Advances in transplantation for lymphomas resulting from CIBMTR lymphoma working committee's research portfolio: A five-year report (2013-2018)
Abstract
The Center for International Blood and Marrow Transplant Research (CIBMTR) is a research collaboration between the National Marrow Donor Program (NMDP)/Be The Match and the Medical College of Wisconsin (MCW). The CIBMTR collaborates with the global scientific community to advance hematopoietic cell transplantation (HCT) and cellular therapy worldwide to increase survival and enrich quality of life for patients. The observation research program within CIBMTR is organized into 15 working committees. This review is aiming to highlight the observational research studies published by the CIBMTR Lymphoma Working committee over the last 5 years (2013-18) and to summarize how these studies have impacted the field by helping inform clinical practice in scenarios where prospective data from high quality randomized trials were not available or where owing to the rarity of a particular transplant indication such data were unlikely to be generated, outside the setting of a large observational research database. The salient findings reviewed include; (a) studies supporting role of autologous HCT in diffuse large B-cell lymphoma (DLBCL) patients with sensitive relapse of disease within 1 year of diagnosis, (b) role of autologous HCT vs allogeneic HCT in follicular lymphoma patients with early therapy failure, (c) prognostic scoring system development for classical Hodgkin lymphoma and DLBCL patients with prior autograft failure, (d) defining the role of alternative donor transplantation in lymphomas, (e) evaluating appropriate conditioning regimens for HCT in lymphoma, and (f) outcomes of HCT in rare lymphoid malignancies.
1 INTRODUCTION
The Center for International Blood and Marrow Transplant Research (CIBMTR) is a research collaboration between the National Marrow Donor Program/Be The Match and the Medical College of Wisconsin. The CIBMTR collaborates with the global scientific community to advance hematopoietic cell transplantation (HCT) and cellular therapy worldwide to increase survival and enrich quality of life for patients. The CIBMTR facilitates critical observational and interventional research through scientific and statistical expertise, a large network of transplant centers, and a unique and extensive clinical outcomes database.1, 2 The prospective and observational research is accomplished through scientific and statistical expertise, a large network of transplant centers and a clinical database of more than 475 000 patients. Support for the program is provided, in part, by grants and contracts from the National Institute of Health and Health Resources and Services Administration. The observation research program within CIBMTR is organized into 15 working committees. These working Committees shape the observational research that leads to publications for CIBMTR.
This review is aiming to highlight the observational research studies published by the CIBMTR Lymphoma Working Committee (LYWC) over the last 5 years (2013-18) and to summarize how these studies have impacted the field by helping inform clinical practice, especially in scenarios where prospective data from high quality randomized trials were not available or where owing to the rarity of a particular transplant indication such data were unlikely to be generated, outside the setting of a large observational research database.
2 FOLLICULAR LYMPHOMA
Although follicular lymphoma (FL) patients generally respond to initial therapy, disease relapse is common. Tools to identify relapsed FL patients destined to do poorly have been lacking, leading to great controversy in selecting appropriate candidates for HCT. The CUP trial conducted in the prerituximab era compared salvage chemotherapy alone to chemotherapy followed by either purged or unpurged autologous (auto)-HCT consolidation, in relapsed/chemosensitive FL. This trial, despite poor accrual (n = 89) and premature closure, showed a significant progression-free survival (PFS) and overall survival (OS) benefit in favor of auto-HCT.2 However, the current clinical relevance of this trial has been questioned, since it was conducted before modern chemotherapies, immunotherapies and targeted agents became available. In addition, the curative potential of high-dose therapy and auto-HCT (if any) in relapse FL has been questioned. Large set of registry data (European Society for Blood and Marrow Transplantation [EBMT] and CIBMTR) do not show a clear plateau in relapse rates postautografting in FL.3, 4 In addition, the risk of second malignancies after auto-HCT (5-15%) has also dampened the enthusiasm for this modality.3, 5 In contrast to high-dose therapy, allogeneic (allo)-HCT is a potentially curative option for FL. A CIBMTR LYWC study reported in 2003 compared auto- vs allo-HCT in the prerituximab era and showed a significantly reduced risk of relapse after allo-HCT, with a clear plateau in relapse curves.4
3 AUTO VS ALLO-HCT IN RELAPSED/REFRACTORY FL
The best way to assess, if there is a preferred HCT approach (auto vs allo) for relapsed FL, is obviously a well-designed randomized trial. The Blood and Marrow Transplant Clinical Trials Network (BMT CTN) 0202 trial tried to answer this question by comparing auto-HCT to a reduced-intensity conditioning (RIC) allo-HCT in a randomized fashion in FL. Unfortunately, the trial was closed early due to poor accrual (N for auto-HCT = 22 and N for allo-HCT = 8).6 For the 30 patients enrolled in the BMT CTN 0202 study, the 3-year OS was 73% with auto-HCT vs 100% following allo-HCT, and 3-year PFS was 63% in the auto-HCT group vs 86% in the allo-HCT cohort. The fact that it is unlikely that another randomized study in the modern era will tackle this question, means that we have to rely on high quality observational databases for decision making.
The CIBMTR LYWC, hence aimed to compare auto- vs RIC allo-HCT as the first transplantation approach in relapsed FL. Included patients were required to received rituximab before HCT to mimic rituximab-era. The first publication from this project limited to WHO grade 1-2 FL suggested that auto- and allo-HCT, when applied as the first transplantation approach provided comparable outcomes in FL; however, the risk of relapse was substantially lower, and nonrelapse mortality (NRM) significantly higher after allo-HCT. In long-term disease-free survivors (>2 years), allo-HCT was associated with a survival benefit (Table 1).7 The second publication from this project limited to patients with grade 3 FL also showed similar findings8 (Table 1). The limitation of these publications is that we cannot be sure whether all FL patients included in these analyses necessarily had a reasonably high-risk disease to warrant transplantation. In addition, while there was a hint of a benefit in long-term survivors in favor of allografting, during pre-HCT patient consultation it is difficult to justify using one modality over another using these data (since benefit of one approach is contingent upon surviving early mortality after allo-HCT).
|
Author (Year) |
Number | NRM | Relapse rate | PFS | OS | Comments |
|---|---|---|---|---|---|---|
|
van Besien (2003)a |
597 vs 176 (auto vs allo) |
8% vs 30% (5 years; auto vs allo) |
58% vs 21% (5 years; auto vs allo) |
– |
55% vs 51% (5 years; auto vs allo) |
Prerituximab era study |
| Klyuchnikov (2015) |
250 vs 268 (auto vs allo) |
5% vs 26% (5 years; auto vs allo) |
54% vs 20% (5 years; auto vs allo) |
51% vs 58% (5 years; auto vs allo) |
74% vs 66% (5 years; auto vs allo) |
Limited to grade 1 or 2 FL, rituximab-era and RIC allo-HCT |
| Klyuchnikov (2016) |
136 vs 61 (auto vs allo) |
4% vs 27% (5 years; auto vs allo) |
61% vs 20% (5 years; auto vs allo) |
36% vs 51% (5 years; auto vs allo) |
59% vs 54% (5 years; auto vs allo) |
Limited to grade 3 FL, rituximab-era and RIC allo-HCT |
|
Casulo (2018) |
175 vs 174 (auto vs no auto) |
– | – | – |
73% vs 60% (5 years; auto vs no auto) |
Limited to FL with POD24. Study showed an OS in favor of auto-HCT if done within 1 year of experiencing POD21 |
|
Smith (2018) |
240 vs 105 vs 94 (auto vs MSD vs MUD) |
5% vs 17% vs 33% (5 years auto vs MSD vs MUD) |
58% vs 31% vs 23% (5 years auto vs MSD vs MUD) |
38% vs 52% vs 43% (5 years auto vs MSD vs MUD) |
70% vs 73% vs 49% (5 years auto vs MSD vs MUD) |
Limited to FL with POD24. Auto-HCT and MDS provide comparable outcomes |
- FL: follicular lymphoma; FCR: fludarabine, cyclophosphamide, rituximab; ATG: antithymocyte globulin; Flu/Bu: fludarabine/busulfan; MSD: matched sibling donor transplantation; MUD: matched unrelated donor transplantation; NRM: nonrelapse mortality; PFS: progression-free survival; OS: overall survival; GVHD: graft vs host disease; auto-HCT: autologous hematopoietic cell transplantation; allo: allogeneic; POD24: FL patients with progression of disease within 2 years of starting first-line chemoimmunotherapies.
- a Only unpurged autologous HCT data shown.
4 HIGH-RISK FL: CAN REGISTRY DATA INFORM DECISION MAKING?
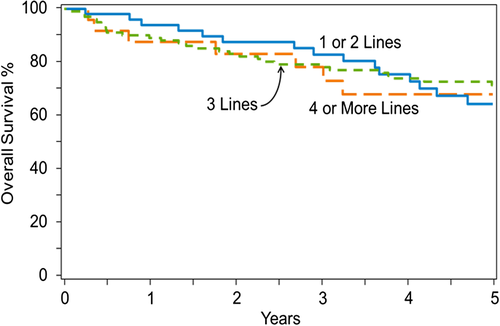
Choosing auto- vs allo-HCT in relapsed FL remains controversial and largely a factor of treating physician and transplant center preferences. Based on mostly prerituximab era data showing greater benefit of auto-HCT early in the disease course, it is a common practice in many centers to limit auto-HCT use in patients who have received 2-3 prior lines of therapies. However, in the rituximab-era unpublished data from the CIBMTR show that lines of prior therapies might not be predictive of auto-HCT outcomes (unadjusted outcomes shown in Figure 1).
The National LymphoCare Study (NLCS) in an important publication reported that early failure of first-line chemoimmunotherapies (<2-years), the so-called POD24 (progression of disease in 24 months from diagnosis), identified a biologically high-risk subset of FL patients with an extremely poor prognosis (5-year OS ~50%).9 These key findings led to an important collaborative study between CIBMTR/NLCS designed to investigate the role of auto-HCT in FL patients with POD24.10 Undertaking an observation study, tackling a transplant vs no transplant questions requires addressing several potential biases in study design. For example, a non-HCT dataset (NLCS in this case) is likely to be enriched for patients at advanced age and those who experience early death after POD24 (eg, death even before start of salvage) precluding a subsequent HCT procedure. Likewise, HCT dataset (in this case CIBMTR) can be enriched for younger patients and those with chemosensitive disease. To address these biases, we excluded patients >70 years of age at FL diagnosis (hence removing patients unlikely to be referred for HCT), but imposed no upper limited on patient age at the time of auto-HCT (so as not to enrich for young, fit patients in HCT cohort). Study excluded NLCS patients who did survive long enough after POD24 to receive salvage therapies and a subsequent auto-HCT. Lack of chemosensitive disease was not considered an exclusion for the auto-HCT CIBMTR patient cohort. Interestingly, the analysis of this carefully selected patient cohort (all meeting NLCS definition of POD24), showed that early application of auto-HCT (ie, within 1 year of POD24) in NLCS-defined high-risk FL patients was associated with an OS benefit when compared to similar patients not undergoing auto-HCT (5 year OS 73% vs 60%; P = 0.02) (Table 1).10 These findings were replicated in a Propensity Score-matched patient population, as well as a simple one-to-one matched subset. This important contribution from the CIBMTR/NLCS will hopefully inform clinical practice about the optimal timing of auto-HCT in high-risk FL.
The next logical question to undertake was whether an allo-HCT offered superior disease control and survival compared to auto-HCT in FL patients with POD24. In a recent CIBMTR study, we compared auto-HCT with either matched sibling donor (MSD) or matched unrelated donor (MUD) allo-HCT in FL with POD24. Among the 440 POD24 FL included in the analysis with a median follow-up of 69 to 73 months, the adjusted probability of 5-year OS was significantly higher after auto-HCT (70%) or MSD-HCT (73%) vs MUD HCT (49%; P = 0.0008). The 5-year adjusted probability of NRM was significantly lower for auto-HCT (5%) vs MSD (17%) or MUD HCT (33%; P < 0.0001). The 5-year adjusted probability of disease relapse was lower with MSD (31%) or MUD HCT (23%) vs auto-HCT (58%; P < 0.0001).11
These two studies have helped clarify the role of HCT in high-risk FL patients with POD24. Among such patients with disease responsive to salvage therapies, auto-HCT should be strongly considered. Allo-HCT preferably with MSD is best reserved for POD24 patient unlikely to benefit from auto-HCT consolidation (eg, those with heavily pretreated disease, active marrow involvement, chemorefractory disease or those with failed stem cell collection).
5 DIFFUSE LARGE B-CELL LYMPHOMA
Diffuse Large B-cell Lymphoma (DLBCL) is the most common type of non-Hodgkin lymphoma (NHL) and one of the most common lymphoma subtypes undergoing HCT in the United State. This histological subtype has been a subject of several prospective and retrospective HCT reports. The landmark PARMA study firmly established auto-HCT consolidation as standard-of-care in relapsed/chemosensitive DLBCL, by demonstrating a survival benefit over salvage chemotherapy alone.12 The CORAL study, while reaffirming the role of auto-HCT in relapsed DLBCL in the rituximab-era, also hinted at the possible futility of this approach in patients experiencing early failure of rituximab-based therapies.13 The findings from the CORAL study, led many investigators to question the role of auto-HCT in DLBCL patients relapsing within 1 year of initial diagnosis. At the CIBMTR, we evaluated the role of auto-HCT in relapsed DLBCL in the rituximab-era, relative to the timing of chemoimmunotherapy failure and noted that DLBCL with early rituximab failure had a higher risk of disease relapse in the first year after transplantation. However, beyond that point no survival difference was noted.14 Thus, auto-HCT remains the standard-of-care for chemosensitive relapsed/refractory DLBCL.
Approximately, 30%-40% of DLBCL will relapse/progression after auto-HCT. In addition, auto-HCT is not appropriate for patients with refractory disease. For such patients, allo-HCT might be a curative option. Easy-to-use prognostic models able to predict allo-HCT outcomes in DLBCL relapsing after a prior auto-HCT could be clinically useful. Unlike auto-HCT, pre allo-HCT positron emission tomography (PET) scans have not consistently been shown to predict transplantation outcomes.15 A CIBMTR study demonstrated that three variables that determined allo-HCT outcomes in DLBCL patients with prior auto-HCT failure, included Karnofsky performance status (KPS) <80, chemoresistance, and auto-HCT to allo-HCT interval <1-year. These three adverse prognostic factors were used to construct a prognostic model for PFS assigning KPS<80 four points, auto-HCT to allo-HCT interval <1-year two points and chemoresistant disease at allo-HCT five points. This CIBMTR prognostic model classified patients into four groups: low-risk (0 points), intermediate-risk (2-5 points), high-risk (6-9 points), or very high-risk (11 points), predicting 3-year PFS of 40%, 32%, 11%, and 6%, respectively, with 3-year OS probabilities of 43%, 39%, 19%, and 11%, respectively.16 This model can be used to identify patients unlikely to benefit from allo-HCT, or those where relapse prevention strategies postallografting should be evaluated.
5.1 Current Limitations for CIBMTR Data for DLBCL
Since CIBMTR does not capture data on lymphoma patients not undergoing HCT, it is not a suitable data source to identify patients unlikely to benefit from a HCT procedure. However, prognostic markers able to identify relapsed/refractory DLBCL patients unlikely to benefit from auto-HCT are urgently needed to investigate alternative therapies. In the recently multicenter retrospective analysis, the presence of at least two out of three risk factors (1. progression while on frontline DLBCL therapy, 2. MYC rearrangement and 3. Intermediate-high/high NCCN-IPI) in relapsed/refractory DLBCL predicted a 2-year OS of only 13% after a subsequent auto-HCT.17 In addition, the information about genomic profile of DLBCL (eg, cell-of-origin, protein expression of c-myc, bcl-2, bcl-6 or data about c-myc, bcl-2, bcl-6 gene rearrangements or alternations) has not been, historically captured by CIBMTR. These important parameters are now part of registry data capture forms (case report forms) released in early 2018 and hopefully in coming years will enhance our ability to analyze HCT outcomes, relative to these important genomic parameters.
6 TRANSFORMED FL
Histologic transformation of FL to DLBCL is not uncommon and generally portends a poor prognosis except in a (now rare) subset of rituximab naïve patients with limited stage disease at the time of transformation, who can expect prolonged disease control with anthracycline-based chemoimmunotherapy salvage. In 2014, CIBMTR reported HCT outcomes in 141 subjects with biopsy-proven DLBCL transformed from FL.18 Report included 108 patients undergoing auto-HCT and 33 receiving allo-HCT. One-year NRM was 8% and 5-year PFS and OS were 35% and 50%, respectively, following auto-HCT. In contrast, allo-HCT was associated with a 1-year NRM of 41%, 5-year PFS of 18% and 5-year OS of 22%. This study suggested that in transformed FL patients with chemosensitive disease auto-HCT was able to provide sustained remissions in a high proportion of subjects.
7 MANTLE CELL LYMPHOMA
The role and timing of transplantation in mantle cell lymphoma (MCL) has been a matter of controversy.19 We examined the timing (early vs late) of auto-HCT and RIC allo-HCT in an important registry analysis.20 In this analysis, early transplantation was defined as HCT in first partial or complete remission with no more than two prior lines of chemotherapy. The late transplantation cohort was defined as all the remaining patients. Interestingly, both auto-HCT and RIC allo-HCT resulted in similar OS from transplantation for both the early (at 5 years: 61% auto-HCT vs 62% RIC allo-HCT; P = 0.95) and late cohorts (at 5 years: 44% auto-HCT vs 31% RIC allo-HCT; P = 0.20). An important observation from this analysis was that OS was superior in MCL patients undergoing early vs late auto-HCT. Results from this analysis support the use of upfront auto-HCT consolidation in MCL patients, with allo-HCT best reserved for patients with more advanced disease status, later in the disease course.
8 PROGNOSIS OF CHILDREN, ADOLESCENTS, AND YOUNG ADULTS (CAYA) WITH HODGKIN LYMPHOMA
The role of auto-HCT in relapsed classical Hodgkin lymphoma with chemosensitive disease is well established. However surprisingly, little data are available regarding the outcomes of this procedure in the children, adolescents, and young adult (CAYA) population. We recently reported outcomes of 606 CAYA patients (median age 23 years) with relapsed/refractory classical Hodgkin lymphoma who underwent an auto-HCT between 1995 and 2010.21 The probabilities of PFS at 1, 5, and 10 years post-HCT were 66%, 52%, and 47%, respectively. Multivariate analysis for PFS demonstrated that at the time of auto-HCT; KPS ≥90, no extranodal involvement and chemosensitive disease predicted significantly improved PFS. Patients with time from diagnosis to first relapse of <1 year had a significantly inferior PFS. A prognostic model for PFS was developed that stratified patients into low-, intermediate-, and high-risk groups, predicting for 5-year PFS probabilities of 72%, 53%, and 23%, respectively. This large study identified a group of CAYA patients with relapsed/refractory Hodgkin lymphoma with very high risk of progression after auto-HCT. We believe that our prognostic model could be used for identifying Hodgkin lymphoma patients with ultra high risk of relapse after high-dose therapies and that such patients should be targeted for novel therapeutic and/or maintenance approaches post-auto-HCT.
9 DOES PET IMAGING DICTATE OUTCOMES OF ALLO-HCT IN LYMPHOMA?
As mentioned in preceding sections that pre allo-HCT PET scans have not consistently been shown to predict transplantation outcomes. The largest analysis tackling this question was under taken by CIBMTR that included 336 chemotherapy-sensitive lymphoma patients (by CAT-scan criteria).15 Patients underwent PET scanning a median of 1 month (range, 0.07-2.83 months) before HCT; 159 were PET-positive and 177 were PET-negative. At 3 years, relapse/progression, PFS, and OS in PET-positive vs PET-negative groups were 40% vs 26%; P = 0.007; 43% vs 47%; P = 0.47; and 58% vs 60%; P = 0.73, respectively. On multivariate analysis, a positive pretransplantation PET was associated with an increased risk of relapse/progression (risk ratio [RR], 1.86; P = 0.001) but was not associated with increased mortality (P = 0.08), worse PFS (P = 0.10), or NRM (P = 0.22). Based on this analysis, we concluded that while a positive PET scan before HCT is associated with increased relapse risk, it should not be interpreted as a barrier to a successful allograft. PET status does not appear to predict survival after allogeneic HCT for NHL.
10 LOOK AT SOME UNCOMMON MATURE T- OR NK-CELL LYMPHOMAS
In 2013, an important CIBMTR analysis showed that allo-HCT could provide durable disease control in patients with mature T-cell NHL.22 After that publication, the LYWC has prioritized a systematic look at the role of allo-HCT in certain rare T-cell NHL subtypes.
The first rare histology we analyzed was mycosis fungoides and Sezary syndrome (MF/SS) undergoing allo-HCT (n = 129). The majority (64%) received RIC regimens. NRM at 1 and 5 years was 19 and 22%, respectively. Risk of disease progression was 50% at 1 year and 61% at 5 years. OS at 1 and 5 years was 54% and 32%, respectively, suggesting that allo-HCT in MF/SS could be curative for about a third of MF/SS patients.23
Extranodal natural killer (NK)/T-cell lymphoma, nasal type (ENKL), is a rare lymphoma entity characterized by predominantly extranodal involvement and association with Epstein-Barr virus. ENKL has a clear predilection for Asian and South American population, constituting up to 5%-15% of all lymphomas in these regions, compared to only 1% in the U.S. Studies evaluating allo-HCT for ENKL in a North American/European cohort are not available. Using the CIBMTR registry, we reported the largest analysis and the only study to include Caucasian patients, evaluating outcomes of ENKL following allo-HCT.24 Eighty-two adult patients with ENKL undergoing allo-HCT were included after central review of pathology reports to confirm diagnosis. The 3-year rates of relapse, NRM, PFS, and OS were 42%, 30%, 28%, and 34%, respectively. Race (Caucasian vs Asian) did not impact 3-year PFS (26% vs 33%, P = 0.58) or OS (35% vs 33%, P = 0.88). The 3-year relapse rate, PFS and OS by pre-HCT PET scan status, remission status, and ENKL prognostic index were similar (P > 0.1). Conditioning intensity (RIC vs myeloablative conditioning [MAC]) had no impact on 3-year relapse rate (50% vs 30%, P = 0.07), NRM (23% vs 40%, P = 0.12), PFS (27% vs 31%, P = 0.75) or OS (36% vs 34%, P = 0.90). The timing of allo-HCT, upfront (after first line therapy) vs late (>1 line of prior therapy) had no impact on 3-year PFS (P = 0.85) and OS (P = 0.39). This study, the largest and only one to include non-Asian cohort, shows that allo-HCT resulted in durable remission in a subset of ENKL. While no relapses were seen beyond 2 years, primary disease was the most common cause of death underscoring need for novel relapse prevention strategies.
Aggressive NK-cell leukemia (ANKL) is an exceedingly rare form of leukemia and carries a poor prognosis with a median survival of only 2 months. In case series, we evaluated outcomes of allo-HCT in patients with ANKL. Twenty-one patients with a centrally confirmed diagnosis of ANKL were included. Median patient age was 42 years and 15 patients (71%) were Caucasian. Fourteen patients (67%) were in complete remission (CR) at the time of allo-HCT, while five patients had active disease. The 2-year estimates of NRM, relapse/progression, PFS, and OS were 21%, 59%, 20%, and 24%, respectively. The 2-year PFS of patients in CR at the time of allo-HCT was significantly better than that of patients with active disease at transplantation (30% vs 0%; P = 0.001). The 2-year OS in similar order was 38% vs 0% (P < 0.001). This analysis showed that allo-HCT can provide durable disease control in a subset of ANKL patients. Achieving CR before transplantation appeared to be a prerequisite for successful transplantation outcomes.25
11 CONDITIONING REGIMENS FOR ALLO-HCT IN LYMPHOMAS
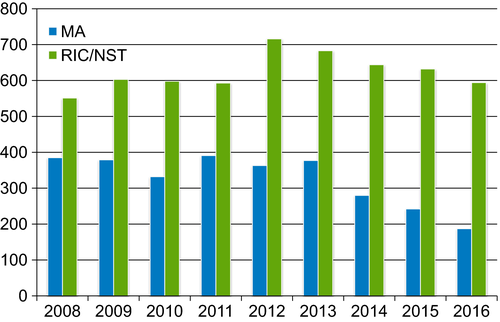
In lymphoma patients undergoing allo-HCT, the choice of intensity and type of conditioning regimen employed is unfortunately not guided by high quality prospective data and remains largely a factor of transplant center preference. Owing to the advanced age of most lymphoma patients and presence of potent graft-vs-lymphoma effects for this disease,26 use of RIC regimens for allo-HCT in the U.S. far outnumbers the usage of MAC regimens (Figure 2). Prior CIBMTR data have shown no clear advantage of MAC over RIC regimens in patients with chemosensitive FL27 or DLBCL.28 Whether MAC offered an advantage in chemorefractory patients was not known. Two large CIBMTR studies compared RIC vs MAC in patients with either refractory aggressive lymphoma (grade 3 FL and DLBCL) or MCL, and showed no significant differences in terms of PFS, relapse rates, and OS between with the two approaches.29, 30 Of note, both studies showed a higher risk of NRM with MAC regimens. In the absence of prospective data proving otherwise, RIC regimens should be considered standard-of-care for allo-HCT in most lymphoma patients.
In B cell NHL, rituximab-containing RIC regimens (R-RIC) have been shown to provide favorable outcomes in single-arm studies. Using the CIBMTR database, we examined the outcomes of R-RIC (n = 379) vs nonR-RIC regimens (n = 1022) in B-NHL.31 Our analysis showed no differences between the R-RIC and nonR-RIC cohorts in terms of GVHD, NRM, relapse/progression or OS. However, R-RIC was associated with a significantly improved PFS (RR = 0.76; 95% CI: 0.62-0.92; P = 0.006). An interesting subgroup analysis showed a survival benefit in R-RIC group patients not receiving busulfan-based RIC. This observation suggested that addition of rituximab might not be warranted in lymphoma patients getting busulfan containing RIC regimens. This provocative observation, prompted us to design a study comparing fludarabine and busulfan (Flu/Bu) vs fludarabine, cyclophosphamide, and rituximab (FCR) RIC conditioning a uniform cohort of FL patients.32 In this analysis, we found no significant differences between the two conditioning approaches, expect a modest (albeit statistically significant) reduction in chronic GVHD risk with FCR conditioning.
12 ALTERNATIVE DONOR TRANSPLANTATION IN LYMPHOMA
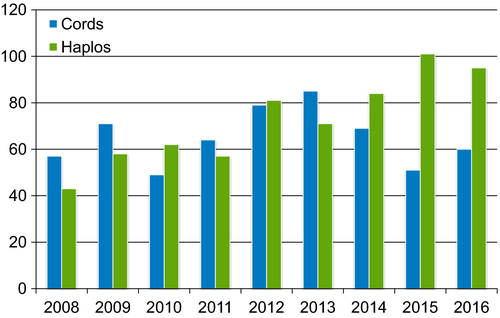
In lymphoma patients being considered for allo-HCT without an available MSD or MUD, alternative donor sources are frequently considered. Current alternative donor options include mismatched unrelated donors (mmURD), umbilical cord blood (UCB) units, or related haploidentical donors (Figure 3). CIBMTR LYWC has been systematically addressing the role of alternative donor transplantation in this setting. In 2015, a CIBMTR study reported comparable survival outcomes in lymphoma patients undergoing MUD, mmURD or UCB allo-HCT (3-year OS of 43% vs 37% vs 41%, respectively), but with significantly higher risk of NRM following mmURD.33 As a next project, we examined the role of haploidentical HCT in lymphoma patients in two studies. In the first report, we evaluated 917 adult lymphoma patients who received haploidentical (n = 185) or MUD transplantation either with (n = 241) or without antithymocyte globulin (ATG; n = 491) following RIC regimens.34 Cumulative incidence of relapse/progression at 3 years was 36%, 28%, and 36% in the haploidentical, MUD without ATG, and MUD with ATG groups, respectively (P = 0.07). Corresponding 3-year OS was 60%, 62%, and 50% in the three groups, respectively. Multivariate analysis showed no difference between the three groups in terms of NRM, relapse, PFS, and OS. A subsequent provocative report compared lymphoma patients undergoing Haploidentical HCT (n = 180) against those receiving MSD-HCT (n = 807) following RIC regimens35 and reported no significant OS and PFS differences between the two groups, but with a possible trend toward higher NRM in the haploidentical cohort. These important studies support the use of alternative donor transplant in lymphoma patients without an available HLA-identical related or URD.
13 ADDRESSING ALLO-HCT ACCESS DISPARITIES FOR MEDICARE PATIENTS:
Unfortunately, allo-HCT is not a covered for NHL patients in the U.S according to current Centers for Medicare and Medicaid services (CMS) guidelines, despite the fact that the median age at diagnosis for NHL in the U.S. is 67 years. This combined with the fact that in the U.S. most patients aged ≥65 years receive their primary insurance coverage through Medicare (and most private insurance providers follow CMS guidelines), means that majority of NHL patients lack access to this potentially curative treatment modality. To address this potential medical care access disparity, we conducted a registry analysis evaluating outcomes of U.S. NHL patients aged ≥65 years undergoing a RIC allo-HCT (n = 446) compared to a younger cohort aged 55-64 years (n = 1183).36 The 4-year adjusted probabilities of NRM, relapse, PFS, and OS of 55-64 years vs ≥65 years groups were 24% vs 30% (P = 0.03), 41% vs 42% (P = 0.82), 37% vs 31% (P = 0.03), and 51% vs 46% (P = 0.07), respectively. On multivariate analysis, compared to the 55-64 years group, the ≥65 years cohort was associated with increased NRM (P = 0.02), but there was no difference between the two cohorts in terms of relapse (P = 0.80), PFS (P = 0.14), or OS (P = 0.10). These data strongly argued that age alone should not be a determinant for allo-HCT eligibility in NHL. We hope that the data from this study will provide the direct evidence needed to justify modification of the current Medicare national coverage determination to allow for allo-HCT in NHL.
14 FUTURE RESEARCH AGENDA
Although rapid strides have been made in lymphoma therapeutics largely due to the better understanding of the molecular pathogenesis, there are still areas that need further investigation and role of HCT ultimately needs to be reassessed. In this regard, role of registries like CIBMTR is going to be even more important. The CIBMTR Lymphoma working committee remains committed to generating clinically relevant data. Our new case report forms were deployed in January 2018 that better capture lymphoma-related genomic information (c-myc, bcl-2, bcl-6 rearrangements etc.), disease-specific prognostic scores, PET scan status using 5-point score, remission status assessment with current criteria and details for posttransplant therapies. Two ongoing studies will evaluate the role of alternative donor transplantation in DLBCL and classical Hodgkin lymphoma. Systematic evaluation of transplant conditioning regimens is also high on priority. One study will evaluate common RIC platforms in NHL and Hodgkin lymphoma, while a study approved in 2018 will assess the role of rituximab in auto-HCT conditioning. In the next 1-2 years, the role of transplant in angioimmunoblastic T-cell lymphoma and chronic lymphocytic leukemia transforming to DLBCL will be evaluated. The registry has also started capturing data on cellular immunotherapies (eg, CAR-T cells) that will enhance our ability to answer many novel questions. Ongoing collaboration between transplant centers across the globe and CIBMTR will hopefully continue to impact patient care decision via high quality research.
ACKNOWLEDGMENTS
Author wishes to acknowledge support of: CIBMTR Reporting Centers and Lymphoma Working Committee members. CIBMTR Milwaukee and Minneapolis Campus Staff. CIBMTR Lymphoma Working Committee MS Statisticians (2013-18): Jeannette Carreras, Alyssa Digilio and Carlos Litovich. CIBMTR Lymphoma Working Committee PhD Statistician (2013-18): Kwang W. Ahn. CIBMTR Lymphoma Working Committee Chairs (2013-18): David Maloney, Sonali Smith, Timothy Fenske, Anna Sureda and Mohammed Kharfan-Dabaja. Prior CIBMTR Lymphoma Working Committee Scientific Directors: Wael Saber and Parameswaran Hari.
ETHICAL STATEMENT
The author confirms that the ethical policies of the journal, as noted on the journal's author guidelines page, have been adhered to. No ethical approval was required as this is a commentary with no original research data.