Ectopic hippocampal neurogenesis in adolescent male rats following alcohol dependence
Abstract
The adolescent hippocampus is highly vulnerable to alcohol-induced damage, which could contribute to their increased susceptibility to alcohol use disorder. Altered adult hippocampal neurogenesis represents one potential mechanism by which alcohol (ethanol) affects hippocampal function. Based on the vulnerability of the adolescent hippocampus to alcohol-induced damage, and prior reports of long-term alcohol-induced effects on adult neurogenesis, we predicted adverse effects on adult neurogenesis in the adolescent brain following abstinence from alcohol dependence. Thus, we examined neurogenesis in adolescent male rats during abstinence following a 4-day binge model of alcohol dependence. Bromodeoxyuridine and Ki67 immunohistochemistry revealed a 2.2-fold increase in subgranular zone cell proliferation after 7 days of abstinence. Increased proliferation was followed by a 75% increase in doublecortin expression and a 56% increase in surviving bromodeoxyuridine-labeled cells 14 and 35 days post-ethanol exposure, respectively. The majority of newborn cells in ethanol and control groups co-localized with NeuN, indicating a neuronal phenotype and therefore a 1.6-fold increase in hippocampal neurogenesis during abstinence. Although these results mirror the magnitude of reactive neurogenesis described in adult rat studies, ectopic bromodeoxyuridine and doublecortin positive cells were detected in the molecular layer and hilus of adolescent rats displaying severe withdrawal symptoms, an effect that has not been described in adults. The presence of ectopic neuroblasts suggests that a potential defect exists in the functional incorporation of new neurons into the existing hippocampal circuitry for a subset of rats. Age-related differences in functional incorporation could contribute to the increased vulnerability of the adolescent hippocampus to ethanol.
Introduction
Alcohol consumption during adolescence dramatically increases the probability of developing an alcohol use disorder (AUD; Grant & Dawson 1997). Remarkably, 6.4% of adolescents drink excessively to the point of meeting the diagnostic criteria for an AUD (Merikangas et al. 2010). The adolescent brain reacts distinctly to alcohol compared with adults, especially the adolescent hippocampus, which appears particularly vulnerable to excessive alcohol intake (White & Swartzwelder 2004). Alcohol-dependent adolescents have reduced hippocampal volumes, and binge-drinking adolescents perform worse on hippocampal-dependent tasks (De Bellis et al. 2000; Schweinsburg et al. 2010; Parada et al. 2011). Studies in animal models support that ethanol affects adolescent hippocampal function differently compared with adults. For example, adolescent rats exhibit greater sensitivity to ethanol-mediated inhibition of trace fear conditioning and spatial memory performance (Markwiese et al. 1998; Yttri, Burk & Hunt 2004). In addition, ethanol inhibits N-methyl-d-aspartate receptor-mediated long-term potentiation of CA1 pyramidal neurons and enhances gamma aminobutyric acid A receptor-mediated tonic inhibition of dentate gyrus granule neurons to a greater extent in adolescent rats compared with adults (Pyapali et al. 1999; Fleming, Wilson & Swartzwelder 2007). These studies support the idea that developmental differences in plasticity contribute to the adolescent hippocampus' vulnerability to ethanol-induced dysfunction.
Adult neurogenesis represents an important form of hippocampal plasticity that is critical for declarative memory consolidation (Deng, Aimone & Gage 2010). Multi-potential neural progenitor cells (NPCs) located in the subgranular zone (SGZ) of the dentate gyrus underlie the continuous generation of granule cells (reviewed in Zhao, Deng & Gage 2008). Neurogenesis is composed of distinct mechanisms: cell proliferation, neuronal differentiation, cell migration, survival and circuitry integration, many of which are targeted by ethanol (reviewed in Nixon 2006; Nixon et al. 2010). In rodents, ethanol intoxication appears to dose-dependently interfere with SGZ cell proliferation and newborn neuron survival (Nixon & Crews 2002; Nixon 2006; Crews et al. 2006b; Richardson et al. 2009; Nixon et al. 2010; Contet et al. 2013; Ehlers et al. 2013). However, ethanol intoxication alters neurogenesis differently in adolescents versus adults (Morris et al. 2010a; McClain et al. 2011a). In adult rats, ethanol decreased S-phase (bromodeoxyuridine, BrdU) and G1-M phase (Ki67) cell cycle markers, suggesting that the number of actively proliferating NPCs is reduced by ethanol (Nixon & Crews 2002; Crews et al. 2006a). In contrast, in adolescents, a 4-day binge model of dependence decreased BrdU, but not Ki67, which indicates that ethanol alters cell cycle kinetics rather than reducing the number of proliferating NPCs (Morris et al. 2010a; McClain et al. 2011a).
Alcohol dependence results in distinct effects on neurogenesis during abstinence, and it is not known whether developmental differences contribute to it. Although long-term exposure decreases NPC proliferation and neurogenesis for weeks (Richardson et al. 2009; Taffe et al. 2010; Ehlers et al. 2013), the 4-day binge model of dependence results in a transient burst in NPC proliferation at 1 week of abstinence, which produces a substantial increase in neurogenesis (Nixon & Crews 2004). Interestingly, this increase in neurogenesis after short-term abstinence closely matches the timeline for ‘reactive neurogenesis’ described in models of hippocampal injury (Parent et al. 2006). As ethanol-induced hippocampal damage has been suggested to stimulate this reactive neurogenesis event (Nixon & Crews 2004) and the adolescent hippocampus appears more susceptible to ethanol-induced damage (White & Swartzwelder 2004), we hypothesized that indices of reactive neurogenesis would be greater in alcohol-dependent adolescent rats.
Materials and Methods
Ethanol exposure
Adolescent male Sprague Dawley rats (n = 99, Charles River Laboratories, Portage, MI, USA) were housed individually and maintained on a 12-hour light/dark cycle. Water was available at all times, although chow was removed during the 4-day exposure period. All procedures were approved by the University of Kentucky Institutional Animal Care and Use Committee and adhered to the Guide for the Care and Use of Laboratory Animals. Postnatal day 35 rats (mid-adolescence; Spear 2000) received ethanol (25% w/v, Pharmco-AAPER, Brookfield, CT, USA) in Vanilla Ensure® Plus (Abbott Laboratories, Abbott Park, IL, USA) or isocaloric control diet (42.75% w/v dextrose) intragastrically every 8 hours for 4 days following a procedure modified from Majchrowicz (1975), as reported previously (Morris et al. 2010b). The model was chosen because it produces dependence in 4 days and blood alcohol levels mimic the truly problematic adolescent AUD population (van Hoof et al. 2011). Briefly, an initial 5 g/kg dose of ethanol was given, with successive doses determined from intoxication behavior assessed from a 6-point scale (Supporting Information Table S1). Physical dependence was confirmed by scoring withdrawal behavior (Supporting Information Table S2) in 30-minute intervals, 10–26 hours following the last dose (identical to Morris et al. 2010b). Tail blood samples were collected 90 minutes following the first dose on the third binge day. Blood was centrifuged (1800 g for 5 minutes) and stored at −20°C until analysis. Blood ethanol concentrations were determined in triplicate from plasma using an AM1 alcohol analyzer calibrated with a 300 mg/dl external standard (Analox Instruments USA, Lunenburg, MA, USA).
BrdU administration
To label proliferating cells, rats were administered BrdU (300 mg/kg, i.p., Fluka, Buchs, Switzerland or Roche, Mannheim, Germany) 2, 7 or 14 days following the last dose and were euthanized 2 hours later (Fig. 1a). To examine differentiation and neurogenesis, a second group of rats was injected with BrdU at 7 days post-binge and was euthanized 28 days later (Fig. 1b).
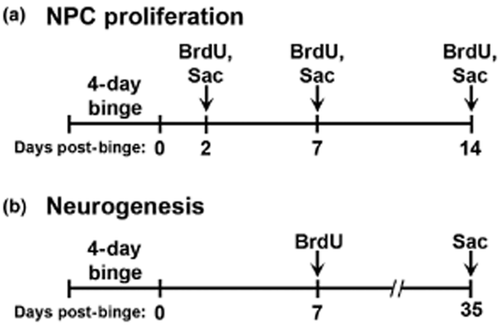
Two bromodeoxyuridine (BrdU) injection paradigms were used. (a) For analysis of subgranular zone (SGZ) NPC proliferation, BrdU (300 mg/kg, i.p.) was injected 2, 7 or 14 days following the 4-day binge ethanol exposure and rats were euthanized (Sac) 2 hours later. (b) For assessment of neurogenesis, BrdU (300 mg/kg, i.p.) was injected 7 days post-ethanol and rats were euthanized 28 days later. Combining detection of BrdU with cell-type specific markers (GFAP and NeuN) allowed assessment of neuronal differentiation with confocal microscopy
Immunohistochemistry
Immunohistochemistry followed standard published procedures (Nixon & Crews 2004; McClain et al. 2011a). Primary antibody information can be found in Table 1. Briefly, rats were euthanized via anesthetic overdose (Nembutal®, MWI Veterinary Supplies, Nampa, ID, USA) and transcardially perfused with 0.1 M phosphate-buffered saline (pH 7.4) followed by 4% paraformaldehyde (pH 7.4). Brains were extracted, post-fixed in 4% paraformaldehyde for 24 hours at 4°C and stored in 0.1 M phosphate-buffered saline at 4°C until sectioning. Twelve series of coronal sections were obtained at 40 μm on a vibrating microtome (Leica Microsystems, Wetzlar, Germany) and stored in cryoprotectant at −20°C until use. Single-label immunohistochemistry was conducted on free-floating tissue sections to detect BrdU (proliferation), Ki67 (proliferation), doublecortin (DCX, immature neurons) and prospero homeobox 1 (Prox1, granule cells). Tissue was washed with Tris-buffered saline (TBS; 20 mM Tris, 500 mM NaCl, pH 7.5, Bio-Rad Laboratories, Inc., Hercules, CA, USA) three times for 5 minutes and then incubated for 30 minutes in 0.6% H2O2 to quench endogenous peroxidase activity. Antigen retrieval (standard sodium citrate for 1 hour at 65°C; McClain et al. 2011a) was used for Ki67 and Prox1, whereas BrdU required DNA denaturing as described by Nixon & Crews (2002). Non-specific binding was blocked in buffer (3% normal serum/0.1–0.2% Triton X-100/TBS) and then sections were incubated in primary antibody for 16 (BrdU) or 42 hours at 4°C. Secondary antibodies were biotinylated horse anti-mouse or goat anti-rabbit IgG (Vector Laboratories, Burlingame, CA, USA) detected with avidin-biotin-peroxidase complex (ABC elite kit, Vector Laboratories) and nickel-enhanced 3,3-diaminobenzidine tetrahydrochloride (Polysciences, Warrington, PA, USA). Ki67 and BrdU stained sections were counterstained with Neutral Red (Fisher Scientific, Pittsburgh, PA, USA) or Cresyl Violet (Fisher Scientific), respectively. All were coverslipped with Cytoseal® (Richard Allen Scientific, Kalamazoo, MI, USA).
| Antibody | Host | Concentration | Source, catalog no. | Used at post-binge timepoint (days) |
|---|---|---|---|---|
| BrdU | Mouse | 0.22 μg/ml | Millipore, Billerica, MA, USA, MAB3424 | 2, 7, 14 |
| BrdU | Rat | 1:400 | Accurate Chemical, Westbury, NY, USA, OBT0030 | 35 |
| Ki67 | Mouse | 1:200 | Vector Laboratories, Burlingame, CA, USA, VP-K452 | 7, 35 |
| DCX | Goat | 0.2 μg/ml | Santa Cruz Biotechnology, Santa Cruz, CA, USA, sc-8066 | 7, 14 |
| NeuN | Mouse | 1:500 | Millipore, MAB377 | 35 |
| GFAP | Rabbit | 1:2000 | DakoCytomation, Carpinteria, CA, USA, Z 0334 | 35 |
| Prox1 | Goat | 1 μg/ml | R&D Systems, Minneapolis, MN, USA, AF2727 | 14 |
- BrdU = bromodeoxyuridine; DCX = doublecortin; GFAP = glial fibrillary acidic protein; NeuN = neuronal nuclei; Prox-1 = prospero homeobox 1.
Fluorescent, triple-label immunohistochemistry for BrdU (rat anti-BrdU), neurons (mouse anti-NeuN) and astrocytes [rabbit anti-glial fibrillary acidic protein (GFAP)] was conducted with goat anti-mouse IgG Alexa Fluor 633, goat anti-rabbit IgG Alexa Fluor 546 and goat anti-rat IgG Alexa Fluor 488 (1:200, Invitrogen, San Diego, CA, USA) secondary antibodies, with Pro-long® Gold anti-fade reagent (Invitrogen) identical to that previously described (Nixon & Crews 2004).
Quantification
All slides were coded to ensure that experimenters were blinded to treatment conditions. For proliferation studies, BrdU and Ki67 labeling were analyzed on an Olympus BX-41 microscope equipped with an UPlanSApo 100× oil immersion lens (1.4 numerical aperture, Olympus, Center Valley, PA, USA). Only cells within the SGZ (∼50-μm ribbon of tissue between the hilus and the granule cell layer) of the dorsal dentate gyrus (bregma −1.8 to −5.52; Paxinos & Watson 2009) were counted. Data were expressed as cells/section. A profile counting methodology was chosen due to the low number of cells to be counted, the non-homogeneous distribution of these cells in the SGZ, the lack of change in the volume in the dentate gyrus in this model (Leasure & Nixon 2010), and previous work demonstrating that this method obtains results similar in relative difference to stereology (Crews, Nixon & Wilkie 2004; Noori & Fornal 2011). For differentiation studies, profile counts of the number of BrdU+ cells within the granule cell layer were obtained on an Olympus BX-51 microscope with ProScan II motorized stage, microcator, DP70 digital camera, 488λ epifluorescence cube and PlanApo 40× lens (0.9 numerical aperture, Olympus). Data were reported as cells/section.
DCX immunoreactivity was measured by Visiomorph image analysis software on the BX-51 system (version 3.6.4.0, Visiopharm, Hørsholm, Denmark). Multi-panel images containing the dorsal dentate gyrus were collected at 100×. The region of interest was drawn around the SGZ and granule cell layer; image segmentation was performed; and DCX immunoreactivity was calculated as pixels/mm2. Ectopic DCX and Prox1-positive profile counts were obtained from the hilus and molecular layer using the BX-51 microscope and were reported as cells/section.
Triple-label immunohistochemistry was quantified similar to previous reports (Morris et al. 2010a) using a laser scanning confocal microscope (Leica TCS SP5, Leica Microsystems) equipped with argon, helium-neon 543 and helium-neon 633 lasers. The phenotype of new cells was determined by sampling 50 BrdU+ cells in the granule cell layer of each subject using a 63× oil immersion objective. Z-plane image stacks (1024 × 1024 pixels) were taken at 0.84 μm thickness, then reconstructed and analyzed for co-labeling in the X, Y and Z plane. BrdU+ cells that co-labeled with NeuN, GFAP or neither were classified as neurons, astrocytes or other, respectively. The number of new neurons, astrocytes or other cells was calculated by multiplying the number of BrdU+ cells/section by the cell-type proportion.
Enzyme-linked immunosorbent assay (ELISA)
Rats were rapidly decapitated at 2, 7 or 14 days following ethanol exposure. Brains were immediately extracted, and the hippocampus was dissected out, snap-frozen on dry ice and stored at −80°C. Identical to previous reports, tissue was homogenized and brain-derived neurotropic factor (BDNF) content was determined via ELISA kit (Millipore CYT306, Millipore, Billerica, MA, USA) following the manufacturer's instructions (McClain et al. 2011b). Duplicates of all samples, standards and positive controls were run. Absorbance was measured at 450 nm on a DXT880 Multimode Detector plate reader (Beckman Coulter, Brea, CA, USA). BDNF protein concentration was divided by the total protein concentration obtained in a bicinchoninic acid assay and expressed as pg of BDNF/mg of total protein.
Statistics
Statistical analyses were conducted using GraphPad Prism® (version 5.04, GraphPad Software, La Jolla, CA, USA). Immunohistochemical data, blood ethanol concentrations (BECs) and ELISA measurements were compared by two-tailed t-test or analysis of variance (ANOVA), with post hoc tests as appropriate. Non-continuous variables (e.g. behavior scores) were analyzed using the non-parametric Kruskal–Wallis test.
Bartlett's test was used to determine equality of variance between groups. When variances were proportional to the square of the group mean (ectopic DCX and Prox1), cell counts were log-transformed prior to ANOVA and post hoc Tukey's tests (Bland & Altman 1996). Correlations between non-continuous variables (intoxication and withdrawal scores) and continuous neurogenesis measures were examined with Spearman's rank correlation coefficients, whereas BECs and neurogenesis were correlated via Pearson's correlation coefficient. Data are reported as mean ± standard error of the mean and differences were considered significant at P < 0.05.
Results
NPC proliferation increased in adolescent rats during early abstinence
In the BrdU proliferation time course study (Fig. 2), two-way ANOVA indicated a significant diet × time interaction on the number of BrdU+ cells located along the dentate gyrus SGZ [F(2,34) = 5.2, P = 0.01]. Specifically, SGZ BrdU increased by 2.2-fold after 7 days of abstinence (P < 0.01), while no difference between control and ethanol groups was observed at the 2- or 14-day timepoints. At the 2-day time-point, BrdU was observed throughout the hippocampus (Fig. 2b). Our previous work demonstrated that these cells are proliferating microglia, an effect that is remarkably similar between adults (Nixon et al. 2008) and adolescents (McClain et al. 2011b). The current study focused solely on the SGZ as it is the neurogenic region of the hippocampus. Changes in BrdU immunoreactivity were confirmed by measuring Ki67, a protein expressed by all proliferating cells regardless of the cell cycle stage (Kee et al. 2002). Similar to BrdU, Ki67 increased by 2.3-fold [t(8) = 3.4, P = 0.009] in ethanol-dependent rats. Changes in BrdU and Ki67 were not due to the differences in intoxication or withdrawal severity as BECs, peak withdrawal and mean withdrawal behavior were similar across groups (Table 2). In addition, BECs and withdrawal scores did not correlate with BrdU immunoreactivity at any timepoint (Supporting Information Tables S3–S5).
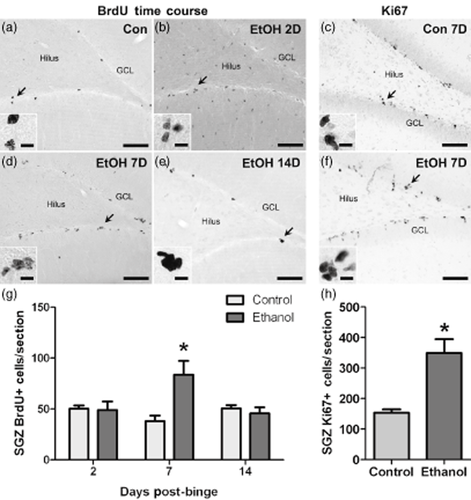
SGZ cell proliferation increases in alcohol-dependent adolescent rats after 1 week of abstinence. Representative images show BrdU immunoreactivity present within multi-cell clusters scattered along the length of the SGZ in control (a) and ethanol-dependent rats after 2 (b), 7 (d) and 14 (e) days of abstinence. Ethanol exposure caused easily discernible changes to BrdU immunoreactivity that varied by recovery timepoint. (g) Profile counts revealed that the number of BrdU+ cells located within the SGZ increased significantly 7 days after binge ethanol exposure. BrdU+ cells detected outside of the neurogenic SGZ at the 2-day timepoint were previously shown to be proliferating microglia (McClain et al. 2011b). Representative images show SGZ Ki67 immunoreactivity in control (c) and ethanol-dependent rats after 7 days of abstinence (f). (h) Similar to BrdU, the number of Ki67+ cells increased significantly after 7 days of abstinence in ethanol-dependent rats. Arrows point to areas represented in the insets. Scale bars = 100 μm (10 μm for insets). *P < 0.05. Samples sizes are as follows: n = 10/group for 2-day timepoint, n = 4–6/group for 7-day timepoint and n = 5/group for 14-day timepoint. BrdU = bromodeoxyuridine; Con = control; EtOH = ethanol; GCL = granule cell layer; SGZ = subgranular zone
| Timepoint (days) | n | BEC (mg/dl) | Peak withdrawal | Mean withdrawal |
|---|---|---|---|---|
| 2 | 10 | 362 ± 20.9 | 3.2 ± 0.14 | 1.5 ± 0.20 |
| 7 | 6 | 314 ± 19.6 | 3.3 ± 0.16 | 1.7 ± 0.26 |
| 14 | 5 | 398 ± 24.5 | 3.1 ± 0.20 | 1.6 ± 0.43 |
- BEC = blood ethanol concentration.
An increase in immature neurons follows increased NPC proliferation
DCX is expressed in developing neuroblasts up to 4 weeks after making the commitment to differentiate into a hippocampal granule cell (Brown et al. 2003). To determine if increased proliferation at the 7-day timepoint enhances granule cell neurogenesis, DCX immunoreactivity was quantified in the dentate gyrus SGZ and granule cell layer after 14 days of abstinence (Fig. 3). A 1.6-fold increase in the ethanol-treated group was observed [t(8) = 5.9, P < 0.001].
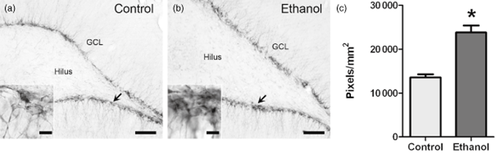
Increased proliferation along the SGZ is followed by enhanced DCX expression 14 days post-ethanol exposure. Representative images show DCX+ cells densely packed along the inner side of the granule cell layer with developing dendrites extending towards the molecular layer 14 days after control (a) or ethanol (b) treatment. Arrows point to areas represented in insets. Scale bars = 100 μm (10 μm for insets). (c) Analysis of immunoreactivity revealed a significant increase in DCX expression in ethanol-dependent rats compared with controls. n = 5/group. *P < 0.05. DCX = doublecortin; GCL = granule cell layer; SGZ = subgranular zone
Neuronal differentiation is not altered during abstinence
Newly born neuroblasts require 3–4 weeks to express mature neuronal markers, such as NeuN (Zhao et al. 2008), and incorporate into the hippocampal circuitry (Espósito et al. 2005). To determine the fate of cells born during the proliferative burst, rats were injected with BrdU at the 7-day timepoint and euthanized 28 days later. Triple-label immunofluorescence (BrdU/NeuN/GFAP) demonstrated that most BrdU+ cells were co-labeled with NeuN (Fig. 4a–i). No BrdU/GFAP co-labeling was observed, similar to previous reports (Morris et al. 2010a), while ∼10% of BrdU+ cells did not co-label with NeuN or GFAP (‘other’). No difference between groups was observed in the proportion of BrdU+ cells that co-labeled with NeuN or neither cell-type marker (‘other’).

Adult hippocampal neurogenesis increases during abstinence, but neuronal differentiation is unaltered. (a) BrdU, (b) NeuN, (c) GFAP and (d) orthogonal view of a representative confocal Z-stack image from a control rat 35 days post-treatment (28 days after BrdU injection to label cells during the proliferative burst). (e) BrdU, (f) NeuN, (g) GFAP and (h) orthogonal view of a representative confocal Z-stack image from an ethanol rat 35 days post-treatment. Scale bars = 20 μm. (i) The graph shows the proportion of sampled BrdU+ cells that were co-labeled with NeuN (neuron), GFAP (astrocyte) or neither (other). No BrdU/GFAP+ cells were observed and there was no effect on the number of BrdU+ cells that were not co-labeled with NeuN or GFAP. (j) The graph shows calculated neurogenesis based on the number of BrdU+ cells present in the granule cell layer multiplied by the cell phenotype proportions. The number of surviving BrdU+ cells was significantly higher in ethanol-treated rats compared with controls. The increase in BrdU combined with cell phenotype data showed that more neurons were generated during abstinence in ethanol-dependent rats than in controls. There was no significant difference in the number of ‘other’ cells generated. n = 11 for the control group and n = 9 for the ethanol group. *P < 0.05. BrdU = bromodeoxyuridine; Con = control; EtOH = ethanol; GFAP = glial fibrillary acidic protein; NeuN = neuronal nuclei
Enhanced NPC proliferation during early abstinence leads to an increase in new granule cells
To determine if the effects on NPC proliferation augment the number of new cells produced during abstinence, surviving BrdU+ cells labeled during the proliferative burst were quantified 28 days later (i.e. 35 days post-ethanol exposure). Profile counts revealed that 1.6-fold more BrdU+ cells survived in the ethanol group compared with the control group [t(18) = 3.3, P = 0.004] (Fig. 4j). The number of new neurons was estimated by multiplying BrdU counts by the proportion of BrdU+ cells that co-localized with NeuN (Fig. 4j). Similarly, a 1.6-fold increase in the number of new granule cells was observed [t(18) = 2.7, P = 0.015]. No statistically significant effect was detected among cells that differentiated into cells other than neurons or astrocytes [t(18) = 2.1, P = 0.054].
Ectopic neurogenesis in adolescent rats that experience severe withdrawal
While quantifying NPC proliferation, a striking number of BrdU+ cells were detected in the dentate gyrus molecular layer of some rats (Fig. 5a,c). In addition, ectopic DCX staining was evident in the hilus and molecular layer of a subset of rats at the 7- and 14-day timepoints, suggesting that immature neuronal cells were generated in non-neurogenic areas (Fig. 5b,d). Knowing the relationship between seizures and ectopic neurogenesis (Parent et al. 2006) and that some rats in this model experience seizures, we examined the relationship between ethanol withdrawal severity and ectopic BrdU and DCX. Therefore, a second study was carried out to generate additional rats for the 14-day timepoint (4 control, 11 ethanol). Intoxication and SGZ DCX measurements closely matched those described for rats used in the 14-day BrdU proliferation experiment (Table 3 and Supporting Information Fig. S1); therefore, rats from both studies were combined (9 control, 16 ethanol).
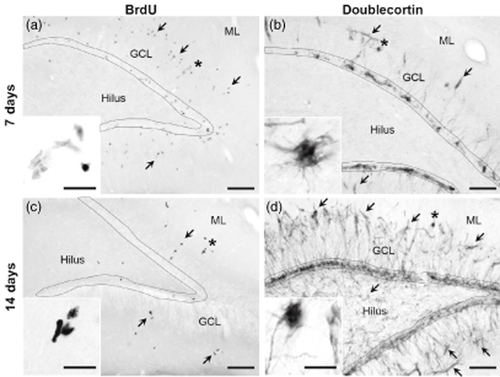
Evidence for ectopic neurogenesis in a subset of rats that experienced severe ethanol withdrawal. Images show a single case study from the 7 and 14 day timepoints to demonstrate ectopic appearance of proliferation (BrdU) and neurogenesis (DCX) markers. The SGZ was outlined to show where BrdU and DCX immunoreactive cells are normally found. Arrows point to ectopic cells in the molecular layer and hilus where BrdU and DCX are not normally expressed. Ectopic BrdU (a) and DCX staining (b) was detected in the molecular layer at the 7-day timepoint in an ethanol-dependent adolescent rat that experienced severe symptoms of ethanol withdrawal. Clusters of BrdU+ cells (c) and DCX+ cells (d) persist in the molecular layer after 14 days of abstinence. DCX+ cells are also evident in the hilus at this timepoint. Compare images of ectopic neurogenesis in this figure to images showing a lack of ectopic neurogenesis in other ethanol-dependent rats in Figs 2 and 3. * denotes areas represented in insets. Scale bars = 100 μm (20 μm for insets). BrdU = bromodeoxyuridine; GCL = granule cell layer; ML = molecular layer
| Timepoint (days) | n | BEC (mg/dl) | Peak withdrawal | Mean withdrawal |
|---|---|---|---|---|
| 2 (ELISA) | 7 | 346 ± 31.1 | 2.6 ± 0.20 | 0.52 ± 0.20 |
| 7 (ELISA) | 6 | 382 ± 14.3 | 3.4 ± 0.14 | 1.20 ± 0.30 |
| 14 (ELISA) | 5 | 428 ± 20.6 | 3.4 ± 0.18 | 1.80 ± 0.31 |
| 14 (IHC) | 11 | 382 ± 16.6 | 3.3 ± 0.17 | 1.50 ± 0.33 |
- BEC = blood ethanol concentration; ELISA = enzyme-linked immunosorbent assay; IHC = immunohistochemistry.
Because of the positive correlation between the number of ectopic DCX+ cells in the hilus and mean (P = 0.04) and peak (P = 0.02) withdrawal scores (Fig. 6), ethanol-dependent rats were divided into two groups: rats that experienced convulsions or had mean withdrawal scores greater than 2.2 were classified as high withdrawal (HW), while rats with mean withdrawal scores less than 2.2 were classified as low withdrawal (LW). Although arbitrary, 2.2 was chosen because it is the starting point for the more severe, rostral symptoms and was approximately the mean withdrawal score (2.13) for all rats that had an observed seizure in this study. Of the 16 ethanol-dependent rats, 7 met the HW criteria. Because variance was obviously non-homogenous (P < 0.0001 for hilus and molecular layer), data were log-transformed and then analyzed via one-way ANOVA. Ethanol withdrawal had a significant effect on the presence of ectopic DCX+ cells in both the molecular layer [F(2,22) = 23, P < 0.001] and the hilus [F(2,22) = 11, P < 0.001]. Post hoc Tukey's tests confirmed that more DCX+ cells were observed in the hilus of the HW group compared with the control and LW groups (Fig. 7) and that the LW group had greater hilar DCX than the control group. In the molecular layer, the HW group had more DCX+ cells than the other two groups; however, the number of DCX+ cells in the LW group was not different from that in the control group (Fig. 7).
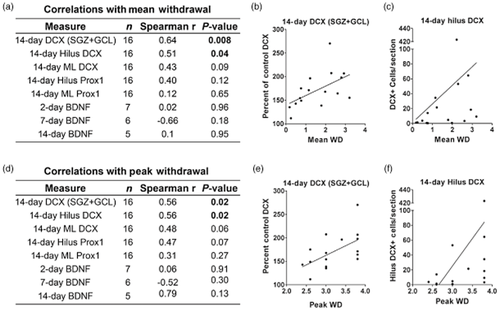
Correlation between measures of ectopic neurogenesis and withdrawal severity. (a) Correlations with mean withdrawal. Spearman's correlation measurements show positive relationships between 14-day DCX (SGZ + GCL) densitometry measurements and mean withdrawal (b) and between 14-day hilus DCX cell counts and mean withdrawal (c). (d) Correlations with peak withdrawal. Spearman correlation measurements show positive relationships between 14-day DCX (SGZ + GCL) densitometry measurements and peak withdrawal (e) and between 14-day hilus DCX cell counts and peak withdrawal (f). BDNF = brain-derived neurotrophic factor; DCX = doublecortin; GCL = granule cell layer; Prox1 = prospero homeobox 1; SGZ = subgranular zone
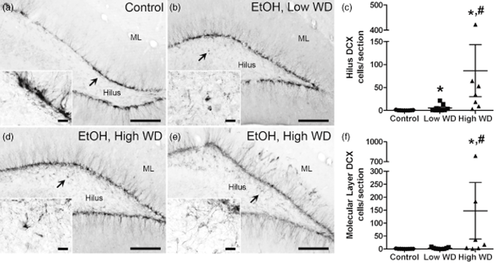
Ectopic neurogenesis was quantified at the 14-day timepoint by counting DCX+ cells in both the molecular layer and the hilus. Representative images show DCX immunoreactivity in control (a, n = 9) and low withdrawal (b, n = 9) groups. Due to the variability within the high withdrawal group (n = 7), two examples are shown. In some, ectopic DCX was restricted to the hilus (d), while in others, ectopic DCX was detected in both the hilus and the molecular layer (e). Scale bars = 200 μm (20 μm for insets). Arrows point to areas represented in the insets. Ethanol withdrawal resulted in a significant increase in the number of ectopic DCX+ cells in the dentate gyrus hilus, regardless of withdrawal severity; however, high withdrawal severity was associated with the greatest degree of ectopic neurogenesis in this region (c). Increased ectopic neurogenesis was only detected in the molecular layer of rats that experienced severe ethanol withdrawal (f). Based on the results from post hoc Tukey's tests, * and # denote significance from control and low WD groups, respectively. Controls are circles, low WD are squares, and high WD are triangles. DCX = doublecortin; EtOH = ethanol; ML = molecular layer; WD = withdrawal
Prox1, a transcription factor expressed by late-stage subgranular NPCs (Karalay et al. 2011; Iwano et al. 2012), was measured to determine if ectopic neuroblasts expressed markers consistent with granule cell commitment (Fig. 8). Variance between groups was statistically different (P < 0.001 for hilus and molecular layer); therefore, data were log-transformed and analyzed by one-way ANOVA. Ethanol withdrawal had a significant effect on the number of Prox1+ cells located in the hilus [F(2,22) = 9.4, P = 0.001]. Post hoc Tukey's tests revealed that the number of hilar Prox1+ cells was greater in the HW group compared with the LW or control groups (Fig. 8c). Surprisingly, no withdrawal effect was observed on molecular layer Prox1 expression [F(2,22) = 2.4, P = 0.12].
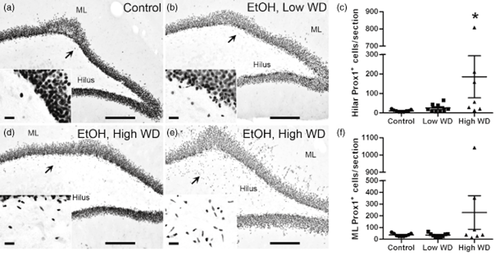
Ectopic cells in the hilus and molecular layer express granule cell-specific Prox1. Representative images show Prox1 expression in control (a, n = 9) and low withdrawal groups (b, n = 9). Due to the variability within the high withdrawal group (n = 7), two examples are shown. In some, ectopic Prox1 was restricted to the hilus (d), while in others, ectopic Prox1 was detected in both the hilus and the molecular layer (e). Scale bars = 200 μm (20 μm for insets). Arrows point to areas represented in the insets. Severe ethanol withdrawal was associated with a significant increase in the number of Prox1+ cells present in the hilus (c). No significant effect of withdrawal severity was found in the molecular layer despite clear evidence of numerous Prox1+ cells observed in a select few rats of the high ethanol withdrawal group (f). Based on the results from post hoc Tukey's tests, * denotes significance compared with the control and low WD groups. Controls are circles, low WD are squares, and high WD are triangles. EtOH = ethanol; ML = molecular layer; Prox1 = prospero homeobox 1; WD = withdrawal
An increase in BDNF can trigger aberrant hippocampal NPC migration (Scharfman et al. 2005). To examine if hippocampal BDNF expression changes in parallel with the appearance of ectopic neuroblasts, rats were subjected to the 4-day ethanol exposure paradigm and then sacrificed 2, 7 or 14 days later (Table 3). BDNF was detected in all samples; however, the amount of hippocampal BDNF was unchanged after 2 (control = 249 ± 8.7 μg BDNF/mg protein; ethanol = 211 ± 25.8 μg BDNF/mg protein), 7 (control = 231 ± 9.7 μg BDNF/mg protein; ethanol = 238 ± 17.8 μg BDNF/mg protein) and 14 (control = 257 ± 10.1 μg BDNF/mg protein; ethanol = 248 ± 18.4 μg BDNF/mg protein) days of abstinence. In addition, BDNF levels did not correlate with withdrawal severity at any timepoint measured (Fig. 6).
Discussion
These studies demonstrate that adolescent rats exhibit reactive hippocampal neurogenesis after ethanol dependence similar to adult rats. However, striking ectopic neurogenesis was observed in severely withdrawing rats, an effect that may be specific to adolescents in this AUD model. An increase in hippocampal cell proliferation was detected 7 days following 4-day binge ethanol exposure, followed by two indices of increased neurogenesis: enhanced expression of the immature neuronal marker, DCX, and increased BrdU/NeuN co-localization 2 and 4 weeks post-intoxication, respectively. The extent of reactive hippocampal neurogenesis in adolescent rats is similar to that first reported in adult rats subjected to the same 4-day exposure paradigm (Nixon & Crews 2004), suggesting that the underlying mechanisms that regulate NPC proliferation and neuronal differentiation during abstinence are similar in the adolescent and adult brain. This was unexpected given the evidence showing that the adolescent brain, especially the hippocampus, appears more vulnerable to the damaging effects of alcohol compared with adults (Crews et al. 2000; De Bellis et al. 2000; White & Swartzwelder 2004) and that alcohol effects on adult neurogenesis differ between adolescent and adult rats (McClain et al. 2011a).
Ectopic neurogenesis and severe ethanol withdrawal
Most distinct in these data was a clear relationship between severe ethanol withdrawal and aberrant or ectopic neurogenesis, an effect that may be adolescent-specific. Evidence for ectopic neurogenesis included detection of BrdU-labeled and DCX immunoreactive cells within the non-neurogenic molecular layer and hilus of the dentate gyrus in rats that experienced withdrawal-induced seizures or had mean withdrawal scores greater than 2.2. The phenotype of ectopic cells was characterized by examining the expression of Prox1, a member of the homeobox transcription factor family that is required for differentiation of NPCs into granule cells (Karalay et al. 2011) and is essential for the maintenance of the granule cell phenotype (Iwano et al. 2012). Comparable to DCX, severe ethanol withdrawal was associated with an increase in the number of Prox1+ cells in the hilus, indicating that aberrantly migrating NPCs maintain a granule cell phenotype. Similar to the effects of severe ethanol withdrawal in adolescent rats, pilocarpine-induced status epilepticus induces the appearance of Prox1+ cells in the hilus 14 days following seizure induction (Parent et al. 2006). This similarity suggests that it is the seizure component of withdrawal that underlies this phenomenon. However, the withdrawal monitoring protocol used has limitations as the rats are observed only 30 minutes/hour and we cannot rule out that seizures and other critical withdrawal symptoms were missed. Intriguingly, ectopic neurogenesis has not been observed in adult rats exposed to this same alcohol paradigm (Nixon & Crews 2004). Given that withdrawal severity is similar between adolescent and adult rats in this model (Morris et al. 2010b), severe ethanol withdrawal-induced ectopic neurogenesis may be adolescent-specific. When considered along with the abnormal functional characteristics described for seizure-induced ectopic granule neurons, the association between adolescence and ethanol-induced ectopic neurogenesis may explain, in part, the high sensitivity of the adolescent hippocampus to alcohol-induced dysfunction (White & Swartzwelder 2004; Schweinsburg et al. 2010; Parada et al. 2011).
The presence of ectopic neurogenesis indicates that severe withdrawal following adolescent alcohol dependence interferes with mechanisms that control hippocampal NPC migration. In the dentate gyrus, a role for BDNF in NPC migration has been suggested as direct infusion of BDNF into the hilus results in the appearance of ectopic granule cells (Scharfman et al. 2005). Coupled with evidence from adult rodent alcohol models and human alcoholics supporting a potential role for BDNF in AUDs (Miller 2004; Costa et al. 2011), we hypothesized that severe ethanol withdrawal and ectopic neurogenesis would be linked with an increase in hippocampal BDNF expression. Surprisingly, analysis of BDNF protein at 2, 7 and 14 days post-ethanol exposure revealed no change in BDNF expression and no correlation between BDNF and measures of withdrawal severity. Although we cannot completely rule out a role for BDNF based on limited ELISA measurements, the data suggest that other factors are involved in aberrant NPC migration. For example, reduced reelin expression and altered radial glial positioning and morphology have been associated with ectopic neurogenesis (Haas et al. 2002; Mooney, Siegenthaler & Miller 2004; Gong et al. 2007). Future studies will examine the contribution of reelin and radial glia to ectopic neurogenesis observed following alcohol dependence in adolescent rats.
Functional consequences of reactive hippocampal neurogenesis
Adult born granule cells become fully integrated into the existing hippocampal circuitry 4–8 weeks following birth (Espósito et al. 2005; Zhao et al. 2008). Although the precise function of adult hippocampal neurogenesis remains heavily debated, evidence from behavioral studies links neurogenesis to hippocampal-dependent learning and memory processes (Deng et al. 2010). The emerging data implicating adult born granule cells in learning and memory suggest that restoration of hippocampal neurogenesis may be involved in cognitive improvement exhibited by abstinent alcoholics (Sullivan et al. 2000; Nixon & Crews 2004; Bartels et al. 2007). The beneficial effects of neurogenesis combined with increased neurogenesis detected in early abstinence supports this intriguing idea. Although ethanol-exposed adolescent rats develop deficits in hippocampal-dependent learning and memory tasks (reviewed in White & Swartzwelder 2004; Yttri et al. 2004), it is not known if alterations in neurogenesis are involved, nor is it clear if reactive neurogenesis contributes to recovery of these deficits. Most tests of hippocampal function occur within several days to 3 weeks of ethanol exposure (Yttri et al. 2004; Coleman et al. 2011), while functional effects of reactive neurogenesis may not be observed until 6 weeks after cell birth (Kee et al. 2007). Additional work is needed to determine if new neurons generated during abstinence properly integrate into the existing hippocampal circuitry to provide beneficial effects on hippocampal function. These studies will be particularly important given the evidence for ectopic neurogenesis in adolescent rats that experienced severe ethanol withdrawal.
Conclusions
Human and animal studies consistently demonstrate that excessive ethanol intake typical of AUDs causes impairments in hippocampal structure and function. These detrimental effects have been hypothesized to underlie the role of the hippocampus in the development of AUDs (Nixon et al. 2010). The effects of adolescent ethanol intoxication on neurogenesis, which include disruption of NPC proliferation and reduced survival of adult born granule neurons (Taffe et al. 2010; Morris et al. 2010a; McClain et al. 2011a), could have important implications for how impairments in hippocampal integrity contribute to the development of addiction (Mandyam & Koob 2012). Reactive neurogenesis may be an attempt to compensate for lost neurogenesis and could represent a key intrinsic mechanism to repair structural damage and improve cognitive function during recovery (Nixon 2006; Mandyam & Koob 2012). Conversely, ectopic neurogenesis potentially disrupts normal dentate gyrus circuitry and may hinder functional recovery (Parent 2007), underscoring the importance for investigating whether neurons born during reactive neurogenesis integrate correctly into the existing hippocampal circuitry. A more complete understanding of the causes and role of alcohol-induced reactive neurogenesis is critical for developing therapies aimed at promoting recovery of hippocampal function in adolescents with AUDs.
Acknowledgements
The authors would like to thank M. Ayumi Deeny for her expert technical assistance with immunohistochemistry and densitometry measurements. This work was supported by NIAAA grants R21 AA016307 and R01 AA016959 and NIDA grant T32 DA016176.
Authors Contribution
JAM, SA Morris and KN were responsible for study concept and design. JAM performed cell proliferation and ectopic neurogenesis experiments. SA Morris completed experiments examining NPC differentiation and survival. SA Marshall performed ELISA experiments. JAM, SA Morris, SA Marshall and KN analyzed data and interpreted the findings. JAM drafted the manuscript. JAM and KN critically evaluated and revised the manuscript for intellectual content. All authors critically reviewed content and approved the final version for publication.




