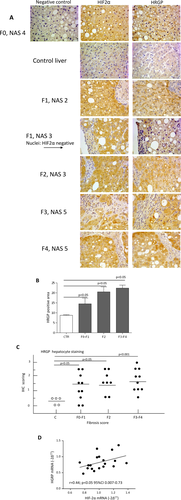
Finally, to further support the possible relevance of the relationships between HIF-2α and HRGP in the human disease, we analyzed for HIF-2α and HRGP immune-staining serial sections (4 μm thick) in our cohorts of NAFLD patients. Figure 7A shows that HRGP positivity was very low in sections from control livers. Conversely, in agreement with previous data,32 HPRG immune-staining was evident and increased in liver samples from NAFLD patients irrespective of the disease evolution (Fig. 7B,C) and colocalized with that of HIF-2α (Fig. 7A). Furthermore, we found a significant positive correlation between HIF-2α and HRGP mRNA levels in those patients of this cohort (n = 19) for whom frozen liver specimens were available (Fig. 7D).
Discussion
As mentioned in the introduction, hypoxia and HIFs have been proposed to play a role in the progression of CLD.8-11 However, data concerning their role in the evolution of NAFLD are quite limited,10 and, more specifically, the contribution of HIF-2α has never been investigated in detail. In the present study we provide evidence that hepatocyte HIF-2α is up-regulated in either human or experimental NAFLD and propose a mechanism by which HIF-2α activation in the liver parenchyma can contribute to the disease progression.
We report that HIF-2α is specifically overexpressed in hepatocytes during the development of human NAFLD. In the majority of the liver biopsies from NAFLD patients, nuclear localization of HIF-2α is also evident, indicating that, by forming a heterodimer with either HIF-1β or HIF-2β, HIF-2α can act as a transcription factor. Interestingly, HIF-2α activation is evident already in the early stage of the disease (F0-F1), and it is maintained up to more advanced (F3-F4) conditions of fibrosis/cirrhosis. Similar findings have been detected in murine models of NAFLD. In this setting, both HIF-2α and HIF-1α transcripts were up-regulated in mice with steatohepatitis, but only those of HIF-2α paralleled the disease progression. Altogether, these data suggest that HIF-dependent responses are an early event in NAFLD evolution and can be involved in the disease progression. Hepatic hypoxia likely represents the most obvious cause for HIF-2α up-regulation in both human and experimental NAFLD. In fact, diffuse lobular staining with the hypoxia-sensitive dye pimonidazole has been documented in mice with NAFLD.21 Nevertheless, we cannot exclude that hypoxia-independent mechanisms, for instance, oxidative stress, might also contribute to stimulate HIF-2α activity.8-11
The actual relevance of HIF-2α in NAFLD progression has been mechanistically confirmed by inducing NAFLD with the MCD or CDAA diet in hHIF-2α–/– mice. We have observed that steatosis, parenchymal injury, inflammatory response, and liver fibrosis are appreciably attenuated in these animals compared with control littermates. The decrease in fatty liver detected in hHIF-2α–/– mice is consistent with the observation that HIF-2α activation is per se capable of promoting fatty liver by up-regulating genes involved in fatty acid synthesis/uptake and lipid storage and by down-regulating those involved in fatty acid catabolism.18, 19 Accordingly, in the CDAA diet model, transcript levels of sterol regulatory element binding protein 1 and of fatty acid synthase were significantly reduced in hHIF-2α–/– mice versus related control littermates exposed to the same diet (Supporting Fig. S8). Moreover, employing murine hepatocytes (AML12 line), we found that exposure of these cells to palmitic acid (i.e., a condition leading to lipid accumulation and mimicking “in vitro” lipotoxicity) resulted in a significant up-regulation of both HIF-2α and HRGP protein levels (Supporting Fig. S9A) and of HRGP transcript levels (Supporting Fig. S9B), suggesting that lipotoxicity may represent an additional condition leading to HIF-2α and HRGP up-regulation.
Furthermore, in a previous study using mice fed a high-fat diet, hepatocyte deletion of HIF-1α effectively prevented NAFLD-associated liver fibrosis without interfering with fat accumulation.21 Interestingly, the same work also showed that hepatocyte HIF-1α deficiency moderately affected parenchymal damage and did not influence the extent of lobular inflammation.21 This indicates that beside the action on steatosis, the activation of HIF-2α in hepatocytes has a key role in favoring NAFLD-associated cell injury and inflammatory responses.
To investigate whether hepatocyte HIF-2α might stimulate the production of mediators with proinflammatory action, we focused attention on the possible role of HRGP, a multifunctional hepatocyte-derived circulating protein34 that has been recently shown to be involved in modulating proinflammatory activity of macrophages in tumors as well during chronic liver injury.32, 35 In particular, Bartneck et al.32 have reported that HRGP deletion leads to a significant protection from liver injury and fibrosis in mice with MCD-induced NASH by down-modulating the recruitment of hepatic macrophages and their M1 polarization. In this setting, our data demonstrate an interplay between HIF-2α and HRGP during NAFLD evolution by showing that the lack of hepatocyte HIF-2α prevents HRGP up-regulation in mice fed the CDAA diet. Furthermore, HRGP staining of liver biopsies from NAFLD patients colocalizes with that of HIF-2α, while in the same subjects a positive linear correlation is evident between the HIF-2α and HRGP transcripts. These findings are supported by in vitro experiments employing HepG2 cells exposed to hypoxia or overexpressing HIF-2α that confirm the strict HIF-2α dependence of HRGP production.
In line with the view that HIF-2α deficiency can reduce hepatic inflammation by preventing HRGP-mediated support to M1 activation of liver macrophages, we have observed that the liver expression of M1 markers TNF-α, IL-12, and C-X-C-motif chemokine ligand 10 is blunted in hHIF-2α–/– mice with NAFLD. Moreover, flow cytometry reveals that the prevalence of proinflammatory Ly6Chigh hepatic macrophages and their capacity to produce IL-12 are lowered in hHIF-2α–/– mice fed the CDAA diet. The HIF-2α influence on macrophage responses is of potential general relevance because it is well known that the polarization of hepatic macrophages is a crucial determinant for the progression of CLDs.36 In particular, the macrophage M1 shift has been reported to correlate with the severity of both alcoholic and nonalcoholic liver disease.37, 38 Accordingly, M2 skewing is associated with a more favorable histology and fewer hepatic lesions in patients with ethanol-related CLD37 as well as in HRGP–/– mice exposed to chronic liver injury.32
The involvement of HRGP in mediating the effects of HIF-2α activation during NAFLD progression does not rule out the possible involvement of other mediators. We previously reported that HIF-2α activation in human liver cancer cells up-regulates Serpin B3 and that this contributes to the development of fibrosis during CLD by sustaining the most relevant profibrogenic responses by activated stellate cells.28, 30 Unfortunately, unlike in humans, SerpinB3 is virtually undetectable in murine livers during CLD; thus, in the present study we did not have the chance to evaluate its contribution to NAFLD progression. However, based on the association between circulating levels of Serpin B3 and the risk of fibrosis in patients with chronic hepatitis C,39 a possible role of Serpin B3 in the evolution of human NAFLD cannot be excluded.
In conclusion, our data indicate that hepatocyte HIF-2α activation during the evolution of human and murine NAFLD has a role in disease progression by mediating the up-regulation of HRGP expression, which in turn critically influences the severity of steatohepatitis and fibrogenesis. These findings point to HIF-2α and HIF-2α-dependent genes as putative targets for future therapeutic strategies in NAFLD, a disease that, at present, has no validated therapy.
REFERENCES
Author names in bold designate shared co-first authorship.