Prognostic and predictive impact of metastatic organ involvement on maintenance therapy in advanced metastatic colorectal cancer: Subgroup analysis of patients treated within the PanaMa trial (AIO KRK 0212)
Parts of this manuscript were presented (poster presentation) at the ASCO Gastrointestinal Cancers Symposium 2023 (Abstract 127).
Abstract
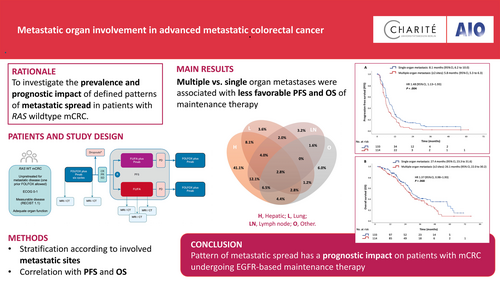
Despite molecular selection, patients (pts) with RAS wildtype mCRC represent a heterogeneous population including diversity in metastatic spread. We investigated metastatic patterns for their prognostic and predictive impact on maintenance therapy with 5-fluorouracil/folinic acid ± panitumumab. The study population was stratified according to (1) number of involved metastatic sites (single vs multiple organ metastasis), liver-limited disease vs (2) liver metastasis plus one additional site, and (3) vs liver metastasis plus ≥two additional sites. Kaplan-Meier method and Cox regressions were used to correlate efficacy endpoints. Single organ metastasis was observed in 133 pts (53.6%) with 102 pts (41.1%) presenting with liver-limited disease, while multiple organ metastases were reported in 114 pts (46.0). Multiple compared to single organ metastases were associated with less favorable PFS (HR 1.48, 95% CI 1.13-1.93; P = .004) and OS (HR 1.37, 95% CI 0.98-1.93; P = .068) of maintenance therapy. While metastatic spread involving one additional extrahepatic site was not associated with clearly impaired survival compared to liver-limited disease, pts with liver metastasis plus ≥two additional sites demonstrated less favorable PFS (HR 1.92, 95% CI 1.30-2.83; P < .001), and OS (HR 2.38, 95% CI 1.51-3.76; P < .001) of maintenance therapy. Pmab-containing maintenance therapy appeared active in both pts with multiple (HR 0.58; 95% CI, 0.39-0.86; P = .006) as well as to a lesser numerical extent in pts with single organ metastasis (HR 0.83; 95% CI, 0.57-1.21; P = .332; Interaction P = .183). These data may support clinical decisions when EGFR-based maintenance therapy is considered.
Graphical Abstract
What's new?
A variety of patterns in metastasis can occur in RAS wildtype metastatic colorectal cancer (mCRC). Heterogeneity in metastatic spread, however, challenges prognostic evaluation for patients with these tumors. Here, the prognostic and therapeutic significance of different metastatic patterns in RAS wildtype mCRC was investigated in patients on maintenance therapy with 5-fluorouracil/folinic acid, with or without panitumumab. In patients who presented with liver-limited disease, spread to one additional organ had limited impact on survival. Survival was less favorable for metastasis to multiple organs. Maintenance therapy involving panitumumab had greater effect in RAS wildtype mCRC patients with multiple organ metastasis.
Abbreviations
-
- 5-FU
-
- 5-fluorouracil
-
- BRAF
-
- B-Raf murine sarcoma viral oncogene homolog
-
- EGFR
-
- Epidermal Growth Factor Receptor
-
- FOLFOX
-
- combination chemotherapy with 5-flourouracil, leucovorin (FU/FA), oxaliplatin
-
- FU/FA
-
- 5-fluorouracil/folinic acid
-
- mCRC
-
- metastatic colorectal cancer
-
- OS
-
- overall survival
-
- PFS
-
- progression-free survival
-
- Pmab
-
- panitumumab
-
- RAS
-
- Rat sarcoma viral oncogene
-
- RECIST
-
- response evaluation criteria in solid tumors
-
- WT
-
- wild type
1 INTRODUCTION
In patients with initially unresectable metastatic colorectal cancer (mCRC), anatomic location of the primary tumor and RAS/BRAF mutational status are used to define treatment strategies.1-6 However, despite the present molecular characterization, the prediction of survival outcomes of metastatic colorectal cancer remains challenging given the remaining biological diversity of the molecular subgroups, and also the heterogeneous metastatic spread within molecularly defined subgroups.1, 7
Metastatic spread to more than one organ has previously been described to worsen survival outcomes in mCRC patients serving as an established prognostic marker.8-10 Likewise, anatomic site and spread of metastases present major drivers of treatment decisions in the management of oligometastatic disease, which is vaguely characterized by limited number of lesions and organs involved.1 The most common site of distant metastasis in mCRC patients is the liver followed by the lung.11 Metastases outside the most commonly affected organs, such as peritoneum, lymph nodes and brain, have been associated with impaired survival in mCRC patients.12-14
As metastatic spread can be easily characterized in the context of routinely performed radiological imaging and provides important information, such as tumor burden, it is commonly used for clinical decision-making, for example, primary or secondary resection of metastases, and antitumor treatment intensity.1, 7, 15-25 Apart from the initial choice of therapy, interventional treatment modalities and the promotion of maintenance therapy are essential parts of a first-line treatment strategy.1, 7
Patients treated in the PanaMa trial received six cycles of 5-flourouracil, leucovorin (FU/FA), oxaliplatin (FOLFOX) and the anti-EGFR antibody panitumumab (Pmab) before randomization (if at least stable disease was observed) into FU/FA plus Pmab vs FU/FA alone.26 PFS of maintenance therapy was the primary endpoint of this trial. This exploratory analysis aims to investigate the prevalence of defined patterns of metastatic spread in patients with RAS wildtype mCRC (number of involved organs focusing on hepatic vs hepatic plus extrahepatic metastasis). Furthermore, the impact of these patterns on the prognosis of patients is evaluated. Lastly, this analysis asks the question if the pattern of metastatic spread interacts with the efficacy of Pmab during maintenance therapy. As this analysis was conducted as a retrospective evaluation of the trial, the results should be interpreted as hypothesis generating.
2 METHODS
2.1 The PanaMa trial
Study design, treatment characteristics, and main inclusion and exclusion criteria of the PanaMa trial (AIO KRK 0212) have been reported previously.26 In brief, patients with RAS WT mCRC received maintenance treatment with either FU/FA plus Pmab or FU/FA alone following first-line induction therapy with six cycles of FOLFOX plus Pmab. PFS of maintenance therapy was the primary endpoint of this trial defined as time from randomization until disease progression or death, whatever occurred first. Patients with RAS WT mCRC, who had an Eastern Cooperative Oncology Group (ECOG) performance status of 0-1, no previous treatment for metastatic disease (one prior application of FOLFOX allowed), measurable disease according to RECIST 1.1,27 and adequate organ function were eligible.
2.2 Assessments and endpoints
Thoracic and abdominal computed tomography (CT) and magnetic resonance imaging (MRI) scans were scheduled within 21 days before first application of induction therapy (baseline) while reassessments were performed after completion of six cycles of induction therapy (corresponding to 12 weeks) and every 8 weeks during maintenance therapy. Metastatic organ involvement was assessed by the local investigator and documented in the electronic case report form (eCRF).
To evaluate prognostic and predictive impact of metastatic organ involvement, information on metastatic site (liver, lung, lymph node, other) as indicated by the local investigator was analyzed with regard to the frequency of mentions in order to define groups large enough for evaluation. For the present analysis, patients were stratified for (1) number of involved metastatic sites (single vs multiple organ metastasis), (2) liver-limited disease vs liver metastasis plus one additional organ, and (3) liver-limited disease vs liver metastasis plus two additional organs. For this analysis, we refrained from including metastatic patterns other than the ones aforementioned owing to limited sample sizes.
PFS of maintenance therapy was defined as time from randomization to first disease progression (according to RECIST 1.1, assessed by the local investigator) or death from any cause. OS of maintenance therapy was defined as time from randomization to death from any cause. Patients without progression or death were censored at the last day of documented progression-free/alive follow-up.
2.3 Statistical analysis
Time-to-event endpoints (PFS and OS) were expressed by Kaplan-Meier method and compared by log-rank testing. Hazard ratios (HR) with 95% confidence intervals (CI) were estimated using Cox regression models. Group differences between both treatment arms were tested nonparametrically (two-sided chi-square and Mann-Whitney U tests for unpaired samples) with a significance level of <.05. Predictive value of metastatic pattern and spread on PFS and OS of maintenance therapy were evaluated by Cox regression models and displayed by forest plots for each subgroup according to treatment arm (FUFA+ Pmab vs FUFA alone). Cases with missing data were omitted and remaining data were analyzed (listwise deletion). All statistical analyses were performed using IBM SPSS Statistics for Macintosh, Version 28.0 (Armonk, NY: IBM Corp).
3 RESULTS
3.1 Patient population
Within the PanaMa trial, a total of 248 patients underwent randomization to receive maintenance therapy with either FU/FA+ Pmab (n = 125) or FU/FA alone (n = 123) after induction therapy with six cycles of FOLFOX+ Pmab (full analysis set). Of those, three comparative groups were defined for subgroup analysis of metastatic pattern: (1) single organ metastasis (n = 133, 53.6%) vs multiple organ metastasis (n = 114, 46.0%), (2) liver-limited disease (n = 102, 41.1%) vs liver metastasis plus one additional organ (n = 61, 24.6%), and (3) liver-limited disease (n = 102, 41.1%) vs liver metastasis plus ≥two additional organs (n = 40, 16.1%) (Figure 1).
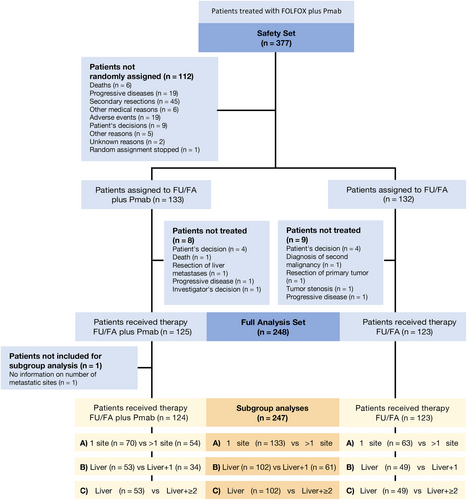
3.2 Baseline characteristics
Patient and tumor characteristics were evaluated according to single vs multiple organ disease and maintenance treatment arm (FU/FA+ Pmab vs FU/FA alone). While sex, age, and onset of metastases (synchronous vs metachronous) were generally balanced between groups, multiple compared to single organ disease was associated with a higher percentage of right-sided primary tumors as well as a reduced performance status in patients. Prior treatment (surgery of primary tumor, and adjuvant oxaliplatin-containing therapy) was reported more often for the Pmab-containing maintenance treatment arm while overall adjuvant chemotherapy appeared to have been applied more often in patients with multiple organ disease (Table 1).
| Single organ disease | Multiple organ disease | |||
|---|---|---|---|---|
| Characteristic | FU/FA+ Pmab | FU/FA | FU/FA+ Pmab | FU/FA |
| (n = 70) | (n = 63) | (n = 54) | (n = 60) | |
| Sex, No. (%) | ||||
| Male | 47 (67.1) | 39 (61.9) | 39 (72.2) | 39 (65.0) |
| Female | 23 (32.9) | 24 (38.1) | 15 (27.8) | 21 (35.0) |
| Age, years | ||||
| Median (range) | 65.5 (45-84) | 65.0 (48-85) | 66.5 (44-81) | 65.0 (30-86) |
| ECOG PS, No. (%) | ||||
| 0 | 49 (70.0) | 39 (63.9) | 21 (38.9) | 35 (58.3) |
| 1 | 21 (30.0) | 22 (36.1) | 33 (61.1) | 25 (41.7) |
| Missing information | – | 2 | – | – |
| Site of primary tumor, No. (%) | ||||
| Left-sided | 58 (82.9) | 56 (88.9) | 41 (75.9) | 44 (73.3) |
| Right-sided | 9 (12.9) | 7 (11.1) | 10 (18.5) | 12 (20.0) |
| Both or unknown | 3 (4.3) | – | 3 (5.6) | 4 (6.7) |
| Onset of metastases, No. (%) | ||||
| Synchronous | 56 (80.0) | 50 (79.4) | 44 (81.5) | 49 (81.7) |
| Metachronous | 14 (20.0) | 13 (20.6) | 10 (18.5) | 11 (18.3) |
| Prior treatment, No. (%) | ||||
| Surgery of primary tumor | 53 (75.7) | 43 (68.3) | 40 (74.1) | 40 (66.7) |
| Adjuvant chemotherapy | 8 (11.4) | 5 (7.9) | 8 (14.8) | 11 (18.3) |
| Adjuvant oxaliplatin | 4 (5.7) | 1 (1.6) | 6 (11.1) | 3 (7.9) |
| Radiotherapy | 8 (11.4) | 6 (9.5) | 6 (11.1) | 8 (13.3) |
| Response to induction, No. (%) | ||||
| ORR | 54 (77.1) | 50 (79.4) | 46 (85.2) | 49 (81.7) |
| SD | 16 (22.9) | 13 (20.6) | 8 (14.8) | 11 (18.3) |
| Response to maintenance, No. (%) | ||||
| ORR | 33 (47.1) | 15 (23.8) | 24 (44.4) | 20 (33.3) |
| SD | 37 (52.9) | 48 (76.2) | 30 (55.6) | 40 (66.7) |
| Involved metastatic sites | ||||
| Liver | 53 (75.7) | 49 (77.8) | 46 (85.2) | 55 (91.7) |
| Lung | 4 (5.7) | 5 (7.9) | 23 (42.6) | 29 (48.3) |
| Lymph node | 7 (10.0) | 1 (1.6) | 38 (70.4) | 34 (56.7) |
| Peritoneum | 5 (7.1) | 4 (6.3) | 8 (14.8) | 18 (30.0) |
| Other | 1 (1.4) | 4 (6.3) | 9 (16.7) | 21 (35.0) |
| Involved metastatic sites, No. (%) | ||||
| 1 | 70 (100) | 63 (100) | – | – |
| 2 | – | – | 42 (77.8) | 32 (53.3) |
| ≥3 | – | – | 12 (22.2) | 28 (46.7) |
| Metastatic sites incl. liver | ||||
| Liver-limited | 53 (75.7) | 49 (77.8) | – | – |
| Liver + 1 organ | – | – | 34 (73.9) | 27 (49.1) |
| Liver + ≥2 organs | – | – | 12 (26.1) | 28 (50.9) |
- Abbreviations: CI, confidence interval; ECOG PS, Eastern Cooperative Oncology Group performance status; FOLFOX, fluorouracil, folinic acid, and oxaliplatin; FU/FA, fluorouracil and folinic acid; left-sided primary tumor, splenic flexure to rectum; right-sided primary tumor, caecum to transverse colon; ORR, objective response rate; OS, overall survival; PFS, progression-free survival; Pmab, panitumumab.
Objective response rate to induction therapy as assessed by the local investigator was numerically higher in patients with multiple compared to single organ disease, independent of treatment arms, whereas higher response rates to maintenance therapy were observed for FU/FA+ Pmab compared to FU/FA alone—irrespective of single compared to multiple organ disease (Table 1).
3.2.1 Metastatic organ involvement at baseline
In both treatment arms, more than half of the patients were diagnosed with single organ metastasis at baseline (FU/FA+ Pmab = 56.5%, FU/FA alone = 51.2%) of which liver-limited disease accounted for the largest proportion (FU/FA+ Pmab = 75.7%, FU/FA alone = 77.8%). In patients with more than one organ affected, metastatic spread including two sites at baseline was more frequent in the FU/FA+ Pmab arm as compared to the FU/FA arm (77.8% vs 53.3%), while vice versa in patients with ≥three metastatic sites (22.2% vs 46.7%) (Table 1).
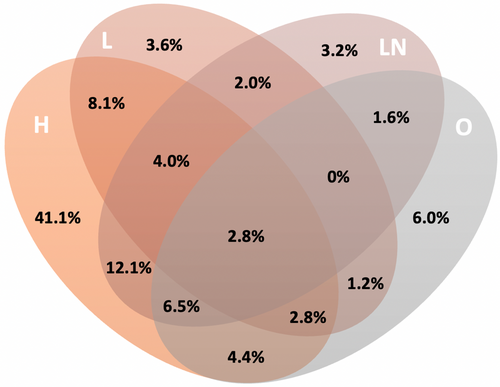
When analyzing metastatic pattern within the PanaMa study population, liver-limited disease was observed most frequently (41.1%) followed by two-organ metastasis including the liver with hepatic plus lymphonodal involvement (12.1%) and hepatic plus pulmonary involvement (8.1%) representing second and third most common distribution patterns. Singular organ involvement other than liver-limited disease was comparatively rare (lung-limited disease: 3.6%, lymph node-limited disease: 3.2%) (Figure 2).
3.3 Prognostic analyses of metastatic organ involvement
Multiple organ metastasis was a negative prognostic factor for PFS of maintenance therapy (5.8 vs 8.1 months; HR 1.48, 95% CI 1.13-1.93; P = .004; Figure 3A) regardless of treatment arm with a trend also for OS of maintenance therapy (26.1 vs 27.4 months; HR 1.37, 95% CI 0.98-1.93; P = .068; Figure 3B).
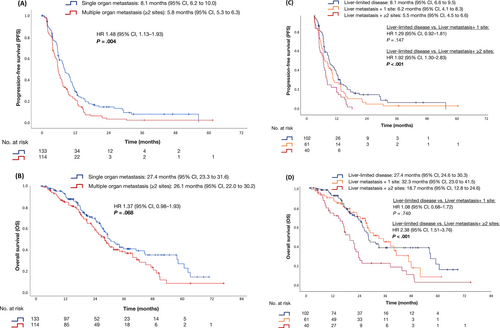
When patients with liver metastasis plus one additional extrahepatic site were compared to patients with liver-limited disease, only little impact on prognosis was evident for PFS and OS (Figure 3C,D).
By contrast, in patients with hepatic plus multiple extrahepatic manifestations (liver metastasis plus ≥2 additional sites) compared to patients with metastases only affecting the liver, PFS (5.5 vs 8.1 months; HR 1.92, 95% CI 1.30-2.83; P < .001; Figure 3C) as well as OS of maintenance therapy (18.7 vs 27.4 months; HR 2.38, 95% CI 1.51-3.76; P < .001; Figure 3D) were less favorable.
3.4 Predictive analyses of metastatic organ involvement
In patients with multiple organ metastatic disease (≥2 metastatic sites), maintenance therapy including Pmab was associated with a pronounced PFS-benefit (7.5 vs 5.0 months; HR 0.58, 95% CI 0.39-0.86; P = .006), and numerically less evident in patients with single organ metastatic disease (9.9 vs 7.2 months; HR 0.83; 95% CI, 0.57-1.21; P = .332; Interaction P = .183; Figure 4A). Regarding OS of maintenance therapy, a numerical, but not significant advantage of the Pmab-containing regimen was evident in both multiple (27.9 vs 22.8 months; HR 0.89, 95% CI 0.55-1.41; P = .609) as well as single organ metastatic disease (30.8 vs 27.3 months, HR 0.84, 95% CI 0.51-1.38; P = .484; Figure 4B).
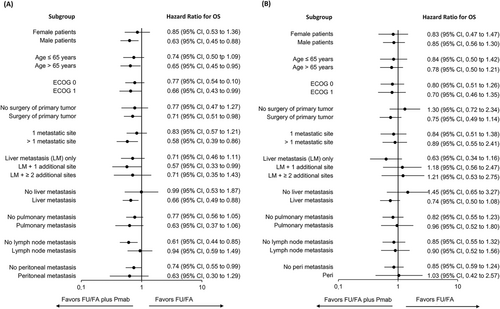
Likewise, in patients with liver metastasis plus one additional extrahepatic site, the Pmab-containing regimen was associated with a significantly prolonged PFS (9.2 vs 5.4 months; HR 0.57, 95% CI 0.33-0.99; P = .046), which was less pronounced in patients with liver-limited disease (10.2 vs 7.2 months; HR 0.71; 95% CI, 0.46-1.11; P = .134; Interaction P = .521; Figure 4A). Regarding OS of maintenance therapy, no benefit of the Pmab-containing maintenance regimen was evident in patients with liver metastases plus one additional organ (34.5 vs 32.3 months, HR 1.18; 95% CI, 0.56-2.47) rather favoring FU/FA while there was a trend toward superior Pmab-efficacy observed in patients with liver-limited disease (32.3 vs 26.5 months; HR 0.63; 95% CI, 0.34-1.16; P = .137; Figure 4B).
Analysis of liver-limited disease compared to liver metastasis plus ≥2 additional metastatic sites fell short of predicting PFS or OS of maintenance therapy (Figure 4). In terms of OS, a nonsignificant trend toward superior efficacy of FU/FA alone compared to FUFA+Pmab was apparent.
Within subgroup analysis of predictive factors for maintenance therapy with FU/FA plus Pmab (Figure 4A), presence of liver metastases as well as absence of lymph node and peritoneal metastases were positively associated with prolonged PFS while none of the subgroups analyzed proofed predictive for OS of maintenance therapy (Figure 4B).
4 DISCUSSION
Patients with RAS wildtype mCRC present with heterogenous metastatic pattern despite their molecular preselection suggesting that biological and clinical differences within this favorable prognostic group exist. There are limited data on the prognostic implications of specific metastatic patterns28 as a detailed metastatic pattern characterization is not routinely reported in clinical trials. However, some studies have been able to prove an association of the extent and anatomical localization of metastatic spread with the prognosis of mCRC patients2, 12, 13, 29, 30 potentially also posing a feasible baseline marker to guide treatment decisions. In this analysis, we aimed to evaluate metastatic organ involvement for its prognostic and predictive value for PFS and OS of maintenance therapy with FU/FA alone or in combination with Pmab, particularly focusing on liver metastases.
We found that single compared to multiple organ metastases were associated with favorable PFS and OS of maintenance therapy. This survival benefit was even more pronounced in patients with liver-limited disease compared to patients with additional extended extrahepatic metastasis. Correspondingly, metastatic spread to more than one organ has been identified as a poor prognostic factor in mCRC patients8-10 leading to the introduction of the subcategories M1A (single metastatic site) and M1b (peritoneal or multiple metastatic sites) within the American Joint Committee on Cancer (AJCC) staging system in 2010.31 Our results support the perspective that the observed survival benefit is not solely attributable to an increased probability of achieving resectability but may also reflect a more favorable tumor biology of single organ metastatic disease at baseline especially if limited to the liver. Interestingly, the impact on long-term survival (OS) was hardly affected by the addition of one metastatic site to liver-metastases. Of cause, the context of this observation in a maintenance therapy setting does not allow for direct extrapolation to patients undergoing conversion therapy. However, if this finding can be confirmed in the latter setting, it might support the clinical decision for resection of liver-metastases in cases, where limited extrahepatic disease is evident or suspected (ie, in the lung or in lymph nodes).
There have been reservations regarding the accuracy of image morphologic lymph node staging based solely on lymph node size. Data show that up to 70% of lymph nodes with metastases in colorectal cancer have a diameter of ≤5 mm,32, 33 posing the risk of understaging patients. Therefore, exploratory analyses were performed (Figure S1), excluding lymphonodal status. Here, the previously described trend regarding a prognostic advantage of limited organ involvement was confirmed. However, all group differences reached significant results, suggesting that image morphologic metastasis assessment without consideration of lymph node status may be more adequate and may be considered for future analyses.
The therapeutic benefit of adding Pmab to FU/FA maintenance therapy was pronounced in patients with multiple organ metastases whereas as a numerically smaller benefit of the Pmab-containing maintenance regimen was observed in patients with single organ metastasis. Similarly, patients with two-organ metastasis including the liver derived a pronounced PFS benefit from FU/FA+ Pmab maintenance therapy compared to patients with liver-limited disease. Therefore, it might be concluded that patients who initially present with extensive metastasis may require more intensive therapy regimens while patients with single organ metastasis do not seem to benefit to the same extent from anti-EGFR antibody-based maintenance therapy. It might be assumed that this observation is somehow biased by on-treatment decisions in the study since the relatively short induction therapy of the PanaMa-trial26 compared to other maintenance trials34-36 may have left some patients with high tumor (organ-) load in a clinical need of rather intense continuation of more active antitumor therapy potentially favoring the use of FU/FA plus Pmab as maintenance therapy vs the control arm.
Corresponding with the overall results of the trial,26 subgroup analyses of metastatic pattern fell short of predicting OS of either maintenance therapy arm. This might be even aggravated by low event numbers which are further reduced by this subgroup evaluation.
There are limitations to the present analysis that need to be taken into account. First, as this analysis was conducted as a retrospective evaluation of the PanaMa trial, the results should be interpreted as hypothesis generating. Second, owing to limited sample sizes, we refrained from performing subgroup analyses on additional metastatic localization patterns, for example, lung-limited or peritoneal-limited metastasis that might be of clinical interest.37 Accordingly, prognostic and predictive impact of other metastatic patterns could not be detected. Third, distribution of metastatic organ involvement of the cohort analyzed might be biased due to the underlying study design. As only patients with at least stable disease during induction were eligible for randomization to maintenance therapy, the cohort considered for the present analysis is preselected excluding patients with primary unsatisfactory response (and potentially also greater metastatic load due to aggressive biology) and also—even more importantly—patients who were potential candidates for secondary resection of metastases. Finally, it should be noted that this evaluation was based on regular computed tomography scans and some organ-involvement (ie, liver metastases) may not have been ideally captured by the scheduled scans. Also, the clear definition of present lung and peritoneal metastases underlies insecurities in the daily clinical practice and may have biased our stratification for clinical subgroups to some extent in a way that the group of liver-limited disease (or one organ disease) patients contains patients that actually had an involvement beyond the liver. Likewise, a more detailed classification of metastasis was not possible within the scope of this evaluation, which would have been valuable for example, in terms of lymph node metastasis (locoregional vs distant) with regard to prognostic characterization.
In conclusion, metastatic organ involvement has prognostic and also predictive impact on PFS in patients with RAS WT mCRC. The efficacy of intensified maintenance therapy including an anti-EGFR antibody is pronounced in patients with multiple organ metastasis with less striking effects observed in patients with single organ metastatic disease. These data may support clinical decisions when EGFR-based maintenance therapy is considered. Prospective trials are warranted to evaluate the role of various metastatic patterns for maintenance treatment with anti-EGFR antibodies.
AUTHOR CONTRIBUTIONS
Greta Sommerhäuser: Data curation, Formal analysis, Writing—Original Draft, Visualization. Meinolf Karthaus: Investigation, Conceptualization, Writing—Review & Editing. Annika Kurreck: Writing—Original Draft, Visualization. Alexej Ballhausen: Writing—Review & Editing. Johanna W. Meyer-Knees: Writing—Review & Editing. Stefan Fruehauf: Conceptualization, Investigation, Writing—Review & Editing. Ullrich Graeven: Conceptualization, Investigation, Writing—Review & Editing. Lothar Mueller: Investigation, Writing—Review & Editing. Alexander O. Koenig: Investigation, Writing—Review & Editing. Ludwig Fischer v. Weikersthal: Investigation, Writing—Review & Editing. Eray Goekkurt: Investigation, Writing—Review & Editing. Siegfried Haas: Investigation, Writing—Review & Editing. Arndt Stahler: Investigation, Writing—Review & Editing. Volker Heinemann: Conceptualization, Investigation, Writing—Review & Editing. Swantje Held: Data Curation, Validation, Writing—Review & Editing. Annabel H. S. Alig: Writing—Review & Editing. Stefan Kasper-Virchow: Conceptualization, Investigation, Writing—Review & Editing. Sebastian Stintzing: Conceptualization, Investigation, Writing—Review & Editing. Tanja Trarbach: Conceptualization, Writing—Review & Editing. Dominik P. Modest: Conceptualization, Methodology, Validation, Formal analysis, Investigation Writing—Original Draft, Visualization, Supervision, Project administration. The work reported in the paper has been performed by the authors, unless clearly specified in the text.
ACKNOWLEDGMENT
Open Access funding enabled and organized by Projekt DEAL. [Correction added on 17 October 2023, after first online publication: The Projekt DEAL funding information has been added in this version.]
FUNDING INFORMATION
The legal funder (sponsor) of the trial was the AIO Studien gGmbH, Berlin, Germany. Amgen Inc (Thousand Oaks, CA, USA) supported the trial with study medication and a research grant to the AIO Studien gGmbH.
CONFLICT OF INTEREST STATEMENT
Annika Kurreck: Honoria: Amgen, Servier; Travel, Accommodations, Expenses: Amgen. Ullrich Graeven: Honoria: Boehringer Ingelheim, Amgen, AstraZeneca, Bristol-Myers Squibb, MSD Oncology, Sanofi Aventis GmbH, Fujifilm, Novartis, Cellrion, Amgen, MSD Oncology. Research funding: Ipsen, Macrogenics. Travel, Accomodations, Expenses: Boehringer Ingelheim, GSK. Arndt Stahler: Honoraria: Roche, Taiho Pharmaceutical, Servier. Expenses for travel and accomodations: Roche, Merck KGaA, Amgen, Pfizer, Lilly Oncology. Volker Heinemann: Honoraria for talks and advisory board role: Merck, Amgen, Roche, Sanofi, SIRTEX, Servier, Pfizer, AstraZeneca, BMS; MSD, Novartis, Boehringer Ingelheim, Pierre-Fabre, Celgene, Terumo, Oncosil, Seagen, NORDIC Pharma; Research funding: Merck, Amgen, Roche, Sanofi, Pfizer, Boehringer-Ingelheim, SIRTEX, Servier, GSK. Annabel H. S. Alig: Honoria: MSD; Travel: Merck Darmstadt. Stefan Kasper-Virchow: Merck Serono, MSD, Novartis, BMS, Amgen, Roche, Sanofi-Aventis, Servier, Incyte and Lilly. Sebastian Stintzing: Honoraria for talks and CME activities: AMGEN, AstraZeneca, Bayer, BMS, ESAI, LEO Pharma, Lilly, Merck KGaA, MSD, Pierre-Fabre, Roche, Sanofi, Servier, Taiho, Takeda; Research funding (institutional): Merck KGaA, Pierre-Fabre, Servier, Roche. Tanja Trarbach: Research Funding: Amgen. Travel, Accommodations, Expenses: Ipsen, Takeda, OMT, AbbVie, Novartis, MSD, Sanofi/Aventis, Amgen, Johnson & Johnson/Janssen. Dominik P. Modest: Honoraria: Amgen, Merck, Servier, Pierre-Fabre, BMS, MSD, Incyte, Lilly, Sanofi, G1, Transgene, Seagen, Onkowissen; Research funding (inst): Servier, Amgen. Lothar Müller: Travel support: Octapharm. Meinolf Karthaus: Travel and Accommodation expenses: Amgen. Siegfried Haas: Travel and Accommodation expenses: Beigene, Pharmamar, Boehringer Ingelheim. Stefan Fruehauf: Advisory Board: Amgen. Alexej Ballhausen: Travel, Accommodation, Expenses: Amgen. Eray Goekkurt: Honoraria: MSD, BMS, AstraZeneca, Servier. The other authors have no conflict to disclose.
ETHICS STATEMENT
The trial recruited patients in accordance with the Declaration of Helsinki and was approved by the ethics committees of all participating centers. All patients provided written informed consent before trial entry. The trial is registered with ClinicalTrials.gov (NCT01991873).
Open Research
DATA AVAILABILITY STATEMENT
With respect to the clinical trial, Randomized phase II study for evaluation of efficacy and safety of maintenance treatment with 5-FU/FA plus panitumumab versus 5-FU/FA alone after prior induction treatment with mFOLFOX6 plus panitumumab and re-induction with mFOLFOX6 plus panitumumab in case of progression for first-line treatment of patients with metastatic colorectal cancer (PanaMa), sponsor code AIO-KRK-0212, EudraCT-No. 2012-005422-30, AIO Studien gGmbH acting as the legal sponsor is committed to provide information about its results to researchers with the goal of facilitating scientific progress. Information that will be considered for disclosure includes individual participant data that underlie the results reported in this article (text, tables, figures, and appendices). Additionally, study Protocol and statistical analysis plan can be made available. All data shared must be anonymized to protect the privacy of the patients who participated in the trial, in accordance with applicable laws and regulations and in compliance with the International Council for Harmonisation and Good Clinical Practice (ICH/GCP). Researchers should provide a scientifically sound proposal directed to [email protected] for approval to gain access to the requested data. Shared data are only to be used to achieve aims of the approved proposal. Further information is available from the corresponding author upon request.