Outcomes of neoadjuvant chemotherapy using gemcitabine and cisplatin in muscle invasive bladder cancer: A retrospective analysis of the patient and treatment factors in a single institute
Abstract
Background
Meta-analysis had shown a significant 5% absolute survival benefit in favour of neoadjuvant chemotherapy (NAC) with cisplatin-based chemotherapy before radical cystectomy (RC) and pelvic lymphadenectomy (PLND) for patients with muscle invasive bladder cancer (MIBC). Those who had pathological complete response (pCR) to NAC could have long-term progression-free survival (PFS) and overall survival (OS).
Aim
To identify the treatment and patient factors which could predict a pCR to NAC and the associated PFS and OS in a single institute.
Methods and Results
We retrospectively reviewed the records of patients who had received NAC with gemcitabine and cisplatin (GC) in our centre from January 2004 to December 2017. The patients' age, tumour stage, baseline estimated glomerular filtration rate (eGFR), chemotherapy chart, and pathological information were recorded.
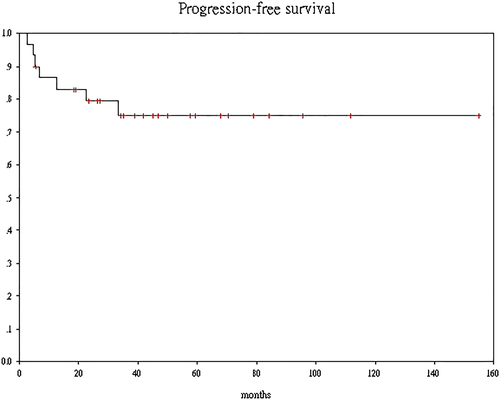
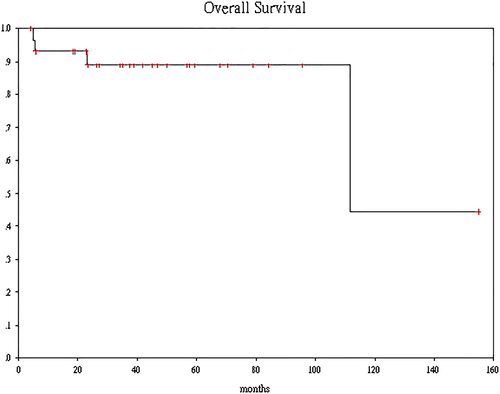
There were 25 men and five women who had received NAC followed by RC. pCR was noted in the surgical specimen of 11 (37%) patients. The mean dose of gemcitabine was significantly higher in the pCR group than the non-pCR group (9850 vs 7852 mg, P = 0.039) as was the dose-intensity of cisplatin (87.4% vs 71.3%, P = 0.044). After a median follow-up of 38 months (range 4.3-154), seven patients had disease progression. The estimated 3-year PFS is 74.9% (95% confidence interval [CI], 66.7%-83.3%). None of the patients who achieved pCR relapsed, while six out of seven patients who had pN1 disease developed distant metastasis (DM). Only two patients died of DM while two other patients died of unrelated causes. The estimated 3-year OS is 88.9% (95% CI 82.8%-95%).
Conclusions
We have demonstrated that the dose intensity of GC is a major determinant of pCR, which predicts longer RFS and OS. Further research in gene expression profiling of MIBC to help selecting patient for NAC is needed.
1 INTRODUCTION
Radical cystectomy (RC) with pelvic lymphadenectomy (PLND) is the gold standard of treatment to provide cure for patients with muscle invasive bladder cancer (MIBC). However, the survival rate after surgery alone for patients who have clinical stage T2-4aN0M0 disease at presentation is still low. It is because MIBC has high chance of distant and lymph node metastasis, which may not be picked up by usual imaging studies preoperatively. Most of these patients who have occult metastatic disease will succumb to overt metastasis at distant sites within 3 years of RC. The European Urology Association (EAU) and the National Cancer Care Network (NCCN) have published guidelines1, 2 that recommend the use of neoadjuvant chemotherapy (NAC) before RC since a meta-analysis3 indicates a significant 5% absolute survival benefit in favour of NAC with cisplatin combination chemotherapy. However, the intake of NAC within the oncological community still remains low, because NAC can cause many toxicities and may not benefit every patients. Many practicing oncologists and urologists still find it difficult to select patients for NAC. Moreover, it remains uncertain which treatment and patient factors could predict a favourable outcome to NAC.
Since 2004, patients with a diagnosis of MIBC in Tuen Mun Hospital were referred to a multidisciplinary team clinic for discussion about the use of NAC. Here, we report the experience and outcomes of patients who have received NAC and then RC over the past 14 years. We also analyse their outcomes in terms of pathological complete remission (pCR) after NAC according to the patient and treatment factors.
2 MATERIALS AND METHODS
2.1 Patients
This is a retrospective review of the clinical records of all patients who have received NAC for MIBC from October 2004 to December 2017. Only patients who received NAC followed by RC were included. The patients' age, clinical stage, renal function, baseline estimated glomerular filtration rate (eGFR), chemotherapy chart, pathological information from the transurethral bladder resection (TUR), and RC specimen were recorded. Patients were classified as high risk (HR) or low risk (LR) according to the criteria by Culp et al.4
2.2 Treatment
The chemotherapy regimens consist of (1) gemcitabine 1000 mg/m2 (D1, D8, and D15), cisplatin 70 mg/m2 (D1 only) every 4 weeks for three cycles or (2) gemcitabine 1000 mg/m2 (D1 and D8), cisplatin 50 mg/m2 (D1 and D8) given with aprepitant, dexamethasone, and a 5-HT3 antagonist every 3 weeks for three cycles. Patients were followed up weekly during chemotherapy with close monitoring on the acute chemotherapy toxicities including complete blood count and serum biochemistry. Chemotherapy toxicities were recorded and graded according to the Common Toxicity Criteria (CTCAE v4). Chemotherapy dose were adjusted according to their tolerance. The absolute total dose of gemcitabine and cisplatin (GP) were recorded as well as the dose intensity calculated as the percentage of given dose divided by the initial planned dose.
After NAC patients were referred back to the urologist and definitive surgery including RC, PLND with urinary diversion or bladder reconstruction were arranged.
2.3 End point
The primary end point of the study is the pCR defined as absence of invasive malignancy in the final RC and LND surgical specimen. Secondary end point include progression-free survival (PFS), defined as the date of definitive surgery to the date of documented progressive disease and overall survival (OS), defined as the date of definitive surgery to the date of death from any cause. Patients were followed up by the urologists and oncologists of Tuen Mun Hospital. Patients who did not have an event had their survival data censored at the last date of their follow-up.
2.4 Statistical analysis
Patients' age, baseline eGFR, chemotherapy dose, and dose intensity were compared according to their pCR status using the independent t test. P value < 0.05 was considered statistically significant. PFS and OS were estimated using the Kaplan-Meier product limit method. Statistical analyses were executed using IBM-SPSS Statistics, version-25 (IBM Corp., Armonk, NY).
3 RESULTS
3.1 Patients and tumour characteristics
There were 25 men and five women who received NAC followed by RC from 2004 to 2018. Their age ranged from 35 to 75 (median 60) at the time of diagnosis. Two patients who had renal impairment due to ureteric obstruction by bladder tumour had percutaneous nephrostomy (PCN) drainage performed prior to chemotherapy. All the other patients had a baseline eGFR of more than 50 mL/min. Tumour characteristics are depicted in Table 1. Fourteen patients had T3/T4 disease on baseline CT scan. Four patients had lymph nodes metastasis as defined by short axis diameter larger than 1 cm on CT scan. Lymphovascular permeation (LVI) was present in the TUR specimen in nine patients. Overall there were 18 patients who belong to the HR group and six of them had two or more HR features.
| High Risk (HR) Feature | Number |
|---|---|
| T3/T4 | 14 |
| N1/N2/N3 | 4 |
| LVI in TUR specimen | 9 |
| Hydonephrosis who required percutaneous nephrostomy (PCN) drainage | 2 |
| Micropapillary/neuroendocrine/small cell carcinoma | 0 |
| Squamous cell/poor differentiated carcinoma | 2 |
| High risk patients | 18 |
| Patients with a least two high risk features | 6 |
- Abbreviation: TUR, transurethral bladder resection.
3.2 Chemotherapy compliance and toxicities
There were two patients who refused further chemotherapy after the first course because of gemcitabine skin rash. Another patient who had history of irradiation to neck for laryngeal cancer developed grade 3 mucositis after the first course of GP because of radiation recall. One patient who had history of chronic obstructive pulmonary disease (COPD) developed pneumothorax and chemotherapy had to be stopped after the second cycle. Another patient developed deep vein thrombosis from rapid disease progression, and she went on to have RC and PLND immediately after the second cycle. One patient did not receive cycle 3 D8 chemotherapy because of delayed recovery from neutropenia. All the other patients completed three cycles of NAC as planned. Overall chemotherapy was well tolerated with grade 3 or above neutropenia in 14 (47%) and thrombocytopenia in three (10%) patients (Tables 2 and 3).
| Number (percent) | Reason of Termination | |
|---|---|---|
| One cycle | 3 (10) |
1. Stopped after C1D1 because of Gemzar skin reaction 2. Stopped after C1D8 because of Gemzar skin reaction 3. Mucositis and severe renal impairment after cycle 1 GP |
| Two cycles | 3 (10) |
1. Pneumothorax after second cycle due to COPD 2. Stop because of progressive disease and DVT 3. C3D8 not given because of neutropenia |
| Three cycles | 24 (80) |
- Abbreviations: COPD, chronic obstructive pulmonary disease; DVT, deep vein thrombosis.
| Toxicity | Grade 1 | Grade 2 | Grade 3 |
|---|---|---|---|
| Anaemia | 26 | 0 | 0 |
| Neutropenia | 11 | 12 | 2 |
| Thrombocytopenia | 19 | 2 | 1 |
| Skin rash | 5 | 0 | 0 |
| Nausea/dyspepsia | 8 | 0 | 0 |
| Mucositis/mouth ulcer | 1 | 1 | 0 |
3.3 Pathological complete response
All patients received radical surgery after their recovery from the acute toxicities from NAC. The median time from completion of chemotherapy to surgery was 55 days. pCR was noted in the surgical specimen of 11 (37%) patients. There was no significant difference in the age and renal function between the patients who achieved pCR and non-pCR. However, the mean dose of gemcitabine was significantly higher in the pCR group than on the non-pCR group (9850 vs 7852 mg, P = 0.039) as was the dose-intensity of cisplatin (87.4% vs 71.3%, P = 0.044) (Table 4).
| pCR | Non-pCR | P value | |
|---|---|---|---|
| Mean age | 62 | 58 | 0.253 |
| Mean eGFR, ml/min | 87 | 76 | 0.125 |
| Mean gemcitabine dose, mg | 9850 | 7852 | 0.039 |
| Mean gemcitabine dose, % | 89.9 | 79.1 | 0.139 |
| Mean cisplatin dose, mg | 406 | 354 | 0.163 |
| Mean cisplatin dose, % | 87.4 | 71.3 | 0.044 |
3.4 Survival and follow-up
There was no patient who had lost to follow-up. After a median follow-up length of 38 months (range 4.3-154) after surgery, seven patients had disease progression. None of the patients who achieved pCR relapsed, while six out of seven patients who had pN1 disease after RC and LND developed distant metastasis (DM) subsequently. The estimated 3-year PFS was 74.9% (95% confidence interval [CI], 66.7%-83.3%) (Figure 1). Among them, two patients who developed brain metastasis as the only site of recurrence were salvaged with definitive surgery or brain irradiation. One patient who developed liver metastasis was receiving immunotherapy, and the two remaining patients were still surviving with best supportive care alone for their peritoneal metastases at the time of data cut-off. Only two patients who developed DM had died while two patients who did not have disease recurrence died of unrelated causes. One of them died of biopsy proven hepatocellular carcinoma more than 10 years after RC; another died of small bowel gangrene at sixth month after RC. The estimated 3-year OS was 88.9% (95% CI, 82.8%-95%) (Figure 2).
4 DISCUSSION
Since the publication of the landmark SWOG study by Grossman et al5 in 2003, NAC has become an option for patients who are diagnosed with MIBC. However, the combination chemotherapy regimen comprising methotrexate, doxorubicin, vinblastine, and cisplatin (MVAC) without growth factor support is considered too toxic with grade 3 or above stomatitis and granulocytopenia occurred in 10% and 50% patients, respectively. The combination of gemcitabine and cisplatin (GP) has been shown to be equally effective as the MVAC regimen but less myelotoxic for patients with locally advanced or metastatic bladder cancer.6 However, there is no head-to-head comparison between GP and MVAC in the neoadjuvant setting. The better toxicity profile of GP may lead to higher completion rate of NAC and therefore better treatment outcomes. Nevertheless, skepticism on the toxicities and efficacy of NAC and patients' reluctance to defer definitive surgery render recruitment of patients into further neoadjuvant trials difficult.
The outcomes of using GP as NAC in our institute appeared to be better than that of using MVAC in the SWOG study. Eleven out of 30 (37%) patients achieved pCR. Besides, our 3-year PFS and OS were 75% and 89%, respectively, while the 3-year OS in the SWOG trial was about 60% only. This may be due to selection bias with better patients and tumour characteristics in our cohort. However, 18 (60%) of our patients actually belong to the HR group by the MD Anderson Cancer Center criteria. Most of them should have died of disease if NAC had not been given. A genuine superior treatment effect by GP over MVAC cannot be ruled out. In fact, we can observe a dose-response relationship between chemotherapy and pCR in the RC specimen. Patients in the pCR group received a higher dose and also dose intensity (greater than 80%) of gemcitabine and cisplatin. It is generally believed that pCR is correlated with long-term survival as well. Therefore, the favourable outcomes of our patients may be due to their better tolerance and compliance to the chemotherapy treatment. Overall, GP was well tolerated, and there was no mortality attributable to treatment after chemotherapy and definitive surgery in our centre.
Research on the biomarkers that can predict better response and outcome to NAC are still ongoing to help optimize delivery and increase uptake of NAC in MIBC patients. Seiler et al7 showed that there may be an impact of molecular subtype in MIBC on predicting response and survival after NAC. By using gene expression profiling techniques, they could subdivide MIBC into four types: claudin-low, basal, luminal, and luminal-infiltrative. Only the patients with TUR specimen showing basal-type tumour could derive the survival advantage from NAC. Patients with claudin-low tumours had the worst survival outcome irrespective of NAC, while patients with luminal-type tumour did equally well with or without NAC. It is also postulated that the more “inflamed” tumour such as the luminal-infiltrative subtype should respond better to immunotherapy. Necchi et al8 in a phase 2 study indicated that pembrolizumab could be a worthwhile neoadjuvant therapy for the treatment of MIBC when limited to patients with PD-L1–positive or high-TMB (tumour mutation burden). Research of using neoadjuvant and adjuvant immunotherapy with check point inhibitors are still ongoing, and patient recruitments into these trials are encouraged to shed more light into future clinical practice.
Several groups9, 10 have reported high OS rates in patients with MIBC who forego immediate RC after a complete clinical response (negative cystoscopy, urine cytology, and cross-sectional imaging). In our cohort, none of the patient who achieved pCR after NAC have recurrence after a median follow-up of more than 3 years. These patients may have been cured of MIBC with NAC alone even without definitive surgery. It would be worthwhile to perform further tissue-based genomic study to elucidate biomarkers in these good responders, as the MSKCC11 had also performed genomic analysis on a subset of these TURBT samples and identified that DNA excision repair protein ERCC-1 alterations predicted a cisplatin response.
5 CONCLUSION
MIBC can be a systemic disease and RC alone may not be enough to provide cure to all patients. Selection of patients for NAC is an important and difficult decision process. We have demonstrated that by selecting patients who have optimal renal function to tolerate adequate dose of cisplatin and gemcitabine can achieve excellent outcomes in terms of pCR, PFS, and OS. The limitations of our study include (1) the use of a retrospective database limited our ability to investigate other sources of potential bias, (2) we used a single centre of accrual with small number of patients (30 only), and (3) short follow-up (median 38 months), and therefore, we cannot conclude cure of those patients who were still alive without recurrence at the time of analysis. Longer follow-up and further research into the molecular science of MIBC to guide neoadjuvant/adjuvant therapy is warranted.
ETHICAL APPROVAL
The study was conducted with approval from the New Territories West Cluster Clinical and Research Ethics Committee.
ACKNOWLEDGEMENT
The authors thank Dr Man Chi Wai for his critical reading of the manuscript.
CONFLICT OF INTEREST
All authors have no conflicts of interest to disclose. All authors had full access to the data, contributed to the study, approved the final version for publication, and take responsibility for its accuracy and integrity.
AUTHORS' CONTRIBUTIONS
All authors had full access to the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. Conceptualization, Eric KC LEE, Peggy SK CHU; Methodology, Eric KC LEE; Investigation, Eric KC LEE, W CHAN; Formal Analysis, Eric KC LEE; Resources, Eric KC LEE, WH MUI; Writing - Eric KC LEE, WH MUI, F.M.L.; Writing - Review & Editing, Eric KC LEE, Peggy SK CHU; Visualization, Eric KC LEE; Supervision, Frank CS WONG; Funding Acquisition, N/A.




