Cytopenic overt primary myelofibrosis at presentation: Analysis of outcomes in the prospective, real-world ERNEST-2 registry
Unlike polycythemia vera (PV), essential thrombocythemia (ET), and prefibrotic primary myelofibrosis (pre-PMF), which show variable expansion of myeloid cell lineages at diagnosis, overt primary myelofibrosis (PMF) may present with uni- or multi-lineage cytosis and isolated or multiple cytopenias, the latter feature configuring a “cytopenic” phenotype (CyP).1 Among 1000 patients with PMF seen at Mayo Clinic, anemia and thrombocytopenia at diagnosis were found in 38%, including 24% requiring transfusions and 18%, respectively; those figures increased to 58%, 46%, and 28% for patients within 1 year from diagnosis.2 Furthermore, cytopenias are exacerbated by treatments, including JAK inhibitors.3, 4 Cytopenic MF is associated with a poorer prognosis compared to the “myeloproliferative” (MyP) counterpart and poses therapeutic challenges because of limited treatment options.5 In the aforementioned study, overall survival (OS) was 7.9 years for patients without anemia compared to 4.9, 3.4, and 2.1 years, respectively, for patients with mild, moderate, and severe anemia. These findings were confirmed in other retrospective studies.6 Also, isolated thrombocytopenia is prognostically adverse.7-9 Anemia, especially if transfusion dependent, and thrombocytopenia, are individually enlisted in conventional risk scores for survival prediction,10-12 including MIPSS70/v2.0 score,13-15 and are associated with increased risk of blast phase (BP).16
We report herein the analysis of data derived from the European multicenter collaborative ERNEST registry, specifically focusing on clinical characteristics and outcomes of MF patients with CyP. The ERNEST project prospectively enrolled patients with primary and secondary MF across tertiary European centers, with the aim of assuring reliability, representativeness, and comparability of real-world data. The project, promoted by the European LeukemiaNet (ELN) and coordinated by FROM (Research Foundation at Papa Giovanni XXIII Hospital in Bergamo), was supported by Novartis through a research collaboration. From February 2013 to May 2014, 1292 patients with WHO diagnosis of MF were included (ERNEST-1); for a subset of 584 overt PMF patients, enrolled in three countries (Italy, Spain, Sweden), extended follow-up data were available until the latest cutoff of December 2020 (ERNEST-2). The Institutional Review Board and Ethical Committee of each Centre approved the study, which was conducted in accordance with the Declaration of Helsinki. After the exclusion of 25 patients with incomplete data at diagnosis, a total of 559 patients constituted the current study population. A CyP was defined by the presence of at least one cytopenia at diagnosis: (i) sex-adjusted anemia (An), that is, hemoglobin (Hb) < 11 g/dL for male and <10 g/dL for female, further categorized as moderate (Hb 9–10.9 and 8–9.9 g/dL) and severe (Hb <9 and <8 g/dL, respectively); the impact of mild anemia (Hb 13–11 and 12–10 g/dL) was also preliminary analyzed; (ii) thrombocytopenia (Thr), as platelet count <100 × 109/L; (iii) leukopenia, as leukocytes <4 × 109/L. Patients showing neither of the above characteristics were considered as MyP.
Descriptive statistic was used to summarize patients' characteristics. Continuous variables were summarized by median and interquartile range and categorical ones as frequencies and percentages. Differences between patient categories were tested with the χ2 test (or Fisher's exact test, where appropriate) or the rank-sum test for categorical or continuous variables, respectively. Univariate and multivariate Fine and Gray's competing risk models were fitted to estimate the association between phenotype and BP, considering death as a competing event; estimated sub-distribution Hazard Ratios (sHRs) and corresponding 95% CIs were reported. The impact of CyP on mortality was estimated by the multivariable Cox regression model; OS was estimated by the Kaplan–Meier method.
The median follow-up was 5.4 years (interquartile range 2.5–9.1); patients' characteristics are presented in Table 1. Preliminary analysis on the impact of different severities of anemia revealed that mild anemia did not impact OS, therefore it was not included in the definition of CyP (Supporting Information S1: Figure 1). A CyP was identified in 275 patients (49.2%), of which 189 (68.7%) with isolated anemia, 32 (11.6%) with isolated thrombocytopenia, and 54 (19.6%) with An + Thr, accounting for 33.8%, 5.7%, and 9.7% of the entire cohort. Mild leukopenia was detected in only 10 cases (1.8%), all included in the An + Thr category, that were not considered separately. Compared to MyP, cytopenic patients were more likely to be older (p < 0.001), less frequently JAK2V617F-mutated (58.4% vs. 71.9%, p = 0.004), included in higher IPSS risk categories (int2/High: 70.8% vs. 29.3%, p < 0.001), and with fewer thrombotic events (12.4% vs. 18.3%; p = 0.051). Anemic patients tended to have higher IPSS risk scores (76.1% vs. 24.1%, p < 0.001) and less frequent splenomegaly (77.4% vs. 100%; p = 0.013) compared to isolated thrombocytopenia. While JAK2V617F-mutated patients were enriched in the anemia group, triple negative (TN) patients constituted 35.3% of the thrombocytopenia cohort compared to only 2.6% of the anemia cohort (p < 0.001); no meaningful differences as regarded systemic symptoms, history of thrombosis, and bleeding were highlighted (Table 1).
| Myeloproliferative phenotype (MyP) | Cytopenic phenotype (CyP) | Anemia (An) | Thrombocytopenia (Thr) | Anemia + Thrombocytopenia (An+Thr) | p | p | p | p | |
|---|---|---|---|---|---|---|---|---|---|
| N = 284 | N = 275 | N = 189 | N = 32 | N = 54 | MyP vs. CyP | An vs. Thr | An vs. An+Thr | Thr vs. An+Thr | |
| Age, mean (SD) | 60.6 (14.0) | 67.2 (11.4) | 67.4 (11.1) | 66.4 (12.3) | 66.8 (12.1) | <0.001 | 0.62 | 0.73 | 0.87 |
| Male, n (%) | 173/284 (60.9) | 189/275 (68.7) | 134/189 (70.9) | 17/32 (53.1) | 38/54 (70.4) | 0.053 | 0.046 | 0.94 | 0.11 |
| IPSS score, n (%) | |||||||||
| Low/Int-1 | 198/280 (70.7) | 79/271 (29.2) | 45/188 (23.9) | 22/29 (75.9) | 12/54 (22.2) | <0.001 | <0.001 | 0.79 | <0.001 |
| Int-2/High2 | 82/280 (29.3) | 192/271 (70.8) | 143/188 (76.1) | 7/29 (24.1) | 42/54 (77.8) | ||||
| Palpable spleen, n (%) | |||||||||
| No palpable spleen | 48/233 (20.6) | 43/223 (19.3) | 35/155 (22.6) | 0/23 (0.0) | 8/45 (17.8) | 0.88 | 0.013 | 0.83 | 0.083 |
| Palpable spleen, <10 cm | 153/233 (65.7) | 146/223 (65.5) | 98/155 (63.2) | 18/23 (78.3) | 30/45 (66.7) | ||||
| Palpable spleen, ≥10 cm | 32/233 (13.7) | 34/223 (15.2) | 22/155 (14.2) | 5/23 (21.7) | 7/45 (15.6) | ||||
| Symptomatic disease at diagnosis, n(%) | |||||||||
| No | 152/270 (56.3) | 128/255 (50.2) | 87/178 (48.9) | 16/26 (61.5) | 25/51 (49.0) | 0.16 | 0.23 | 0.99 | 0.30 |
| Yes | 118/270 (43.7) | 127/255 (49.8) | 91/178 (51.1) | 10/26 (38.5) | 26/51 (51.0) | ||||
| Hb (g/dL), median (IQR) | 12.5 (11.7–14.1) | 9.4 (8.2–10.3) | 9.2 (8.2–9.9) | 11.8 (11.2–12.9) | 8.8 (7.6–9.7) | <0.001 | <0.001 | 0.051 | <0.001 |
| WBC × 109/L, median (IQR) | 10.5 (7.4–16.3) | 7.3 (4.1–10.8) | 7.5 (4.8–10.0) | 9.3 (4.1–17.9) | 5.5 (3.2–9.4) | <0.001 | 0.29 | 0.023 | 0.028 |
| WBC > 20 × 109/L, n (%) | 50/284 (17.6) | 24/272 (8.8) | 17/186 (9.1) | 5/32 (5.6) | 2/54 (3.7) | 0.002 | 0.26 | 0.19 | 0.051 |
| PLT × 109/L, median (IQR) | 386.0 (251.0–657.0) | 168.0 (78.0–296.0) | 237.0 (165.0–419.0) | 58.0 (44.5–74.0) | 59.6 (39.0–75.0) | <0.001 | <0.001 | <0.001 | 0.90 |
| PLT > 450 × 109/L, n (%) | 126/283 (44.5%) | 44/271 (16.2) | 44/185 (23.8) | 0/32 (0) | 0/54 (0) | <0.001 | - | - | - |
| Driver mutations, n (%) | |||||||||
| JAK2 V617F | 161/224 (71.9) | 118/202 (58.4) | 93/143 (65.0) | 8/25 (32.0) | 17/34 (50.0) | 0.004 | 0.002 | 0.10 | 0.17 |
| Homozygous | 32/93 (34.4) | 14/57 (24.6) | 9/42 (21.4) | 2/4 (50.0) | 3/11 (27.3) | 0.27 | 0.24 | 0.70 | 0.56 |
| Heterozygous | 61/93 (65.6) | 43/57 (75.4) | 33/42 (78.6) | 2/4 (50.0) | 8/11 (72.7) | 0.27 | 0.24 | 0.70 | 0.56 |
| MPL W515 | 10/215 (4.7) | 11/186 (5.9) | 9/136 (6.6) | 1/21 (4.8) | 1/29 (3.4) | 0.66 | 1.00 | 1.00 | 1.00 |
| CALR | 27/197 (13.7) | 14/153 (9.2) | 12/115 (10.4) | 2/17 (11.8) | 0/21 (0.0) | 0.24 | 1.00 | 0.21 | 0.19 |
| Type 1 | 23/197 (11.7) | 9/153 (5.9) | 7/115 (6.1) | 2/17 (11.8) | 0/21 (0.0) | 0.091 | 0.33 | 0.60 | 0.19 |
| Type 2 | 4/196 (2.0) | 5/153 (3.3) | 5/115 (4.3) | 0/17 (0.0) | 0/21 (0.0) | 0.51 | 1.00 | 1.00 | - |
| Triple-negative | 8/197 (4.1) | 12/153 (7.8) | 3/115 (2.6) | 6/17 (35.3) | 3/21 (14.3) | 0.16 | <0.001 | 0.047 | 0.25 |
| Abnormal karyotype, n (%) | 42/153 (27.5) | 41/116 (35.3) | 22/79 (27.8) | 6/13 (46.2) | 13/24 (54.2) | 0.17 | 0.18 | 0.017 | 0.64 |
| Previous events, n (%) | |||||||||
| Previous thrombosis | 52/284 (18.3) | 34/275 (12.4) | 25/189 (13.2) | 2/32 (6.3) | 7/54 (13.0) | 0.051 | 0.27 | 0.96 | 0.33 |
| Previous bleeding | 7/284 (2.5) | 12/275 (4.4) | 6/189 (3.2) | 1/32 (3.1) | 5/54 (9.3) | 0.25 | 1.00 | 0.070 | 0.40 |
- Note: JAK2 V617F homozygous and heterozygous status was defined in the presence of a variant allele frequency (VAF) of >50% and ≤50%, respectively. Definition of abnormal karyotype was according to Tefferi et al.1 Bold values indicate statistically significant.
A total of 392 patients (70.1%) died, 59.9% of MyP and 80.7% of CyP (p < 0.001); the incidence rate (IR) of death was 17.3% pt-y (95% CI, 15.5–19.8) and 7.8% pt-y (95% CI, 6.6–9.1) among cytopenic and myeloproliferative patients, respectively (p < 0.001). Patients with An+Thr had the highest incidence rate (IR) of death (26.8% pt-y; 95% CI, 20.0–35.9) (p = 0.003) (Supporting Information S1: Table 1). OS was significantly shorter in CyP compared to MyP patients, with a median OS of 4.0 (interquartile range [IQR]: 1.7–7.5) versus 8.4 (IQR 4.6–16.1) years (p < 0.001) (Figure 1A–C), and 10-year cumulative incidence (CuI) of death of 80.9% (95% CI, 75.7%–85.7%) and 56.6% (95% CI, 50.6%–62.8%), respectively (Supporting Information S1: Table 2). Patients with An+Thr had the worst median survival (1.7 years; IQR 0.7–4.4), with a 10-year survival of 14.1% (95% CI, 6.2–25.1). Analysis of variables predicting death by the multivariable Cox model (Supporting Information S1: Figure 2A) identified age (with an increased risk of death of 6% for each additional year; HR 1.06, 95% CI, 1.05–1.07; p < 0.001), male gender (HR: 1.63, 95% CI, 1.25–2.13; p < 0.001), peripheral blasts ≥1% (HR 2.18, 95% CI, 1.56–3.05; p < 0.001), systemic symptoms (HR: 1.34, 95% CI, 1.05–1.72; p = 0.019), and leukocytes ≥20 × 109/L (HR 1.87, 95% CI, 1.28–2.72; p < .001) as being significantly associated with shortened survival. Splenomegaly was not a significant factor for death. By using the cohort of MyP patients as the reference category, moderate anemia (HR 1.59, 95% CI, 1.19–2.15; p = 0.002), severe anemia (HR 1.82, 95% CI, 1.21–2.72; p = 0.004), thrombocytopenia (HR 2.44, 95% CI, 1.36–4.36; p = 0.003), and An+Thr (HR 3.62, 95% CI, 2.20–4.82; p < 0.001) were all significantly associated with risk of death.
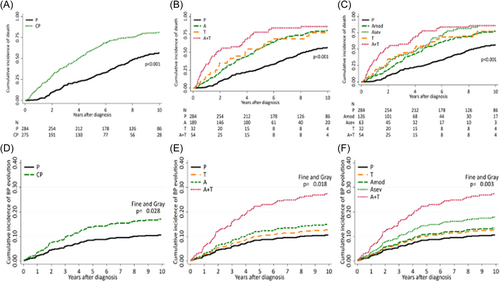
Seventy-five events of BP transformation occurred (13.4%); 45 (16.4%); and 30 (10.6%) among CyP and MyP patients (p < 0.0001), respectively, corresponding to an IR of 3.6% pt-y (95% CI, 2.7%–4.8%) and 1.4% pt-y (95% CI, 1.0%–2.0%) (p < 0.001), (Supporting Information S1: Table 1). Considering death as a competing event, the 10-yr CuI of BP was significantly greater for cytopenic (17.0%; 95% CI, 12.6–21.7) than MyP (10.6%; 95% CI 7.7–15.3) patients (p = 0.028) (Supporting Information S1: Table 2), particularly for An+Thr patients (27.4%; 95% CI, 16.9-41.2; p = 0.018) compared to other cytopenia categories (Figure 1D–F). Predictors of BP in a multivariable Fine & Gray competing risk model, with death as a competing event, were peripheral blasts ≥1% (HR: 3.17; 95% CI, 1.56–6.46; p = 0.001) and An+Thr phenotype (HR: 3.13; 95% CI, 1.41–6.97; p = 0.005) (Supporting Information S1: Figure 2B).
The current collection of real-world patients with PMF from the ERNEST-2 registry represents the largest prospective analysis focused on cytopenic patients, and owing to the maturity of follow-up (70% of patients died within the observation period and 13% developed BP), it allowed a thorough evaluation of clinical correlates and outcomes. In line with other retrospective series, 34% and 6% of PMF patients at diagnosis had anemia and thrombocytopenia, and 10% had both; this latter category was burdened by the worst outcome, with 10 y survival of only 14% and an OS of less than 2 years; furthermore, the cumulative incidence of BP was as high as 27.4%. These data point to anemic and thrombocytopenic patients as being at exceedingly high risk of dying and evolving to BP, which should prompt decisions regarding allogeneic stem cell transplantation possibly irrespective of conventional risk scores. Another novel finding of the study was the striking association between thrombocytopenic phenotype and triple-negativity, found in 35% of the patients, which might contribute to explain the previously reported unfavorable impact of TN;17 conversely, less than 3% of anemic patients were TN. Though splenomegaly is enlisted among the diverse mechanisms contributing to anemia in PMF, we did not find meaningful differences between MyP and CyP patients as regarded to frequency and size of splenomegaly, if not for more splenomegaly patients in the thrombocytopenic category. Such findings might help to explain the lack of correlations between spleen volume reduction by ruxolitinib and/or fedratinib and improvement of anemia and thrombocytopenia, which may actually worsen in most cases representing on-target JAK2 inhibition. On the other hand, this gives support to the prevalent ACVR1 (Activin A receptor, type 1)-dependent inhibition, more than spleen volume reduction, as the mechanism underlining the favorable effects of momelotinib and pacritinib in anemic patients. Limitations of the present study include the lack of detailed information about the impact of transfusion dependence in severely anemic patients, the profile of non-driver mutations, in particular U2AF1 that was associated with anemia and/or thrombocytopenia,18, 19 and treatments used. This notwithstanding, we hope that this set of data will be useful for designing future clinical trials with molecules specifically addressing the unmet needs of cytopenic PMF patients.
ACKNOWLEDGMENTS
Open access publishing facilitated by Aziende Socio Sanitarie Territoriale Papa Giovanni XXIII, as part of the Wiley – SBBL agreement.
AUTHOR CONTRIBUTIONS
Paola Guglielmelli and Alessandro M. Vannucchi collected data, analyzed data, and drafted the manuscript together with Arianna Ghirardi. Arianna Ghirardi, Alessandra Carobbio, and Arianna Masciulli performed statistical analysis. Lucrezia Morrone, Barbara Mora, Elisa Rumi, Ana Triguero, Maria C. Finazzi, Helna Pettersson, Valentina Boldrini, Daniele Vanni, Alessandro Rambaldi, Francesco Passamonti, Alberto Alvarez-Larràn, and Bjorn Andreasson contributed data, Tiziano Barbui was responsible for the ERNEST1-2 project and contributed to data analysis and manuscript drafting. All authors provided critical inputs and revised and approved the final draft.
CONFLICT OF INTEREST STATEMENT
Paola Guglielmelli received fees for lectures and advisory boards from Novartis and GSK. Alessandro M. Vannucchi received fees for lectures and advisory boards from Novartis, GSK, AbbVie, AOP, and Italfarmaco. Barbara Mora fees for Lectures for Novartis and GSK, and Advisory Board for GSK. Francesco Passamonti received honoraria for lectures and advisory boards from Novartis, Bristol-Myers Squibb/Celgene, Sierra Oncology, AbbVie, Janssen, Roche, AOP Orphan, Karyopharm, Kyowa Kirin, and MEI. Alessandro Rambaldi received fees for consultancies and participation in meetings, boards, and symposia sponsored by Amgen, Pfizer, Novartis, Kite-Gilead, Jazz, Astellas, Abbvie, Incyte, and Omeros. Tiziano Barbui received Gsk research grant support from GSK, and fees for the advisory board of Italfarmaco, AOP, and Ionis, and for lectures from AOP. For Elisa Rumi, Alberto Alvarez-Larràn, Daniele Vanni, Arianna Masciulli, Arianna Ghirardi, Alessandra Carobbio, Lucrezia Morrone, Valentina Boldrini, AT, Maria C. Finazzi, Helna Pettersson, and Bjorn Andreasson, no relevant conflicts of interest were declared.
ETHICS STATEMENT
The project, promoted by the European LeukemiaNet (ELN) was coordinated by FROM (Research Foundation at Papa Giovanni XXIII Hospital in Bergamo, Italy). The Institutional Review Board and Ethical Committee of each participating Centre approved the study, which was conducted in accordance with the Declaration of Helsinki. Patients signed an informed consent.
FUNDING
The ERNEST registry is supported by Novartis Pharma through a research collaboration. The study was also supported by a research grant by AIRC 5×1000 called “Metastatic disease: the key unmet need in oncology” to MYNERVA project, #21267 (MYeloid NEoplasms Research Venture AIRC). A detailed description of the MYNERVA project is available at https://progettomynerva.it (A.M.V, P.G.); and Bando Ricerca Finalizzata Ministero della Salute project NET-2018-12365935 (P.G., F.P.).
Open Research
DATA AVAILABILITY STATEMENT
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Aggregated data only are available upon reasonable request.




