Does vitamin D supplementation impact serotonin levels? A systematic review and meta-analysis
Abstract
Background and Aims
Vitamin D deficiency impacts a significant proportion of the world's population, and this deficiency has been linked to various conditions characterized by imbalanced serotonin regulation. The objective of this systematic review and meta-analysis was to evaluate the effect of vitamin D supplementation on serum serotonin levels.
Methods
We conducted a comprehensive search of PubMed, Scopus, Cochrane Central for Randomized Clinical Trials, and Web of Science up to September 2022, without any language restrictions. The effect sizes were calculated using the standard mean difference (SMD) and 95% confidence interval (CI).
Results
Six randomized clinical trials involving 356 participants were included in the analysis. Our findings indicated no significant changes in serotonin levels between the intervention and control groups (SMD: 0.24 ng/mL, 95% CI: −0.28, 0.75, p > 0.10). Subgroup analysis also did not reveal any significant changes in serotonin levels among children, participants with autism spectrum disorders, interventions lasting 10 weeks or longer, or those receiving vitamin D doses below 4000 IU/day.
Conclusion
Although the results obtained in this systematic review are inconclusive, they support the need for further well-designed randomized trials to assess the potential role of vitamin D supplementation in regulating serotonin levels and potentially ameliorating depression and related disorders.
1 INTRODUCTION
Serotonin (5-hydroxytryptamine; 5-HT) is a crucial neurotransmitter involved in various physiological functions within the body. Its synthesis relies on the conversion of l-tryptophan, an essential amino acid, through a cascade of enzymatic reactions.1, 2 Serotonin's effects are mediated by a diverse array of serotonin receptors distributed throughout human tissues.3, 4 Notably, serotonin influences both peripheral functions, such as platelet aggregation and immune responses, as well as central functions, including mood regulation and memory.5, 6
Vitamin D, a fat-soluble vitamin and prohormone, is recognized for its role in calcium and phosphorus metabolism and bone health.7, 8 The primary source of vitamin D is cutaneous synthesis induced by ultraviolet B rays, although it can also be obtained from dietary sources and supplements.9, 10 Beyond its traditional role in bone health, vitamin D exerts significant effects on gene regulation, cardiovascular function, immune response, and hormonal interplay through the vitamin D receptor.11-13 Vitamin D deficiency affects a substantial portion of the global population and has been implicated in various disorders where serotonin dysregulation is involved.14, 15
Despite the potential interplay between vitamin D and serotonin, the precise relationship and its implications remain largely unknown. The existing knowledge gap calls for further research to elucidate the connection between vitamin D status and serotonin levels, as well as their indirect effects on the regulation of the serotonergic system and associated diseases. Notably, no comprehensive synthesis of evidence regarding the impact of vitamin D supplementation on serotonin levels has been published to date. Therefore, this systematic review and meta-analysis of randomized controlled trials (RCTs) aims to investigate the overall effect of vitamin D supplementation on serum serotonin concentrations, while exploring potential dose–response relationships.
By bridging this research gap, we can gain valuable insights into the interplay between vitamin D and serotonin and uncover potential therapeutic implications. Understanding the relationship between these two crucial factors may contribute to the development of novel interventions for disorders where serotonin dysregulation is implicated.
2 MATERIALS AND METHODS
This systematic review was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA).16, 17 The systematic review protocol has been registered in PROSPERO (CRD42021242804).
2.1 Search strategy and study selection
We performed a systematic review by searching databases PubMed, Scopus, Cochrane Central for Randomized Clinical Trials, and Web of Science up to September 2022. The following keywords were used for identifying studies on vitamin D and serum serotonin level: (“Vitamin D” OR “Cholecalciferol” OR “Hydroxycholecalciferols” OR “Ergocalciferols” OR “25-Hydroxyvitamin D 2” OR “Dihydrotachysterol” OR “Calcitriol” OR “1 alpha,25-Dihydroxyvitamin D3” OR “1,25 Dihydroxyvitamin D3” OR “Vitamin D3” OR “1 alpha,25-Dihydroxycholecalciferol” OR “1,25(OH)2-20epi-D3” OR “1,25(OH)-D3”) AND “Serotonin” OR “Hydroxytryptamine” OR “5-Hydroxytryptamine” OR “5-HT” OR “5-Methoxytryptamine” AND “randomized controlled trial” OR “controlled clinical trial” OR “randomized controlled trials” OR “random allocation” OR “double blind method” OR “single blind method” OR “clinical trial” OR “clinical trials” OR “placebos” OR “placebo” OR “random.” The complete search strategy and syntaxes for all databases are provided in Appendix A1. We had no language, time, and location restrictions for identified studies. The reference lists of relevant reviews were manually screened by two independent reviewers (J. H. and M. M.) to find other eligible articles.
2.2 Eligibility criteria
Eligibility criteria were based on the PICOS (Population, Intervention, Comparison, Outcome, and Study Design) approach: population (P): unhealthy adults; intervention (I): vitamin D (in any doses and durations); comparison (C): placebo or no intervention; outcome (O): serum serotonin level; and study design (S): RCTs. Published studies with the following criteria were excluded: (1) animal or in vitro studies, (2) duplicate or overlapping data, (3) cross sectional, case reports, and case series studies (4) less than 1 week follow up after intervention (5) incomplete data about the serotonin levels of intervention and placebo groups, and (6) articles without a full text available.
2.3 Data extraction
Date extraction was performed by two independent reviewers (J. H. and M. M.). The data extraction form was designed to collect: the first author's name, year of publication, country, study design, total sample size, intervention dose, characteristics of participants (age, gender, and BMI [body mass index]), duration, supplementation, study quality, and outcomes.
2.4 Risk of bias assessment
We evaluated the quality of each eligible study based on the Cochrane Risk-of-Bias tool for randomized trials (RoB 2) with the following seven domains: random sequence generation (selection bias), allocation concealment (selection bias), blinding of participants and assessors (performance bias), blinding of outcome assessment (detection bias), incomplete outcome data (attrition bias), selective reporting (reporting bias), and other bias. Each item was categorized as low, high, or an unclear risk of bias.
2.5 Statistical analysis
We extracted all data in mean ± standard deviation (SD) format and converted other formats to SD if necessary. For evaluating the effects of vitamin D supplementation on serum serotonin level, standard mean difference (SMD) and 95% confidence interval (CI) were applied. All statistical analyses were performed using STATA (version 15). Heterogeneity was assessed using the I2 statistic. If I2 was less than 50%, we used a fixed effects model, and a random effects model was applied for I2 more than 50%. p < 0.10 was considered as statistically significant for our systematic review. A random effects model was used for calculating pooled effect size for a variable of interest. We additionally applied the Beg funnel plot and Egger funnel plot tests to evaluate publication bias. Sensitivity analysis was performed to evaluate each individual study's effect on main pooled effect size. We also conducted subgroup analysis based on Disease type (Depressive disorders vs. Autism-related disorders vs. Other diseases), Duration of vitamin supplementation (≥10 weeks vs. <10 weeks), Vitamin D dose (≥4000 vs. <4000 IU/day), and Age (Children vs. Middle-aged adults).
3 RESULTS
3.1 Study selection
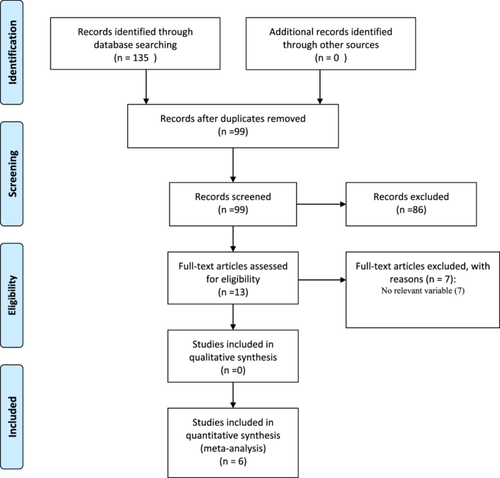
Figure 1 illustrates the screening and selection process of eligible studies. The initial multi-database search yielded 135 publications. After removing duplicates, 99 records were screened and 86 were excluded. The remaining 13 articles were assessed for eligibility and six articles18-23 that met the inclusion criteria were included in the meta-analysis.
3.2 Study characteristics
A total of six clinical trials involving 356 participants were selected in this systematic review and meta-analysis. Table 1 provides the main characteristics of the included studies. These trials were published between 2019 and 2020. All trials apart from one18 followed a double-blind design. The duration of interventions ranged from 8 to 19 weeks and sample size from 30 to 78 participants. Age and mean BMI ranged between 3 and 81 years, and from 24 to 30 kg/m2, respectively. The studies were performed in Iran,20-23 Norway,19 and Saudi Arabia.18 Five studies included both males and females18, 20-23 and one study was conducted only with adult males.19 The trials enrolled patients with generalized anxiety disorder,18 autism spectrum disorders (ASD),20 depression,21 irritable bowel syndrome,23 attention-deficit/hyperactivity disorder,22 and stress.19 The trials were conducted among children20, 22 and adults.18, 19, 21, 23 The dose of vitamin D administered was between 1600 IU once a day and 50,000 IU once a week. Most studies indicated that serum serotonin concentrations did not significantly change (p > 0.0.5) in the vitamin D group compared to the placebo group19-23 but in one trial, serum serotonin levels were significantly increased in vitamin D-treated patients compared to the untreated.18
| Study, (reference) | Country | Condition | Duration (week) | Vitamin D Dose | N (analyzed) | Female gender n (%) | Participants | Age (years) | Age, years Mean ± SD | BMI, kg/m2 Mean ± SD | Serum Serotonin (ng/mL)* | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Intervention | Control | Intervention | Control | ||||||||||
| Eid et al.18 | Saudi Arabia | GAD | 12 | 50,000 IU (once/week) | 30 (♀♂) | 13 (43) | Adult | 18−65 | 40 ± 2 | 28.8 | (↑) | ||
| Seyedi et al.22 | Iran | ADHD | 12 | 2000 IU/day | 75 (♀♂) | 23 (26) | Children | 6−12 | 10. ± 3 | 10 ± 4 | (↔) | ||
| Javadfar et al.20 | Iran | ASD | 15 | 300 IU/kg daily to 6000 IU/d | 43 (♀♂) | 7 (16) | Children | 3−13 | 8 ± 2 | 8 ± 3 | (↔) | ||
| Kaviani et al.21 | Iran | Depression | 8 | 50,000 IU/2weeks | 56 (♀♂) | 50 (89) | Adult | 18−60 | 43 ± 9 | 42 ± 8 | 30.0 ± 4.7 | 28.6 ± 5.3 | (↔) |
| Sikaroudi et al.23 | Iran | IBS | 9 | 50,000 IU/week | 74 (♀♂) | 39 (44) | Adult | 18−65 | 34 ± 12 | 36 ± 8 | 24.5 ± 4.6 | 26.2 ± 4.4 | (↔) |
| Hansen et al.19 | Norway | Having Stress Individuals | 19 | 1600 IU/day | 78 (♂) | 0 | Adult | 31−81 | 48 ± 11 | 49 ± 11 | 30.0 ± 7.0 | 30.0 ± 7.0 | (↔) |
- Abbreviations: ADHD; Attention-Deficit/Hyperactivity Disorder; ASD, Autism spectrum disorders; IBS, Irritable bowel syndrome (IBS); GAD, Generalized Anxiety Disorder; ♂, Male; ♀, Female;
- * Statistical significance (p < 0.05) as either increased (↑), decreased (↓), or no difference (↔) between vitamin D versus placebo group in terms of serum serotonin.
3.3 Effect of vitamin D on serotonin
Figure 2 shows a forest plot for vitamin D supplementation effect on serum serotonin levels. Changes in serum serotonin concentrations following vitamin D supplementation were compared between vitamin D or untreated (placebo) groups in six clinical trials. The studies comprised a total of 356 participants in both arms (180 for vitamin D treated and 176 for untreated or placebo).
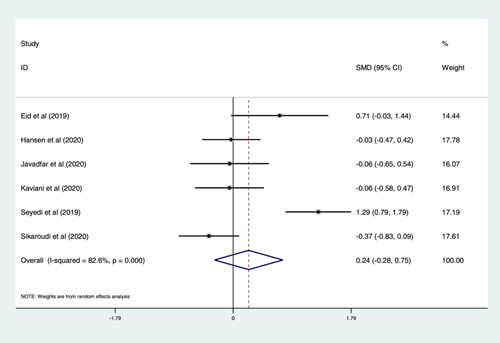
A meta-analysis of data indicated no significant changes in serum serotonin concentrations following supplementation with vitamin D compared to the untreated group (SMD: 0.24 ng/mL, 95% CI: −0.28, 0.75, p > 0.05) using a random-effects model, with a large heterogeneity (I2 = 82.6%, p < 0.001). The results of categorical subgroup analyses for the effect of vitamin D on serum serotonin levels are summarized in Table 2. The subgroup analyses suggest no significant differences in mean serum serotonin concentrations between subgroups in terms of disease type, duration of intervention, the dose of vitamin D, and age of participants. However, serum serotonin levels were higher but nonsignificant in participants with ASDs, children, interventions with duration ≥ 10 weeks and dose of vitamin D less than 4000 IU/day compared to other patients, adults, trials with duration < 10 weeks, and dose of vitamin D ≥ 4000 IU/day, respectively (Table 2).
| Sub-grouped by | No. of arms | Effect size (SMD) | 95% CI | I2 (%) | p for heterogeneity | |
|---|---|---|---|---|---|---|
| Disease type | Depressive disorders | 3 | 0.12 | −0.28, 0.53 | 38.3 | 0.198 |
| Autism related disorders | 2 | 0.63 | −0.69, 1.95 | 91.3 | 0.001 | |
| IBS | 1 | −0.37 | −0.83, 0.09 | - | - | |
| Duration | ≥10 weeks | 4 | 0.47 | −0.22, 1.17 | 83.9 | 0.000 |
| <10 weeks | 2 | −0.23 | −0.58, 0.11 | 00.0 | 0.381 | |
| Vitamin D dose | ≥4000 IU/day | 3 | 0.04 | −0.55, 0.62 | 65.8 | 0.054 |
| <4000 IU/day | 3 | 0.40 | −0.46, 1.26 | 89.3 | 0.000 | |
| Age | Children | 2 | 0.63 | −0.69, 1.95 | 91.3 | 0.001 |
| Adults | 4 | −0.01 | −0.38, 0.36 | 48.8 | 0.119 | |
- Note: Bold values indicate statistically significant p-values.
- Abbreviations: CI, confidence interval; SMD, standard mean difference.
3.4 Risk of bias assessment
A summary assessment of the risk of bias for the included studies is provided in Appendix A2. The results indicated that a low risk for random sequence generation bias was found in three trials,20, 22, 23 followed by those with unclear risk.18, 19, 21 In addition, risk of allocation concealment bias was low in four trials19, 20, 22, 23 and unclear in two18, 21 trials. As one trial was not double-blinded, it was deemed to be at high risk of performance bias,18 whilst low and unclear risk were considered for two19, 20 and three21-23 trials, respectively. Three trials18, 21, 22 were judged to be at high risk of detection bias, while one trial had unclear risk23 and the other two trials19, 20 were considered to have a low risk. Furthermore, attrition bias was low in two21, 23 and unclear in four18-20, 22 trials. Three trials19, 21, 22 exhibited a low risk of reporting bias, while the remaining trials18, 20, 23 had an unclear risk. The majority of trials were judged to be at low risk for other bias18-22 except one,23 which had an unclear risk of bias.
4 DISCUSSION
This is the first known systematic review and meta-analysis to consider the effect of vitamin D supplementation compared to placebo or no intervention on serum serotonin levels. We included six studies reporting on 356 participants across three countries. Our generalized results were inconclusive, indicating no significant effect on serum serotonin concentrations following supplementation compared to the placebo or no treatment group in a largely heterogeneous population. However, subgroup analyses indicated a nonsignificant increase in serotonin levels in those with ASD, children, interventions ≥ 10 weeks, and with a dose < 4000 IU/day. Nevertheless, we recognize that the findings of subgroups are merely observational, and further studies examining these specific subgroups are needed to better understand the exact effects of vitamin D supplementation on serotonin levels.
Although we identified no other systematic reviews reporting any results about the effect of vitamin D supplementation on serotonin levels, several systematic reviews evaluating the effect of supplementation on depression24 have presented similarly inconclusive results with largely heterogenous populations, with significant findings only for subgroup analyses. Shaffer et al. found no significant effect of vitamin D supplementation on depressive symptoms, but observed a significant effect of supplementation on participants with clinically significant depressive symptoms or depressive disorders.25 A nonsignificant effect was also observed in patients without significant depression. Similarly, Spedding et al. reported inconclusive results for depression, finding that studies of differing methodological quality either improved or worsened depression severity.26 Comparatively, Jamilian et al. found a significant reduction in the Beck Depression Inventory score, which evaluates the depression levels, following supplementation in patients with psychiatric disorders.27 Although we recognize that the effect on serotonin levels and depression is not comparable, serotonin levels and available receptors have been proposed to play a role in depression. These largely inconclusive findings highlight the need for randomized trials of high methodological quality evaluating the effect of various methods and durations of treatment of vitamin D supplementation, particularly considering the overall effect on serotonin levels.28
There are several suggested mechanisms about the effect of vitamin D supplementation on serotonin levels. Wang et al. in their large-scale in silico and microarray-based study reported over 900 genes containing putative DR3 vitamin D response elements (VDREs) upstream of the promoter regions, including tryptophan hydroxylase 1 (TPH1) and tryptophan hydroxylase 2 (TPH2).29 It has been speculated that VDREs present in the regulatory regions of TPH2 and TPH1 respond to the hormonal action of vitamin D in an inverse mode with increasing doses of vitamin D causing a repression of TPH1 expression30 and, on the contrary, TPH2 transcription is seemingly activated in the brain. Taken together, these observations provide additional clarification for the potential disproportion between blood and brain serotonin concentrations.31
Further, according to preclinical data, 1,25-dihidroxyvitamin D significantly increases the expression of both TPH 1 and 2 and represses the expression of serotonin reuptake transporter (SERT) and monoamine oxidase-A (MAO-A), two major players in serotonin metabolic pathway, which may explain the observed increase in serotonin concentration in treated cells.32 Thus, if vitamin D is proposed to suppress serotonin reuptake and metabolization it may induce higher levels of serotonin which, in turn, is thought to improve neuropsychiatric disorders, such as depression, particularly when a dysregulation of serotonergic neurotransmission is observed. However, to date, only limited animal models have sought to clarify this phenomena, and various vitamin D doses have yielded nonsignificant results. As such, this mechanism must be viewed with caution, and further studies aiming to clarify the exact effects of vitamin D on the brain are warranted.32 We acknowledge the significance of the dosages employed in our included trials, which effectively raised levels to above 50 nmol/l, potentially correcting deficiencies. We agree that considering higher vitamin D levels (>75 nmol/L) for serotonin effects warrants attention, especially given the relatively high doses administered. While some countries prescribe doses up to 50,000 IU per week for up to 8 weeks for deficient individuals, exceeding the tolerable upper intake of 4000 IU per day may raise concerns at the population level.33
The primary strength of the present meta-analysis lies in its inclusion of studies from various countries and diverse ethnic communities. However, the adult population represented is very homogeneous but there are two trial that were carried out with children and so the generalization of the results is limited. Additionally, various subgroups and inconsistencies in the means and regimens of vitamin D supplementation led to potentially unreliable results. However, this systematic review has some limitations that need to be addressed. First, the limited number of included studies affects our results. Second, the high heterogeneity and low quality of included studies also need to be acknowledged. Third, the lack of data on baseline serum vitamin D status and post-supplementation levels is a notable limitation that deserves attention. Finally, it was not defined in the included studies during which month of the year the study was conducted, which impacts exposure to the sun and the generation of vitamin D.
5 CONCLUSION
This study found inconclusive results regarding the effect of vitamin D supplementation, compared to placebo or no treatment, on serotonin levels. Given the variation in vitamin D supplementation dose and duration, this could result in significant differences between the results of included studies. Animal models propose a potential mechanism for vitamin D supplementation on improving serotonergic neurotransmission, indicating the importance of this research field. Ultimately, further randomized trials performed in the general population alongside subgroups with various neuropsychiatric diseases that evaluate various amounts, duration, frequency, and method of vitamin D supplementation on serum serotonin levels are warranted.
AUTHOR CONTRIBUTIONS
Javad Heshmati and Malek Alimohammadi-Kamalabadi: Conceptualization, methodology, manuscript preparation. Malek Alimohammadi-Kamalabadi, Milad Mehrbod and Shooka Mohammadi: Data extraction, writing—original draft preparation. Somayeh Ziaei: Data analysis, investigation. Maria Dulce da Mota Antunes de Oliveira Estêvão and Mahsa Malekahmadi: Writing—original draft preparation, writing—reviewing and editing. Emma Persad and Andrej Belančić: Writing—reviewing and editing, final draft validation. Mehrnaz Morvaridi, Elnaz Daneshzad and Motahareh Hasani: Writing—original draft.
CONFLICT OF INTEREST STATEMENT
The authors declare no conflict of interest.
ETHICS STATEMENT
All authors have read and approved the final version of the manuscript and Javad Heshmati had full access to all of the data in this study and takes complete responsibility for the integrity of the data and the accuracy of the data analysis.
TRANSPARENCY STATEMENT
The lead author Javad Heshmati affirms that this manuscript is an honest, accurate, and transparent account of the study being reported; that no important aspects of the study have been omitted; and that any discrepancies from the study as planned (and, if relevant, registered) have been explained.
APPENDIX A1: SEARCH STRATEGY
A systematic review and meta-analysis of the Vitamin D supplementation effects on serum serotonin.
Javad Heshmati (corresponding author)
Tehran, Iran.
E-mail: [email protected]
Telephone: +98 (021) 44442393
| Groups | Descriptors |
|---|---|
| Outcome | Serotonin OR Hydroxytryptamine OR 5-Hydroxytryptamine OR “5-HT” OR 5-Methoxytryptamine |
| Exposure | Vitamin D OR Cholecalciferol OR Hydroxycholecalciferols OR Ergocalciferols OR 25-Hydroxyvitamin D 2 OR Dihydrotachysterol OR Calcitriol OR 1 alpha,25-Dihydroxyvitamin D3 OR 1,25 Dihydroxyvitamin D3 OR Vitamin D3 OR 1 alpha,25-Dihydroxycholecalciferol OR 1,25(OH)2-20epi-D3 OR 1,25(OH)-D3 |
| Setting | Randomized controlled trial OR controlled clinical trial OR randomized controlled trials OR random allocation OR double blind method OR single blind method OR clinical trial OR clinical trials OR placebos OR placebo OR random |
PUBMED
Number of localized studies: 15
Limits: humans
Number of studies after applying limits: 11
| Descriptors | Number of studies reached | |
|---|---|---|
| #1 | "serotonin"[MeSH Terms] OR "serotonin"[All Fields] OR "serotonins"[All Fields] OR "serotonin s"[All Fields] OR "serotonine"[All Fields] OR "hydroxytryptamines"[All Fields] OR "serotonin"[MeSH Terms] OR "serotonin"[All Fields] OR "hydroxytryptamine"[All Fields] OR "serotonin"[MeSH Terms] OR "serotonin"[All Fields] OR "5 hydroxytryptamine"[All Fields] OR "serotonin"[MeSH Terms] OR "serotonin"[All Fields] OR "5 ht"[All Fields] OR "5 methoxytryptamine"[MeSH Terms] OR "5 methoxytryptamine"[All Fields] OR "5 methoxytryptamine"[All Fields] | 156,258 |
| #2 | ((((((((((("Vitamin D"[Mesh]) OR "Cholecalciferol"[Mesh]) OR "Hydroxycholecalciferols"[Mesh]) OR "Ergocalciferols"[Mesh]) OR "25-Hydroxyvitamin D 2"[Mesh]) OR "Dihydrotachysterol"[Mesh]) OR "Calcitriol"[Mesh]) OR 1 alpha,25-Dihydroxyvitamin D3) OR 1,25 Dihydroxyvitamin D3) OR Vitamin D3) OR 1 alpha,25-Dihydroxycholecalciferol) | 70,692 |
| #3 | ((((((((("Randomized Controlled Trial"[Publication Type] OR "Controlled Clinical Trial"[Publication Type]) OR "Randomized Controlled Trials as Topic"[Mesh]) OR "Random Allocation"[Mesh]) OR "Double-Blind Method"[Mesh]) OR "Single-Blind Method"[Mesh]) OR "Clinical Trial"[Publication Type]) OR ("clinical trial"[Publication Type] OR "clinical trials as topic"[MeSH Terms] OR "clinical trials"[All Fields])) OR "Placebos"[Mesh]) OR ("placebos"[MeSH Terms] OR "placebos"[All Fields] OR "placebo"[All Fields])) OR ("random allocation"[MeSH Terms] OR ("random"[All Fields] AND "allocation"[All Fields]) OR "random allocation"[All Fields] OR "random"[All Fields]) | 1,747,727 |
| #4 | #1 AND #2 AND #3 | 15 |
WEB OF SCIENCE
Number of localized studies: 30
Limits: documents types (articles)
Number of studies after applying limits: 14
| Descriptors | Number of studies reached | |
|---|---|---|
| #1 | TS=(Serotonin) OR TS=(Hydroxytryptamine) OR TS=(5-Hydroxytryptamine) OR TS=(5-HT) OR TS=(5-Methoxytryptamine) | 139,118 |
| #2 | TS=(“Vitamin D”) OR TS=(“Cholecalciferol”) OR TS=(“Hydroxycholecalciferols”) OR TS=(“Ergocalciferols”) OR TS=(“25-Hydroxyvitamin D 2”) OR TS=(“Dihydrotachysterol”) OR TS=(“Calcitriol”) OR TS=(“1 alpha,25-Dihydroxyvitamin D3”) OR TS=(“1,25 Dihydroxyvitamin D3”) OR TS=(“Vitamin D3”) OR TS=(“1 alpha,25-Dihydroxycholecalciferol”) OR TS=(“1,25(OH)2-20epi-D3”) OR TS=(“1,25(OH)-D3”) | 104,635 |
| #3 | TS=(Randomized controlled trial) OR TS=(controlled clinical trial) OR TS=(randomized controlled trials) OR TS=(random allocation) OR TS=(double blind method) OR TS=(single blind method) OR TS=(clinical trial) OR TS=(clinical trials) OR TS=(placebos) OR TS=(placebo) OR TS=(random) | 1,851,952 |
| #4 | #1 AND #2 AND #3 | 30 |
SCOPUS
Number of localized studies: 264
Limits: document type (article and article in press)
Number of studies after applying limits: 94
| Descriptors | Number of studies reached | |
|---|---|---|
| #1 | (TITLE-ABS-KEY (serotonin)) OR (TITLE-ABS-KEY (hydroxytryptamine)) OR (TITLE-ABS-KEY (5-hydroxytryptamine)) OR (TITLE-ABS-KEY (5-ht)) OR (TITLE-ABS-KEY (5-methoxytryptamine)) | 232,149 |
| #2 | ((TITLE-ABS-KEY (vitamin AND d) OR TITLE-ABS-KEY (cholecalciferol) OR TITLE-ABS-KEY (hydroxycholecalciferols) OR TITLE-ABS-KEY (ergocalciferols) OR TITLE-ABS-KEY (25-hydroxyvitamin AND d 2))) OR ((TITLE-ABS-KEY (dihydrotachysterol) OR TITLE-ABS-KEY (calcitriol) OR TITLE-ABS-KEY (1 alpha,25-dihydroxyvitamin AND d3) OR TITLE-ABS-KEY (1,25 AND dihydroxyvitamin AND d3) OR TITLE-ABS-KEY (vitamin AND d3))) OR (TITLE-ABS-KEY (1 alpha,25-dihydroxycholecalciferol)) | 158,434 |
| #3 | (TITLE-ABS-KEY (randomized AND controlled AND trial) OR TITLE-ABS-KEY (controlled AND clinical AND trial) OR TITLE-ABS-KEY (randomized AND controlled AND trials) OR TITLE-ABS-KEY (random AND allocation) OR TITLE-ABS-KEY (double AND blind AND method) OR TITLE-ABS-KEY (single AND blind AND method) OR TITLE-ABS-KEY (clinical AND trial) OR TITLE-ABS-KEY (clinical AND trials) OR TITLE-ABS-KEY (placebos) OR TITLE-ABS-KEY (placebo) OR TITLE-ABS-KEY (random)) | 3,260,115 |
| #4 | #1 AND #2 AND #3 | 264 |
COCHRANE
Number of localized studies: 16
Limits: -
Number of studies after applying limits: 16
| Descriptors | Number of studies reached | |
|---|---|---|
| #1 | Me ("Serotonin") or ("Hydroxytryptamine"):ti, ab, kw or ("5-Hydroxytryptamine"):ti, ab, kw or ("5-HT"):ti, ab, kw or ("5-Methoxytryptamine"):ti, ab, kw | 2791 |
| #2 | Me ("Vitamin D") or ("Cholecalciferol"):ti, ab, kw or ("Hydroxycholecalciferols"):ti, ab, kw or ("Ergocalciferols"):ti, ab, kw or ("25-Hydroxyvitamin D 2"):ti, ab, kw or ("Dihydrotachysterol"):ti, ab, kw or ("Calcitriol"):ti, ab, kw or ("1 alpha,25-Dihydroxyvitamin D3"):ti, ab, kw or ("1,25 Dihydroxyvitamin D3"):ti, ab, kw or ("Vitamin D3"):ti, ab, kw or ("1 alpha,25-Dihydroxycholecalciferol"):ti, ab, kw or ("1,25(OH)2-20epi-D3"):ti, ab, kw or ("1,25(OH)-D3"):ti, ab, kw | 7744 |
| #3 | #1 AND #2 | 16 |
Excluded articles
APPENDIX A2: Assessment of the risk of bias in the included studies
| Author, year | Random sequence generation (selection bias) | Allocation concealment (selection bias) | Blinding of participants and personnel (performance bias) | Blinding of outcome assessment (detection bias) | Incomplete outcome data (attrition bias) | Selective reporting (reporting bias) | Other bias |
|---|---|---|---|---|---|---|---|
| Eid et al.18 | ? | ? | + | + | ? | ? | _ |
| Hansen et al.19 | ? | _ | _ | _ | ? | _ | _ |
| Javadfar et al.20 | _ | _ | _ | _ | ? | ? | _ |
| Kaviani et al.21 | ? | ? | ? | + | _ | _ | _ |
| Seyedi et al.22 | _ | _ | ? | + | ? | _ | _ |
| Sikaroudi et al.23 | _ | _ | ? | ? | _ | ? | ? |
- Abbreviations: +, High risk; −, Low risk; ?, Unclear.
Open Research
DATA AVAILABILITY STATEMENT
Not Applicable.