Epidermal Growth Factor Receptor Mutations and Their Prognostic Value with Carcinoembryonic Antigen in Pathological T1 Lung Adenocarcinoma
Abstract
Aims. The prognostic value of epidermal growth factor receptor (EGFR) mutations in the context of serum carcinoembryonic antigen levels remains controversial in T1 lung adenocarcinoma. Methods. Clinical and pathological characteristics, preoperational carcinoembryonic antigen levels, EGFR mutations, and disease-free and overall survival were analysed retrospectively in 573 pathological T1 patients in East China. Results. EGFR mutations were detected in 220 of 573 patients (38.4%). Patients with serum carcinoembryonic antigen levels ≥ 2.12 ng/mL had worse disease-free (P < 0.001) and overall survival (P < 0.001) than had others, although survival was comparable between patients with and without EGFR mutations. However, patients with exon 21 mutations in EGFR had significantly better overall survival than had patients with exon 19 mutations (P = 0.016), although disease-free survival was comparable (P = 0.424). Among patients with serum carcinoembryonic antigen levels ≥ 2.12 ng/mL, disease-free (P = 0.019) and overall survival (P < 0.001) was also better than that in those with exon 21 mutations. Finally, the exon 19 deletion was found to be an independent predictor of unfavourable overall survival (P = 0.037). Conclusions. EGFR mutations were associated with preoperational serum carcinoembryonic antigen levels ≥ 2.12 ng/mL. In patients with levels above this threshold, those with the exon 19 deletion have less favourable prognosis than have those with the exon 21 mutation.
1. Introduction
Lung adenocarcinoma is the most prevalent subtype of non-small cell lung cancer, and the 5-year overall survival remains poor [1–3]. Lung adenocarcinoma may arise from an accumulation of genetic mutations, of which those in epidermal growth factor receptor (EGFR) are some of the most important and are associated with tumour progression, proliferation, and survival [4]. EGFR mutations are the most common genetic lesions in adenocarcinoma but are very rare in squamous cell carcinoma [5]. However, the median progression-free survival in squamous cell carcinoma patients treated with EGFR tyrosine kinase inhibitors is worse than that in adenocarcinoma patients with EGFR mutations [6]. Thus, EGFR mutations may be predictive of the therapeutic response to such inhibitors [7]. Similarly, non-small cell lung cancer patients with mutated EGFR also have higher median disease-free survival and improved overall survival [8–10]. However, the predictive value of EGFR mutations in patients with pathological T1 lung adenocarcinoma is still unclear. Strikingly, deletion of EGFR exon 19 and a point mutation in exon 21 account for up to 90% of EGFR mutations in the clinic and correspond to two distinct tumour subtypes with different clinical characteristics and response to EGFR tyrosine kinase inhibitors [11, 12]. Hence, the prognostic role of EGFR mutations in T1 lung adenocarcinoma is probably well-defined.
In contrast, carcinoembryonic antigen has been used as a biomarker of prognosis and therapeutic efficacy in non-small cell lung cancer. Notably, Cai [13] reported that carcinoembryonic antigen levels gradually increase with the rate of EGFR mutations. Moreover, carcinoembryonic antigen levels were reported to be independently prognostic in lung adenocarcinoma patients without EGFR mutations [14]. Indeed, we also found that lung adenocarcinoma patients with carcinoembryonic antigen levels above 2.12 ng/mL have a poor prognosis [15]. Nevertheless, other surveys indicated that carcinoembryonic antigen levels are normal in most patients with early-stage lung cancer. Hence, the purpose of this study was to investigate EGFR mutations in the context of carcinoembryonic antigen levels and to assess the prognostic value of such mutations in patients with pathological T1 lung adenocarcinoma.
2. Methods
2.1. Patients
Patients who underwent surgical resection for pathological T1 adenocarcinoma of the lung (N = 573) were enrolled retrospectively at Zhoushan Hospital, Zhejiang, China, from July 2011 through March 2016. Histological subtypes were assigned by two pathologists, in accordance with World Health Organization classification and new criteria from the International Association for the Study of Lung Cancer, American Thoracic Society, and European Respiratory Society [16]. The staging of all patients with lung cancer was redefined according to the proposed 8th edition of lung cancer classification [17–19]. The maximum diameter of the resected lesion was measured by a pathologist. Patients were also genotyped for EGFR mutations. Clinicopathological features including age, sex, comorbidities, smoking history, lymphatic vessel invasion, vascular vessel invasion, pleural invasion, tumour maximum diameter, tumour stage, tumour histologic subtype, and preoperative serum carcinoembryonic antigen levels were analysed systematically. Patients with resected tumours greater than 3 cm in maximum diameter were excluded. Patients with incomplete records and follow-up data were also excluded, along with patients who died within 30 days after surgery. Patients were monitored over time, using computed tomography (CT) to assess recurrence. Overall survival was calculated as the period from surgical resection to end of follow-up, which was considered to be the time of death, or at the final follow-up of surviving patients. Disease-free survival was calculated as the period between surgery and initial detection of recurrence and metastasis. The study was approved by the Ethical Review Committee of the Zhoushan Municipal Government, and written informed consent was obtained from subjects or their families.
2.2. Genomic DNA Extraction and EGFR Genotyping
Resected tumours were fixed with 10% formalin, embedded in paraffin, and sectioned at 10 μm. Genomic DNA was extracted from five sections using a QIAamp DNA FFPE Tissue Kit (QIAGEN, Hilden, Germany). DNA concentration and purity were assessed on a Quawell Q3000 spectrophotometer (Quawell Technology, Sunnyvale, CA, USA). EGFR was genotyped on a 7500 Real-Time PCR System (ABI, Foster City, CA, USA) using an amplification refractory mutation system (Yuanqi Diagnostics, Shanghai, China), following the manufacturer’s instructions.
2.3. Statistical Analysis
Data were analysed in GraphPad Prism 5.0 (GraphPad Software Inc., San Diego, CA) and SPSS 17.0 (SPSS Institute, Chicago, IL, USA). Pearson’s chi-squared test or Fisher’s exact test (t < 1 or n < 40) was performed to compare differences between categorical groups. The Gaussian distribution was examined according to the Kolmogorov-Smirnov test. Then, the Mann–Whitney U test [data shown as the median (P25, P75)] or unpaired t-test (data shown as the mean ± SD) was used to analyse the difference in CEA level between EGFR mutated and wild-type patients. Disease-free and overall survival was evaluated by Kaplan-Meier curves. Multivariate analysis was performed using Cox’s proportional hazards regression model for all prognostic factors with univariate P < 0.05. All statistical tests were two-sided, with P < 0.05 considered significant.
3. Results
3.1. Relationship between EGFR Mutations and Clinicopathological Features
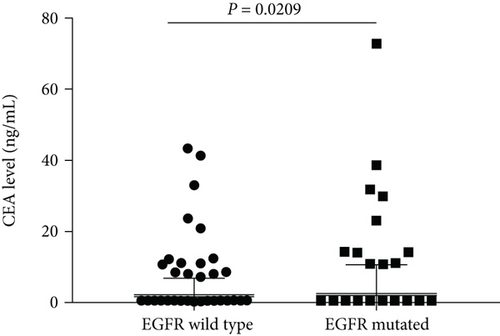
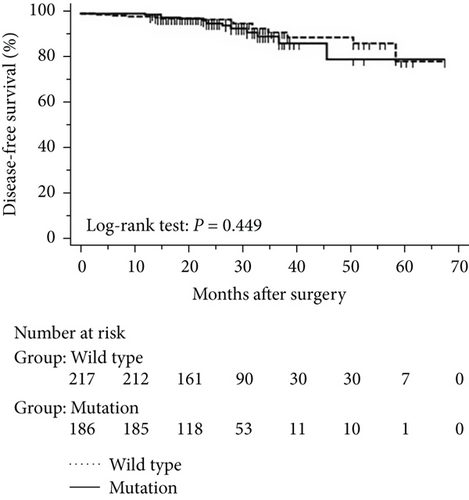
Clinical and pathological characteristics are summarized in Table 1 for 573 patients in eastern Chinese islands who had lung adenocarcinoma with a maximum diameter of 3.0 cm or less. Of these patients, 220 were found postsurgery to harbour EGFR mutations (38.4%), consisting of G719X in exon 18 (n = 6, 1.0%), exon 19 deletion (n = 64, 11.2%), exon 20 insertion (n = 1, 0.2%), L858R and/or L861Q in exon 21 (n = 145, 25.3%), and combined mutations in exons 18 and 19 (n = 1, 0.2%), exons 20 and 21 (n = 1, 0.2%), and exons 19 and 21 (n = 2, 0.3%). The CEA level was higher in EGFR mutated patients than in the wild-type patients [1.83 (1.22, 2.91) versus 1.61 (1.05, 2.54), P = 0.0209, Figure 1]. Mutations were more likely to occur in patients with carcinoembryonic antigen ≥ 2.12 ng/mL (P = 0.030), a threshold identified in our previous survey [15]. Mutations were also associated with tumour size pT1b and pT1c (P < 0.001), pleural invasion (P = 0.016), histology (P < 0.001), lymphatic metastasis (P = 0.026), and stage (P < 0.001). All other clinical features were comparable between patients with and without EGFR mutations (Table 1).
| Characteristics | All (n = 573) | EGFR mutated (n = 220) | EGFR wild type (n = 353) | P |
|---|---|---|---|---|
| Age, mean y (range) | 57.0 ± 10.0 (26–84) | 60.5 ± 10.0 (28–84) | 55.0 ± 10.5 (26–77) | <0.001 ∗a |
| Sex | ||||
| Male | 175 (30.5) | 69 (39.4) | 106 (60.6) | 0.780b |
| Female | 398 (69.5) | 151 (37.9) | 247 (62.1) | |
| Smoking | ||||
| Nonsmoker | 468 (81.7) | 184 (39.3) | 284 (60.7) | 0.375b |
| Current/former | 105 (18.3) | 36 (34.3) | 69 (65.7) | |
| CEA | ||||
| <2.12 IU/mL | 378 (66.0) | 133 (35.2) | 245 (64.8) | 0.030 ∗b |
| ≥2.12 IU/mL | 195 (34.0) | 87 (44.6) | 108 (55.4) | |
| Type of surgery | ||||
| Lobectomy | 392 (68.4) | 156 (39.8) | 236 (60.2) | 0.356b |
| Limited | 181 (31.6) | 64 (35.4) | 117 (64.6) | |
| Tumour size | ||||
| pT1a | 305 (53.2) | 76 (24.9) | 229 (75.1) | <0.001 ∗b |
| pT1b | 197 (34.4) | 103 (52.3) | 94 (47.7) | |
| pT1c | 71 (12.4) | 41 (57.7) | 30 (42.3) | |
| Histology | ||||
| AIS | 170 (29.7) | 34 (20.0) | 136 (80.0) | <0.001 ∗b |
| MIA | 167 (29.1) | 57 (34.1) | 110 (65.9) | |
| IAC | 236 (41.2) | 129 (54.7) | 107 (45.3) | |
| IAC subtype (n = 236) | ||||
| Lepidic | 53 (22.5) | 30 (56.6) | 23 (43.4) | 0.041 ∗c |
| Acinar | 65 (27.5) | 40 (62.5) | 25 (37.5) | |
| Papillary | 105 (44.5) | 57 (54.3) | 48 (45.7) | |
| Solid | 9 (3.8) | 2 (22.2) | 7 (77.8) | |
| Mucinous variant | 4 (1.7) | 0 | 4 (100.0) | |
| Pleural invasion | ||||
| No | 528 (92.1) | 195 (36.9) | 333 (63.1) | 0.016 ∗b |
| Yes | 45 (7.9) | 25 (55.6) | 20 (44.4) | |
| Lymph nodes | ||||
| pN0 | 546 (95.3) | 204 (37.4) | 342 (62.6) | 0.026 ∗b |
| pN1 or 2 | 27 (4.7) | 16 (59.3) | 11 (40.7) | |
| Pathological stage | ||||
| 0 | 170 (29.7) | 34 (20.0) | 136 (80.0) | <0.001 ∗c |
| IA | 339 (59.2) | 149 (44.0) | 190 (56.0) | |
| IB | 37 (6.4) | 21 (56.8) | 16 (43.2) | |
| IIA, IIB, IIIA | 27 (4.7) | 16 (59.3) | 11 (40.7) | |
- CEA: carcinoembryonic antigen; AIS: adenocarcinoma in situ; MIA: minimally invasive adenocarcinoma; IAC: invasive adenocarcinoma. ∗P < 0.05 by at-test, bPearson’s chi-squared test, and cFisher’s exact test.
Among patients with carcinoembryonic antigen levels < 2.12 ng/mL (Table 2), EGFR mutations were associated with age (P < 0.001), tumour size (P < 0.001), histology (P < 0.001), and stage (P < 0.001). In contrast, EGFR mutations in patients with carcinoembryonic antigen levels ≥ 2.12 ng/mL were associated with age (P = 0.002), nonsmokers (P = 0.038), tumour size (P = 0.010), histology (P < 0.001), and stage (P < 0.001, Table 2).
| Characteristics | CEA < 2.12 IU/mL (n = 378) | P | CEA ≥ 2.12 IU/mL (n = 195) | P | ||
|---|---|---|---|---|---|---|
| EGFR mutated | EGFR wild type | EGFR mutated | EGFR wild type | |||
| Age, mean y (range) | 58.4 ± 10.2 (26–83) | 52.9 ± 10.5 (26–83) | <0.001 ∗a | 63.7 ± 8.3 (38–84) | 59.8 ± 8.6 (38–84) | 0.002 ∗a |
| Sex | ||||||
| Male | 37 (16.3) | 190 (83.7) | 0.260b | 55 (49.1) | 57 (50.9) | 0.143b |
| Female | 96 (63.6) | 55 (36.4) | 32 (38.6) | 51 (61.4) | ||
| Smoking | ||||||
| Nonsmoker | 115 (35.0) | 214 (65.0) | 0.873b | 69 (49.6) | 70 (50.4) | 0.038b |
| Current/former | 18 (36.7) | 31 (63.3) | 18 (32.1) | 38 (67.9) | ||
| Type of surgery | ||||||
| Lobectomy | 89 (36.3) | 156 (63.7) | 0.574b | 67 (45.6) | 80 (54.4) | 0.738b |
| Limited | 44 (33.1) | 89 (66.9) | 20 (41.7) | 28 (58.3) | ||
| Tumour size | ||||||
| pT1a | 58 (23.9) | 185 (76.1) | <0.001 ∗b | 18 (29.0) | 44 (80.0) | 0.010 ∗b |
| pT1b | 60 (53.6) | 52 (46.4) | 43 (50.6) | 42 (49.4) | ||
| pT1c | 15 (65.2) | 8 (34.8) | 26 (54.2) | 22 (45.8) | ||
| Histology | ||||||
| AIS | 29 (21.5) | 106 (78.5) | <0.001 ∗b | 5 (14.3) | 30 (85.7) | <0.001 ∗b |
| MIA | 35 (28.2) | 89 (71.8) | 22 (51.2) | 21 (48.8) | ||
| IAC | 69 (58.0) | 50 (42.0) | 60 (51.3) | 57 (48.7) | ||
| IAC subtype (n = 236) | ||||||
| Lepidic | 20 (60.6) | 13 (39.4) | 0.344c | 10 (50.0) | 10 (50.0) | 0.123c |
| Acinar | 19 (41.3) | 27 (58.7) | 21 (56.8) | 16 (43.2) | ||
| Papillary | 30 (76.9) | 9 (23.1) | 27 (56.3) | 21 (43.7) | ||
| Solid | 2 (22.2) | 7 (77.8) | ||||
| Mucinous variant | 0 | 1 (100.0) | 0 | 3 (100.0) | ||
| Pleural invasion | ||||||
| No | 124 (34.3) | 238 (65.7) | 0.105b | 71 (42.8) | 95 (57.2) | 0.231b |
| Yes | 9 (56.3) | 7 (43.7) | 16 (55.2) | 13 (44.8) | ||
| Lymph nodes | ||||||
| pN0 | 130 (34.8) | 244 (65.2) | 0.127c | 74 (43.0) | 98 (57.0) | 0.267b |
| pN1 or 2 | 3 (75.0) | 1 (25.0) | 13 (56.5) | 10 (43.5) | ||
| Pathological stage | ||||||
| 0 | 29 (21.5) | 106 (78.5) | <0.001 ∗c | 5 (14.3) | 30 (85.7) | 0.001 ∗c |
| IA | 93 (41.5) | 131 (58.5) | 56 (48.7) | 59 (51.3) | ||
| IB | 8 (53.3) | 7 (46.7) | 13 (59.1) | 9 (40.9) | ||
| IIA, IIB, IIIA | 3 (75.0) | 1 (25.0) | 13 (56.5) | 10 (43.5) | ||
- ∗P < 0.05 by at-test, bPearson’s chi-squared test, and cFisher’s exact test.
The clinical features of patients with an exon 19 deletion or an exon 21 point mutation are listed in Table 3. Exon 21 point mutations were more common in patients with the lepidic predominant invasive adenocarcinoma (IAC) subtype (P = 0.022). However, the frequency of mutations was comparable among other IAC subtypes and was not associated with other clinical and pathological features (Table 3).
| Characteristics | Exon 19 (n = 64) | Exon 21 (n = 145) | P |
|---|---|---|---|
| Age, mean y (range) | 59.7 ± 10.7 (30–80) | 61.2 ± 9.4 (28–84) | 0.324a |
| Sex | |||
| Male | 24 (35.8) | 43 (64.2) | 0.263b |
| Female | 40 (28.2) | 102 (71.8) | |
| Smoking | |||
| Nonsmoker | 50 (28.6) | 125 (71.4) | 0.158b |
| Current/former | 14 (41.2) | 20 (58.8) | |
| CEA | |||
| <2.12 IU/mL | 42 (33.9) | 82 (66.1) | 0.218b |
| ≥2.12 IU/mL | 22 (25.9) | 63 (74.1) | |
| Type of surgery | |||
| Lobectomy | 48 (31.6) | 104 (68.4) | 0.737b |
| Limited | 16 (28.1) | 41 (71.9) | |
| Tumour size | |||
| pT1a | 16 (24.2) | 50 (75.8) | 0.231b |
| pT1b | 32 (31.1) | 71 (68.9) | |
| pT1c | 16 (40.0) | 24 (60.0) | |
| Histology | |||
| AIS | 6 (20.0) | 24 (80.0) | 0.267b |
| MIA | 15 (27.8) | 39 (72.2) | |
| IAC | 43 (34.4) | 82 (65.6) | |
| IAC subtype (n = 125) | |||
| Lepidic | 3 (10.7) | 25 (89.3) | 0.022 ∗c |
| Acinar | 16 (40.0) | 24 (60.0) | |
| Papillary | 23 (41.8) | 32 (58.2) | |
| Solid | 1 (50.0) | 1 (50.0) | |
| Pleural invasion | |||
| No | 54 (29.3) | 130 (70.7) | 0.278b |
| Yes | 10 (40.0) | 15 (60.0) | |
| Lymph nodes | |||
| pN0 | 59 (30.6) | 134 (69.4) | 0.955b |
| pN1 or 2 | 5 (31.3) | 11 (68.7) | |
| Pathological stage | |||
| 0 | 6 (20.7) | 24 (79.3) | 0.216b |
| IA | 43 (30.1) | 99 (69.9) | |
| IB | 10 (47.6) | 11 (52.4) | |
| IIA, IIB, IIIA | 5 (31.3) | 11 (68.7) | |
- CEA: carcinoembryonic antigen; AIS: adenocarcinoma in situ; MIA: minimally invasive adenocarcinoma; IAC: invasive adenocarcinoma. ∗P < 0.05 by at-test, bPearson’s chi-squared test, and cFisher’s exact test.
3.2. Disease-Free and Overall Survival for Pathological T1 Lung Adenocarcinoma
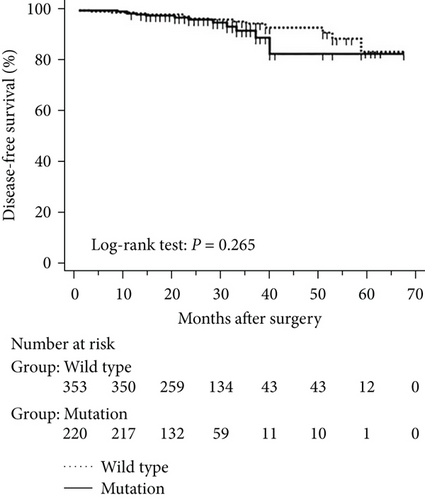
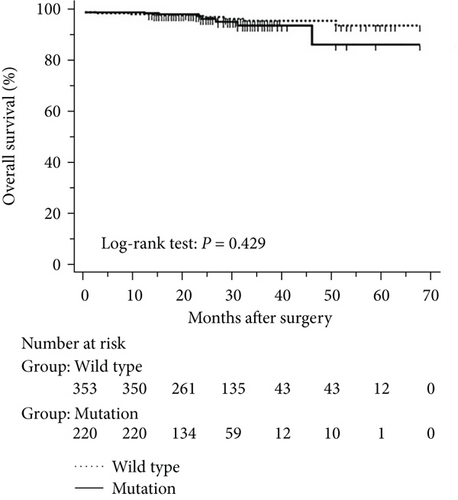
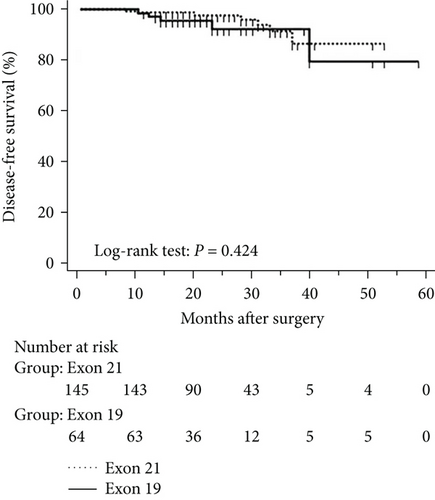
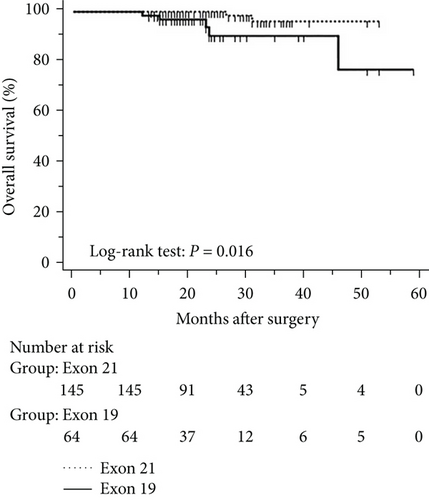
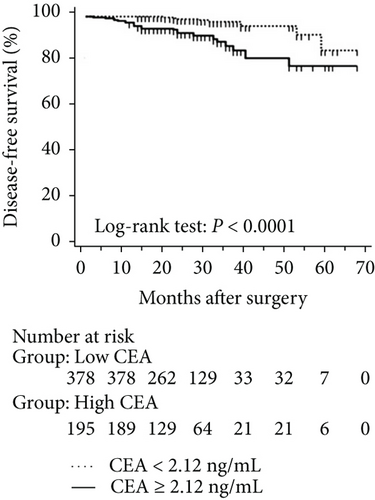
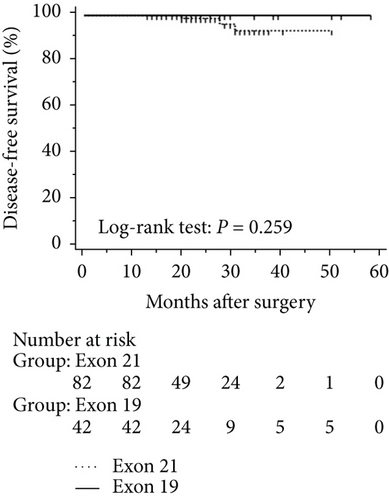
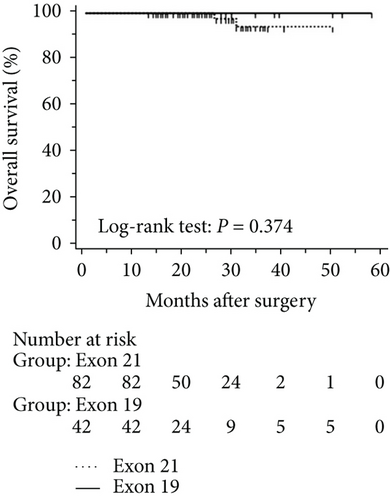
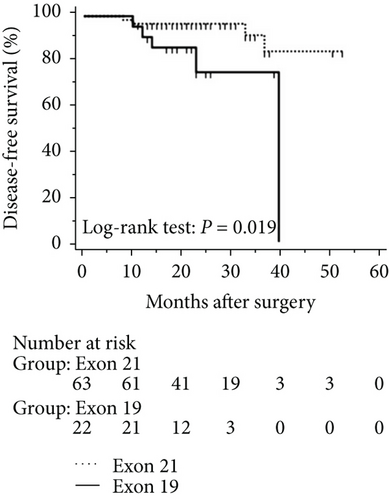
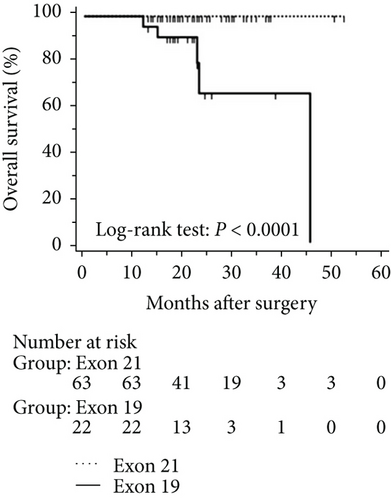
The mean follow-up time was 27.7 months, with a median of 25 months and range of 2.5–68 months. Disease-free and overall survival was comparable between patients with and without EGFR mutations. However, patients with exon 21 mutations had significantly better overall survival than had patients with exon 19 mutations (P = 0.016), although disease-free survival was comparable (P = 0.424, Figure 2). In addition, patients with serum carcinoembryonic antigen levels ≥ 2.12 IU/mL exhibited worse disease-free (P < 0.0001) and overall survival (P < 0.0001) than did others (Figures 3(a) and 3(b)). Among patients with carcinoembryonic antigen levels below the threshold, disease-free (P = 0.259) and overall survival (P = 0.374) was comparable between those with exon 21 mutations and exon 19 deletions (Figures 3(c) and 3(d)). Among patients with carcinoembryonic antigen levels above the threshold, disease-free (P = 0.019) and overall survival (P < 0.0001) was better in those with exon 21 mutations than in those with exon 19 deletions (Figures 3(e) and 3(f)).

3.3. Univariate and Multivariate Analyses for Pathological T1 Lung Adenocarcinoma
Univariate and multivariate Cox regression analysis results for disease-free and overall survival are summarized in Table 4. In the multivariate analysis, unfavourable disease-free survival was associated with preoperational carcinoembryonic antigen levels above 2.12 ng/mL (P = 0.022), IAC pathology (P = 0.046), confirmed lymphatic metastasis (P < 0.001), and advanced pathological stage (P = 0.033). The exon 19 deletion was an independent predictor of reduced overall survival (P = 0.037).
| Factor | Univariate analysis | Multivariate analysis | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | P | HR | 95% CI | P | |
| Disease-free survival | ||||||
| Sex (female versus male) | 2.303 | 1.111–4.775 | 0.025 ∗ | 1.268 | 0.583–2.758 | 0.550 |
| CEA (<2.12 versus ≥2.12) | 4.999 | 2.211–11.302 | <0.001 ∗ | 2.877 | 1.166–7.097 | 0.022 ∗ |
| Tumour size (pT1a versus pT1b versus pT1c) | 2.709 | 1.664–4.411 | <0.001 ∗ | 1.454 | 0.786–2.687 | 0.233 |
| Histology (AIS and MIA versus IAC) | 6.300 | 2.400–16.540 | <0.001 ∗ | 3.204 | 1.023–10.040 | 0.046 ∗ |
| Lymphatic metastasis (absent versus present) | 8.901 | 3.934–20.139 | <0.001 ∗ | 11.201 | 2.658–47.209 | 0.001 ∗ |
| Pathological stage (stage 0 versus I, II, and IIIA) | 2.128 | 1.430–3.168 | <0.001 ∗ | 0.361 | 0.163–0.800 | 0.012 ∗ |
| Overall survival | ||||||
| Sex (female versus male) | 3.472 | 1.320–9.130 | 0.012 ∗ | 2.070 | 0.308–13.899 | 0.454 |
| Smoking (non versus current/former) | 3.235 | 1.231–8.506 | 0.017 ∗ | 1.276 | 0.129–12.663 | 0.835 |
| CEA (<2.12 versus ≥2.12) | 9.200 | 2.641–32.049 | 0.001 ∗ | 2.626 | 0.352–19.583 | 0.346 |
| EGFR mutation (exon 21 versus exon 19) | 6.170 | 1.162–32.768 | 0.033 ∗ | 7.153 | 1.124–45.516 | 0.037 ∗ |
| Tumour size (pT1a versus pT1b versus pT1c) | 4.056 | 2.046–8.039 | <0.001 ∗ | 5.793 | 0.934–35.944 | 0.059 |
| Histology (AIS and MIA versus IAC) | 21.184 | 2.187–124.759 | 0.003 ∗ | 36416.346 | 0.000–3.671E220 | 0.967 |
| Lymphatic metastasis (absent versus present) | 9.309 | 3.275–26.457 | <0.001 ∗ | 14.820 | 0.172–1276.425 | 0.236 |
| Pathological stage (stage 0 versus I, II, and IIIA) | 2.201 | 1.319–3.673 | 0.003 ∗ | 0.448 | 0.045–4.468 | 0.494 |
- HR: hazard ratio; CI: confidence interval; CEA: carcinoembryonic antigen; AIS: adenocarcinoma in situ; MIA: minimally invasive adenocarcinoma; IAC: invasive adenocarcinoma. ∗P < 0.05.
Among patients with carcinoembryonic antigen levels above the threshold (Table 5), exon 19 deletion (P = 0.031), large tumour size (P = 0.001), IAC pathology (P = 0.012), confirmed lymphatic metastasis (P = 0.001), and advanced pathological stage (P = 0.029) were found by univariate analysis to be unfavourable for disease-free survival. In contrast, large tumour size (P = 0.009) and lymphatic metastasis (P = 0.003) were predictive of worse overall survival. In the multivariate analysis, none of the clinical features were significantly associated with disease-free survival, although lymphatic metastasis was an independent predictor of reduced overall survival (P = 0.039).
| Factor | Univariate analysis | Multivariate analysis | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | P | HR | 95% CI | P | |
| Disease-free survival | ||||||
| EGFR mutation (Ex21 versus Ex19) | 4.281 | 1.138–16.105 | 0.031 ∗ | 2.705 | 0.657–11.136 | 0.168 |
| Tumour size (pT1a versus pT1b versus pT1c) | 2.910 | 1.506–5.623 | 0.001 ∗ | 2.813 | 0.551–14.358 | 0.214 |
| Histology (AIS and MIA versus IAC) | 13.031 | 1.748–97.143 | 0.012 ∗ | 76752.907 | 0.000–6.609E256 | 0.970 |
| Lymphatic metastasis (absent versus present) | 4.811 | 1.926–12.018 | 0.001 ∗ | 9.748 | 0.262–362.601 | 0.217 |
| Pathological stage (stage 0 versus I, II, and IIIA) | 1.664 | 1.054–2.627 | 0.029 ∗ | 0.772 | 0.099–6.005 | 0.804 |
| Overall survival | ||||||
| Tumour size (pT1a versus pT1b versus pT1c) | 2.966 | 1.318–6.674 | 0.009 ∗ | 3.204 | 0.989–10.382 | 0.052 |
| Lymphatic metastasis (absent versus present) | 5.216 | 1.725–15.7737 | 0.003 ∗ | 2.444 | 1.044–5.720 | 0.039 ∗ |
- HR: hazard ratio; CI: confidence interval; AIS: adenocarcinoma in situ; MIA: minimally invasive adenocarcinoma; IAC: invasive adenocarcinoma. ∗P < 0.05.
3.4. Disease-Free and Overall Survival Excluding Stage 0 Patients
The progression of lung cancer is typically divided into five stages (0–IV); however, the stage 0 lung cancer patients had better prognosis with 100% disease-free survival and overall survival, and we thus excluded stage 0 patients to analyse the data again [17–19]. The mean follow-up time was 28.0 months, with a median of 25.5 months and range of 2.5–68 months. As shown in Figures 4 and 5, the results were similar to those for the whole cohort, and patients with exon 21 mutations had significantly extended overall survival than had patients with exon 19 mutations (P = 0.019, Figure 4). Additionally, patients with a higher level of serum carcinoembryonic antigen levels showed worse disease-free (P < 0.001) and overall survival (P < 0.001) than did others, and those with exon 21 mutations had better disease-free (P = 0.025) and overall survival (P < 0.001) than had those with exon 19 deletions (Figure 5).

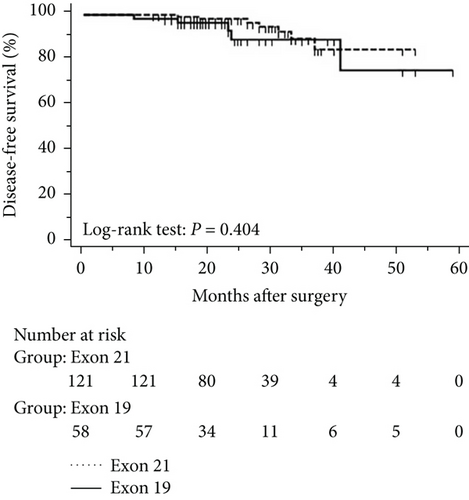
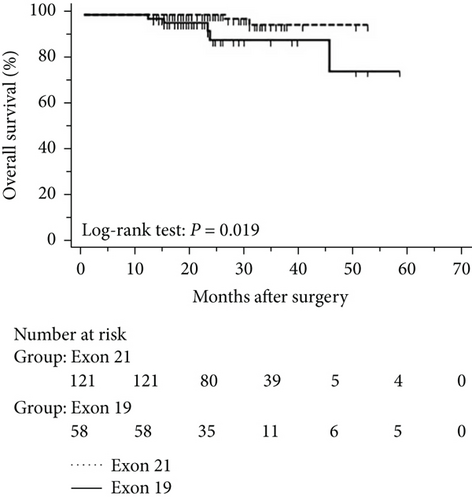
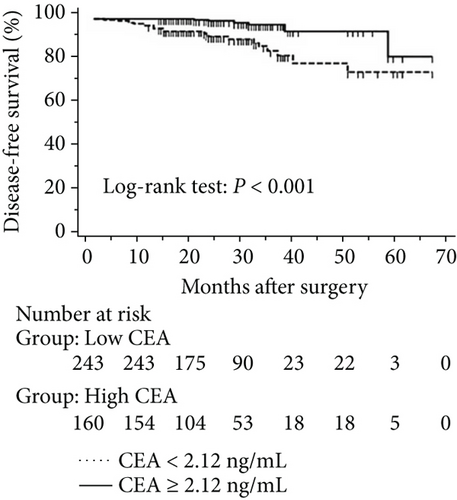
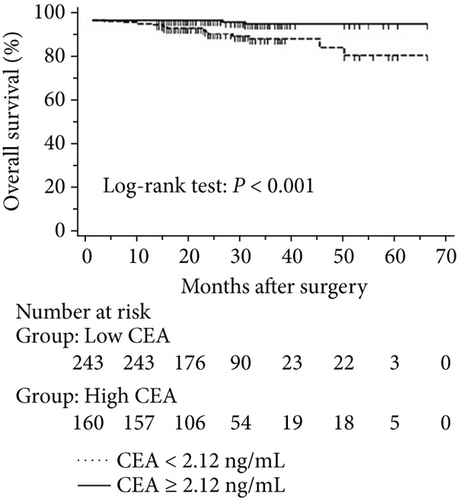
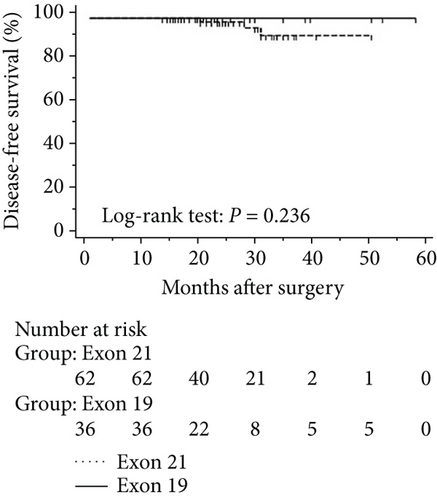
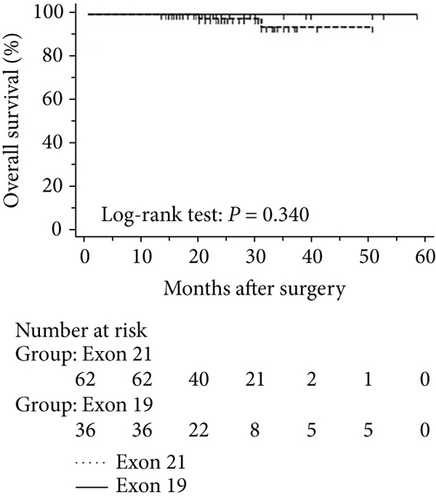
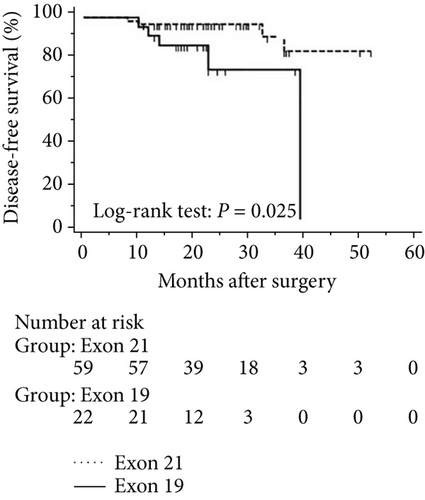
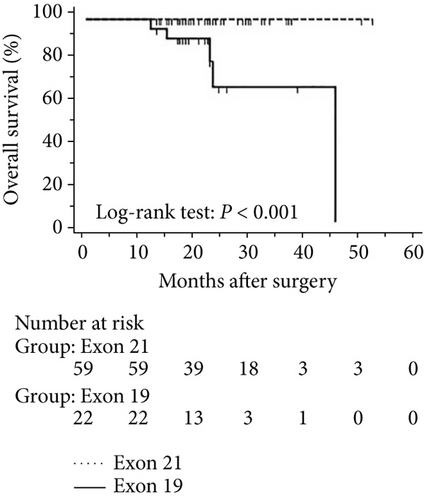
3.5. Univariate and Multivariate Analyses for Pathological T1 Lung Adenocarcinoma
Univariate and multivariate Cox regression analysis results for disease-free and overall survival of patients excluding those in stage 0 are summarized in Table 6. In the multivariate analysis, unfavourable disease-free survival was correlated with preoperational carcinoembryonic antigen levels above 2.12 ng/mL (P = 0.031), confirmed lymphatic metastasis (P < 0.001), and advanced pathological stage (P = 0.012). The exon 19 deletion was an independent predictor of reduced overall survival (P = 0.036).
| Factor | Univariate analysis | Multivariate analysis | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | P | HR | 95% CI | P | |
| Disease-free survival | ||||||
| Sex (female versus male) | 2.320 | 1.072–5.023 | 0.033 ∗ | 1.377 | 0.601–3.157 | 0.450 |
| CEA (<2.12 versus ≥2.12) | 5.203 | 2.086–12.977 | <0.001 ∗ | 3.016 | 1.104–8.241 | 0.031 ∗ |
| Tumour size (pT1a versus pT1b versus pT1c) | 2.235 | 1.464–3.412 | <0.001 ∗ | 1.580 | 0.800–3.119 | 0.188 |
| Histology (MIA versus IAC) | 7.971 | 1.881–33.772 | 0.005 ∗ | 1.913 | 0.224–16.317 | 0.553 |
| Lymphatic metastasis (absent versus present) | 7.183 | 3.117–16.553 | <0.001 ∗ | 19.612 | 3.888–98.923 | <0.001 ∗ |
| Pathological stage (stage I versus II, and IIIA) | 2.054 | 1.263–3.340 | 0.004 ∗ | 0.342 | 0.147–0.793 | 0.012 ∗ |
| Overall survival | ||||||
| Sex (female versus male) | 3.332 | 1.208–9.188 | 0.020 ∗ | 2.090 | 0.312–14.004 | 0.447 |
| Smoking (non versus current/former) | 2.817 | 1.047–7.578 | 0.040 ∗ | 1.263 | 0.127–12.561 | 0.842 |
| CEA (<2.12 versus ≥2.12) | 11.244 | 2.552–49.537 | 0.001 ∗ | 2.643 | 0.352–19.842 | 0.345 |
| EGFR mutation (exon 21 versus exon 19) | 5.974 | 1.124–31.746 | 0.036 ∗ | 7.221 | 1.133–46.003 | 0.036 ∗ |
| Tumour size (pT1a versus pT1b versus pT1c) | 3.351 | 1.698–6.613 | <0.001 ∗ | 6.019 | 1.036–34.958 | 0.046 ∗ |
| Lymphatic metastasis (absent versus present) | 7.147 | 2.479–20.606 | <0.001 ∗ | 14.645 | 0.170–1258.671 | 0.238 |
| Pathological stage (stage I versus II, and IIIA) | 1.898 | 1.011–3.564 | 0.046 ∗ | 0.449 | 0.045–4.503 | 0.496 |
- HR: hazard ratio; CI: confidence interval; CEA: carcinoembryonic antigen; AIS: adenocarcinoma in situ; MIA: minimally invasive adenocarcinoma; IAC: invasive adenocarcinoma. ∗P < 0.05.
4. Discussion
We surveyed the prognostic value of EGFR mutations in a cohort of 573 patients from East China who underwent surgical resection of pathological T1 lung carcinoma. In particular, we analysed EGFR mutations in patients with preoperational carcinoembryonic antigen levels ≥ 2.12 ng/mL, a key prognostic threshold identified in our previous study [15]. The data suggest that although disease-free and overall survival was comparable between patients with or without EGFR mutations, patients with exon 21 mutations had extended overall survival in comparison with patients with exon 19 deletion. Extended disease-free and overall survival was also observed in patients with exon 21 mutations who had carcinoembryonic antigen levels above the threshold, but not for patients with the same mutations who had carcinoembryonic antigen levels below the threshold. Accordingly, exon 19 deletion was an independent predictor of reduced overall survival.
EGFR mutations have been reported to be more frequent in Chinese and other Asian populations than in Western populations. Indeed, we detected EGFR mutations in 38.4% of our cohort, which was in line with previous surveys of stage I lung adenocarcinoma [20], but slightly lower than the rate in another report [21]. We attribute this difference to the inclusion in our cohort of a number of patients who had tumours of the adenocarcinoma in situ (AIS) subtype (29.7%), in which the frequency of EGFR mutations was reported to be 27.3% or 23.8% [22, 23]. In contrast, EGFR mutations in our cohort were most prevalent in patients with acinar forms (62.5%), and then in patients with lepidic (56.6%), papillary (54.3%), minimally invasive adenocarcinoma (MIA) (34.1%), solid (22.2%), and AIS (20.0%) forms, in line with other studies [23, 24]. In addition, clinical characteristics were comparable between patients with exon 19 and exon 21 mutations, except that lepidic subtypes were more common in the latter than in the former. Conversely, Japanese and Chinese surveys demonstrated that exon 21 mutations were more common than exon 19 mutations in lepidic tumours [11, 25].
On the basis of our previous study, we stratified the patients by serum carcinoembryonic antigen levels [15]. The new data indicate that serum carcinoembryonic antigen levels ≥ 2.12 ng/mL are associated with EGFR mutations, as previously observed in lung cancer [13, 26, 27]. Strikingly, these mutations were more frequent in nonsmokers with serum carcinoembryonic antigen levels ≥ 2.12 ng/mL, even though the prevalence was comparable between smokers and nonsmokers in the entire study population. Notably, we observed that the prevalence was lower at stage 0 than at stages I-III, in contrast to previous surveys [13, 24]. Moreover, we report for the first time that EGFR mutations are less frequent in patients with tumour size pT1a than in patients with tumour size pT1b and pT1c. We note, however, that our cohort included a greater proportion of lung adenocarcinoma at stage 0 and tumour size pT1a than had previous cohorts covering stages II to IV. Hence, further prospective surveys are necessary to confirm this result.
Previous surveys of the prognostic value of preoperational serum carcinoembryonic antigen levels have been contradictory, although we note that such surveys use different thresholds [13, 26]. Previously, we determined that levels higher than 2.12 ng/mL were associated with the prognosis of non-small cell lung cancer, and we now report that patients with levels above this threshold had worse disease-free and overall survival than had patients with levels below this threshold. Indeed, carcinoembryonic antigen levels above 2.12 ng/mL were an independent predictor of unfavourable prognosis. Similarly, Yang et al. [25] found that levels above 5 ng/mL were an independent predictor of recurrence-free and overall survival in stage I lung adenocarcinoma. In contrast, in lung adenocarcinoma patients treated with EGFR tyrosine kinase inhibitors and platinum-based doublet chemotherapy, carcinoembryonic antigen levels > 5 ng/mL were associated with unfavourable prognosis in patients without EGFR mutations, but not in patients with EGFR mutations [14, 28]. Strikingly, we found that overall survival, but not disease-free survival, was poorer for patients with exon 21 mutations than for patients with exon 19 mutations. This result contradicts findings in patients with advanced unresectable lung adenocarcinoma who were treated with EGFR tyrosine kinase inhibitors but is consistent with a survey by Nishii et al. [29]. In patients with carcinoembryonic antigen levels ≥ 2.12 ng/mL, overall and disease-free survival was also better in patients with exon 21 mutations than in those with exon 19 mutations. However, this relationship was not observed in patients with serum carcinoembryonic antigen levels below 2.12 ng/mL, presumably because the antigen is antiapoptotic. In addition, activation of downstream molecules by EGFR mutants may promote antiapoptotic activity, or mutated EGFR may elicit abundant expression of the antigen [26, 30]. We also analysed the prognosis of patients excluding those at stage 0; however, the results were similar to those obtained for the overall cohort, which confirms the prognostic role of carcinoembryonic antigen and EGFR.
Ultimately, we found that carcinoembryonic antigen levels ≥ 2.12 ng/mL, IAC subtype, lymphatic metastasis, and advanced pathological stage were predictors of worse progression-free survival. Similarly, exon 19 mutations in EGFR were an independent predictor of reduced overall survival, especially in patients with carcinoembryonic antigen levels above 2.12 ng/mL. The mechanism underlying the association of carcinoembryonic antigen with EGFR mutations remains unclear, and molecular studies are needed to investigate the difference in proliferation and survival between tumours with exon 19 and exon 21 mutations.
Our survey is limited by its retrospective nature, inclusion of several stage 0 patients, small sample size, and patient recruitment in a single institution in Eastern China, which may have resulted in selection bias. Nevertheless, the data imply that in pathological T1 lung adenocarcinoma, EGFR mutations are associated with preoperational serum carcinoembryonic levels ≥ 2.12 ng/mL. The data also imply that patients with exon 19 deletions in EGFR have less favourable prognosis than had those with exon 21 mutations after curative resection of the lung, especially in patients with carcinoembryonic antigen levels above ≥2.12 ng/mL. Thus, carcinoembryonic antigen levels and EGFR genotype should be considered together to assess prognosis in pathological T1 lung adenocarcinoma.
Disclosure
The funders had no role in study design, data collection or analysis, decision to publish, or preparation of the manuscript.
Conflicts of Interest
The authors declare that they have no competing interests.
Authors’ Contributions
Wang-Yu Zhu and Hai-Feng Li contributed equally to this work.
Acknowledgments
This research was supported in part by grants from Natural Science Foundation of Zhejiang Province (China) (Grant no. LQ17H160001) and Medical Bureau of Zhejiang Province (China) (Grant nos. 2015ZDA032 and 2016RCB020) to Wang-Yu Zhu and Han-Bo Le, from Science and Technology Department of Zhejiang Province (China) (Grant no. 2016C37008) to Xiao-Fei Hu, and from the Science and Technology Bureau of Zhoushan to Wang-Yu Zhu (Grant no. 2011C12040).