Nationwide Assessment of Patient Trajectories in Mantle Cell Lymphoma: The Swedish MCLcomplete Project
Previous presentation: Initial data were presented at the American Society of Hematology annual meeting 2020.
Supplemental digital content is available for this article.
Graphical Abstract
Abstract
Mantle cell lymphoma (MCL) is a B-cell malignancy currently considered incurable. Although some patients obtain prolonged remission after first-line chemoimmunotherapy, many will need several treatment lines. Here, we present a nationwide assessment of treatment strategies, time to progression and survival in MCL. All patients diagnosed with MCL 2006–2018 were identified in the Swedish Lymphoma Register. Information on all lines of therapy was extracted from the medical records. Overall and progression-free survival (OS and PFS) were assessed through August 2021. In total, 1367 patients were included (median age, 71 years) and median follow-up was 6.8 years. Two hundred and one (15%) were managed initially with watch-and-wait, but 1235 (90%) eventually received treatment. The most frequently used first-line regimens were rituximab-bendamustine (BR) (n = 368; 30%) and Nordic MCL2 (n = 342; 28%). During follow-up, 630 patients (46%) experienced relapse/progression and 546 (40%) received second-line treatment. The most frequently used second-line regimen was BR (n = 185; 34%) but otherwise a wide variety of second-line treatments were used. Further, 382 and 228 patients experienced a second or third relapse/progression, respectively. Median PFS after first (PFS-1), second (PFS-2), third (PFS-3), and fourth (PFS-4) treatment lines was 29.4, 8.9, 4.3, and 2.7 months. Patients with early progression, defined as a PFS-1 <24 months, had an inferior median OS of 13 versus 37 months in patients with later relapse. For patients treated with frontline BR, however, time to relapse had no impact on later outcome. By use of nationwide population-based data, we provide important benchmarks for future studies of all treatment lines in MCL.
INTRODUCTION
Mantle cell lymphoma (MCL) is a B-cell malignancy mainly diagnosed in the elderly population, and more commonly among males. It is among the more recently defined forms of B-cell lymphomas, when the association between centrocytic morphology and the presence of a translocation t(11;14) was made in the 1990s,1. defining what is now classified as MCL.
Since then, the lymphoma community has been increasingly aware that patients with MCL require specific treatment, compared with other B-cell lymphomas. The first piece of evidence was that consolidation with high-dose chemotherapy with autologous stem cell transplant (ASCT) improved progression-free survival (PFS) in younger patients.2. The outcome of this group was shown to improve further by the addition of cytarabine and rituximab,3.,4. the latter both during induction and as maintenance.5.
In patients ineligible for ASCT, several chemoimmunotherapy combinations are used. These include R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone) with rituximab maintenance, showing superiority compared with rituximab-fludarabine-cyclophosphamide.6. Other commonly used combinations are BR (rituximab-bendamustine), which showed superior PFS and quality of life compared with R-CHOP.7. Also, VR-CAP (bortezomib, rituximab, cyclophosphamide, doxorubicin, and prednisone) has shown superior overall survival (OS) compared with R-CHOP,8. but is associated with more hematological toxicity compared with BR. The benefit of rituximab maintenance, also after BR, was recently confirmed in a large US real-world analysis.9.
As MCL is still not a curable disease, patients will eventually, if surviving long enough, experience a relapse. The optimal sequence of therapies for patients relapsing after frontline treatment is currently not settled. One important factor is time to relapse. Patients initially receiving intensive regimens including high-dose cytarabine with disease progression within 24 months from start of treatment (POD24) have an inferior survival, and benefit more from treatment with ibrutinib compared with chemoimmunotherapy in second line.10.
Here, we present the first data from the Swedish MCLcomplete project, aiming to provide a more complete view of treatment trajectories in MCL on a national population-based level, to serve as a benchmark for comparing efficacy against novel agents and combinations in different lines of therapy. We also aimed to investigate outcomes by type of first-line treatment, number of treatment lines, and timing of progression.
PATIENTS AND METHODS
The Swedish lymphoma register (SLR) is a nationwide quality-of-care register, which has been in use since 2000. Data on baseline clinical characteristics and primary treatment are entered through a standardized electronic form. Compared with the Swedish Cancer Register, to which all new cases of malignancies are reported by a compulsory reporting system, cases identified and reported to SLR has a coverage of ≈95%.11. The SLR is regularly linked to the Swedish population register for information on vital status and dates of death, with virtually no loss to follow-up.
In the present study, the SLR was used to identify all patients diagnosed with MCL during the period 2006–2018. For this population, medical records were reviewed to validate the information regarding response to first-line treatment and occurrence of relapse, and complement with second and additional treatment lines and relapses. Specially trained research nurses and/or medical staff at local hospitals performed the medical record review and updated the register within the regular register reporting system. In Sweden, all residents diagnosed with MCL are managed in specialist care covered by the state-financed social security system.
Clinical characteristics at diagnosis (age, sex, performance status, Ann Arbor stage, and prognostic factors needed for the calculation of the MCL International Prognostic Index [MIPI]) were also retrieved from the updated SLR.
Recorded information on watch-and-wait in the medical records was retrieved during the data collection for >80% of the patients in the cohort. For the subset of patients that did not have this information, watch-and-wait was defined as treatment start >3 months after the diagnosis.
First-, second-, and third-line treatments were categorized according to the received regimen (regardless of if it was administered after an initial period of watch-and-wait or not) into chemoimmunotherapy with or without radiotherapy, single radiotherapy or single rituximab (second- and third-line). The most common specific types of systemic treatment in each treatment line were also presented.
Progression of disease was defined as either progressive or stable disease as best response at evaluation after end of treatment or as relapse. Progressions occurring within 24 months from treatment start were defined as POD24.
Statistical methods
Clinical characteristics were summarized for all patients at diagnosis.
Patients were followed either from date of treatment start (in the analysis where the patients were categorized according to the treatment regimens) or otherwise from date of primary diagnosis or date of relapse/progression).The end point for OS was defined as death due to any cause and for PFS it was defined as either progression or death. Patients were additionally censored at the date of the last medical record review, between June 2020 and August 2021, depending on at which hospital the patient was treated.
OS and PFS were shown by treatment regimen (first- and second-line), initial management (initially treated versus watch-and-wait), by order of relapse/progression (first-, second-, and third progression), and by early versus late first and second progression (POD24) using the Kaplan-Meier method. Patients were further stratified into age-groups (>70 years/≤70 years) according to their age at primary diagnosis or relapse/progression.
In analysis of maintenance treatment, delayed entry was used to avoid immortal time bias (ie, follow-up was started at 6 months after the initial treatment ended). For the same reason, in the analysis of initially treated versus watch-and-wait, we only included patients with known intent from diagnosis (collected in the medical records review).
Due to the observational nature of the data (ie, the presence of confounding by indication of administered treatment), we did not test for significant differences in PFS or OS via log-rank tests.
All statistical analyses were performed using Stata (StatCorp, 2017. Stata Statistical Software: Release 17. College station, TX: StataCorp LLC).
Ethics
The study was approved by the Regional Board of the Ethical Committee in Lund, Sweden (2018/739).
RESULTS
In total, 1412 patients were recorded with a diagnosis of MCL in the SLR between January 1, 2006 and December 31, 2018. Of those 1367 (96.8%) were included in this study. Reasons for exclusion were either that the medical record could not be retrieved (n = 24; 1.7%) or that the MCL diagnosis was revised as a result of the medical record review (n = 21; 1.5%) (Figure 1). Clinical data for the 1367 patients in the final cohort are presented in Table 1. Median age at diagnosis was 71 years, the majority of the patients were male (72%), and 69% initially presented with stage IV disease.
Initial management
Most patients received active treatment at diagnosis (n = 1075; 78.6%) whereas 201 (14.7%) patients were managed with a watch-and-wait approach (of whom 134 were recorded as watch-and-wait in the medical records review and 67 patients were treated at hospitals from where information on watch-and-wait was not recorded but had a delayed treatment start of >3 months). In 91 patients (6.7%), treatment was never administered (for other reasons than watch-and-wait), or data on initial treatment was missing. For the 134 patients managed with a known watch-and-wait strategy from diagnosis, median time to treatment start or death was 1.7 years (95% confidence interval [CI], 1.1-1.9), which can be compared with a median PFS of 2.7 years (95% CI, 2.3-3.0) for patients treated at diagnosis. Median OS in the watch-and-wait group was 7.1 years (95% CI, 5.1-9.9), compared with 4.7 years (95% CI, 4.3-5.3) among patients receiving initial treatment (Suppl. Table S1).
| Total | 1367 | (100) |
|---|---|---|
| Median follow-up in years (IQ range) | 3.9 | (1.3–6.8) |
| Sex | ||
| Male | 986 | (72) |
| Female | 381 | (28) |
| Median age in years (IQ range) | 71 | (64–79) |
| Elevated S-LD | ||
| No | 758 | (55) |
| Yes | 564 | (41) |
| Missing | 45 | (3) |
| Performance status (WHO) | ||
| 0 | 691 | (51) |
| 1 | 450 | (33) |
| 2 | 113 | (8) |
| 3 | 66 | (5) |
| 4 | 24 | (2) |
| Missing | 23 | (2) |
| MIPI | ||
| Low | 168 | (12) |
| Intermediate | 385 | (28) |
| High | 635 | (47) |
| Missing | 179 | (13) |
| Stage | ||
| Ann Arbor I | 78 | (6) |
| Ann Arbor II | 125 | (9) |
| Ann Arbor III | 164 | (12) |
| Ann Arbor IV | 945 | (69) |
| Missing | 55 | (4) |
- IQ = interquartile; MIPI = MCL International Prognostic Index; S-LD = S-lactate dehydrogenase; WHO = World Health Organization.
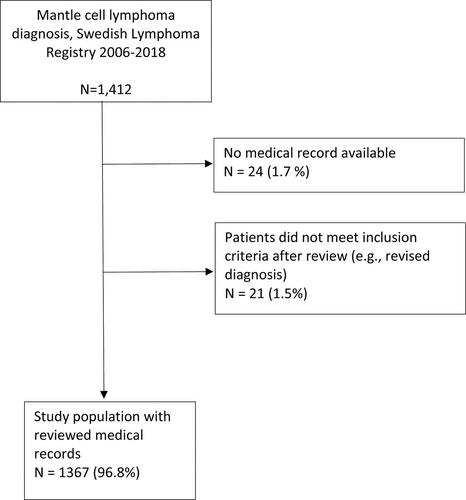
Flowchart describing the inclusion and exclusion of all 1412 patients diagnosed with mantle cell lymphoma in Sweden 2006–2018.
Chemoimmunotherapy
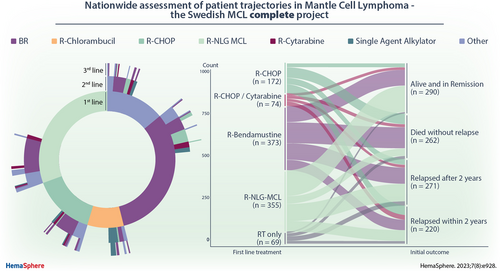
A total of 1235 patients were treated with any type of therapy (including patients treated after a period of watch-and-wait). Of these, 117 patients (9.5%) received treatment within a clinical trial. A majority, 1145 (92.7%) patients, received chemoimmunotherapy (28 in combination with radiotherapy). The most common regimens were BR (n = 368; 28.9%), the Nordic MCL2 protocol (n = 342; 27.7%), and R-CHOP (n = 165; 13.4%) (Figure 2). Treatment patterns changed during the evaluated time period, most notably with an increase in use of BR (Suppl. Figure S1). In total, 303 (24.5%) received consolidation with ASCT, all after Nordic MCL2. Median PFS from treatment initiation for BR, Nordic MCL2, and R-CHOP was 2.7 (95% CI, 2.1-3.2), 5.1 (95% CI, 4.2-6.1), and 1.5 (95% CI, 1.1-2.0) years, and median OS 4.1 (95% CI, 3.5-5.0), 11.7 (95% CI, 7.8-not reached), and 2.9 (95% CI, 2.2-4.1) years, respectively (Figure 3). Patient characteristics differed between the groups in terms of age and MIPI (Suppl. Table S2).
A minority of patients (n = 137; 11.9%) started postremission maintenance with rituximab. For this group, median PFS and OS, calculated from 6 months after end of first-line treatment, was 3.8 (95% CI, 3.2-5.4) and 6.0 (95% CI, 4.1) years, compared with 2.8 (95% CI, 2.4-3.1) and 4.7 (95% CI, 4.1-5.2) years, respectively, for the population not receiving maintenance rituximab (Suppl. Table S3).
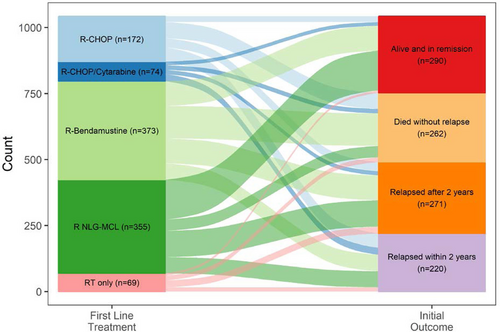
Sankey plot of first-line treatment types and outcome among MCL patients diagnosed in Sweden 2006–2018 and followed through 2021. MCL = mantle cell lymphoma.
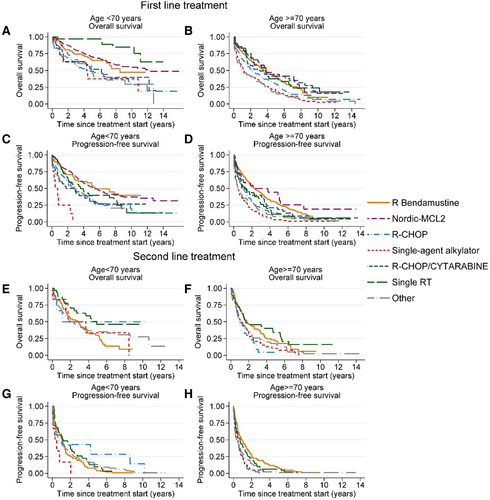
Overall and progression-free survival from treatment start for the most common treatment regimens used in first- and second-line of treatment. (A) Overall survival, first-line, <70 y. (B) Overall survival, first-line, ≥70 y. (C) Progression-free survival, first-line, <70 y. (D) Progression-free survival, first-line, ≥70 y. (E) Overall survival, second-line, <70 y. (F) Overall survival, second-line, ≥70 y. (G) Progression-free survival, second-line, <70 y. (H) Progression-free survival, second line, ≥70 y. *The group “other” includes R-CVP, R-FC, R-Cytarabine, Ibrutinib, and other more uncommon regimens. R-CVP = rituximab-cyclophosphamide, vincristine, prednisone; R-FC = rituximab-fludarabine-cyclophosphamide.
Radiotherapy
Among 69 patients who received radiotherapy as single primary treatment (5.6% of all treated patients, median age 71 years), median PFS was relatively short (2.2 years [95% CI, 1.3-3.4]) but median OS was 10.2 years (95% CI, 6.1; not reached), (Figure 3). Fifty-five of the 69 (79.7%) presented with stage I–II disease, indicating that radiotherapy was given with curative intent. For this subgroup, the median PFS was 2.8 years (95% CI, 1.8-4.1) and median OS 11.1 years (95% CI, 7.6; not reached). Fourteen of the 55 (25%) presented with MALT-type MCL, with predominantly extranodal disease.12.
Relapse treatment
During a median follow-up of 3.9 years (range, 0–16.1 years) from primary diagnosis, 630 patients (46%) experienced at least 1 relapse, and 382 (27.9%) experienced 2 or more relapses. In total, 854 (62.5) patients died during follow-up.
Among patients that relapsed (n = 630), 546 (86.7%) received second-line treatment (Figure 4) and of those, 423 (77.5%) received chemoimmunotherapy. The most common second-line regimen was BR (n = 185; 33.9%), but otherwise, a wide range of treatments were used, and no other large treatment subgroups could be identified (Figure 5). Median PFS from initiation of second-line treatment was very short regardless of regimen, for example, for BR PFS was 1.2 years (95% CI, 0.9-1.4), and median OS 2.2 years (95% CI, 1.6-3.0), respectively (Figure 3).
Three-hundred and eighty-two patients experienced a second relapse, and most (n = 283; 74.1%) received a third treatment line (Figure 4), mainly with chemoimmunotherapy (n = 201). Also in third line, BR was the most used regimen (n = 73; 25.8%). Further, 160 of 228 patients (70.2%) with a third relapse/progression received a fourth treatment line, 91 of 123 (74.0%) a fifth, 49 of 74 (66.2%) a sixth, 21 of 36 (58.3%) a seventh, 16 of 20 (80.0%) an eighth, 4 of 8 (50.0%) a ninth, 4 of 5 (80.0%) a tenth treatment line. One patient received 12 lines of therapy.
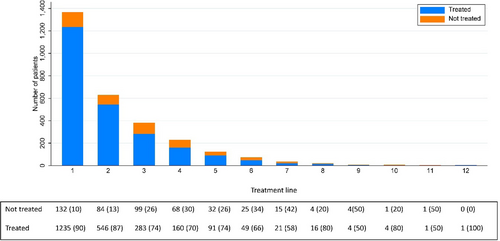
Number of patients receiving treatment or not, according to the treatment line/number of relapse/progression.
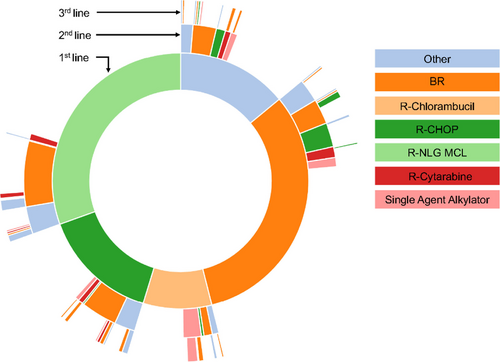
Overview of treatment sequencing in MCL patients diagnosed 2006–2018 in Sweden. The top 5 first-line treatments and the second-/third-line treatments administered to ≥2% of patients are shown. The inner, middle, and outer rings represent first-line, second-line, and third-line treatments, respectively. The individual treatments are shown in descending order of frequency. MCL = mantle cell lymphoma.
OS and PFS after relapse
The median PFS and OS for patients after the first relapse were 0.8 years (95% CI, 0.6-1.0) and 1.6 years (95% CI, 1.5-1.9). PFS and OS were shorter in elderly patients after first relapse (>70 years), but in patients experiencing a second or a third progression, age was of less importance (Figure 6). When evaluating PFS and OS by treatment line, according to the time period, we found no clear improvement in patients diagnosed during the later time period (2013–2018) compared with patients diagnosed earlier (Suppl. Figure S2).
Progression of disease within 24 months
Out of the 630 first progressions, 355 (56.3%) were reported within 24 months from first treatment initiation. Median PFS from time of first progression for patients relapsing within 24 months (POD24) was 0.5 years (95% CI, 0.4-0.6), compared with 1.5 years (95% CI, 1.3-2.0) for patients with a later progression (Figure 7). PFS and OS differed by POD24 to various extents across types of frontline therapy. In patients who received BR as first-line treatment, the OS and PFS were similar in early and late POD24, in contrast to patients receiving Nordic MCL2 or R-CHOP (Figure 8). Of the 382 second progressions, 324 (84.8%) occurred within 24 months of initiation of relapse treatment after the first relapse (POD2). Similar to after first progression, patients with early POD2 had an inferior outcome compared with patients with late POD2 (median PFS 0.3 years [95% CI, 0.3-0.4] versus 1.6 years [95% CI, 1.0-2.0]). In several aspects, the late POD2 population was different from patients with early POD2. More patients with late POD2 only received radiotherapy as first-line treatment (17% versus 4%), or at first relapse (26% versus 13%). At baseline, more patients with late POD2 were without symptoms (75% versus 53%), and more often presented with stage I–II disease (22% versus 10%) and low-risk MIPI (24% versus 7%).
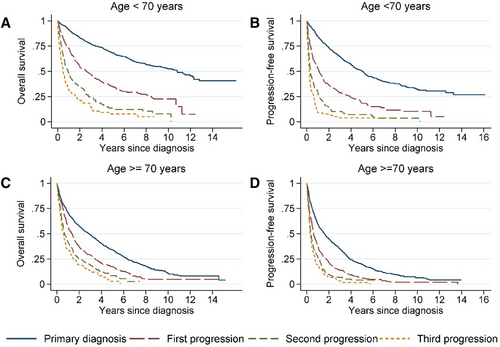
Overall and progression-free survival from date of primary diagnosis, first, second, and third relapse/progression stratified by age <70 vs ≥70 y. Left column: overall survival; Right column: progression-free survival, (A–B) age <70 y; (C–D) age ≥70 y.
Allogeneic stem cell transplantation
In total, 20 patients underwent an allogeneic stem cell transplantation (allo-SCT) during follow-up. Fifteen (75.0%) patients received allo-SCT after the first relapse, 4 (20.0%) after second relapse, and 1 patient after 3 relapses. The median PFS and OS for transplanted patients was 2.0 years (95% CI, 0.8) and 5.9 years (95% CI, 1.2), respectively.
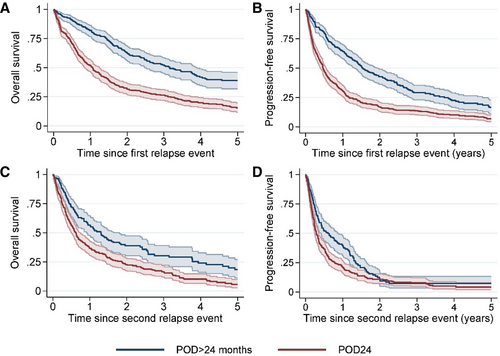
Outcome (5-y overall survival and progression-free survival) in patients with first and second relapse/progression within 24 months from treatment start (POD24) vs patients relapsing later than 24 months from treatment start (POD > 24). (A) Overall survival, first relapse. (B) Progression-free survival, first relapse. (C) Overall survival, second relapse. (D) Progression-free survival, second relapse. POD24 = disease progression within 24 months from start of treatment.
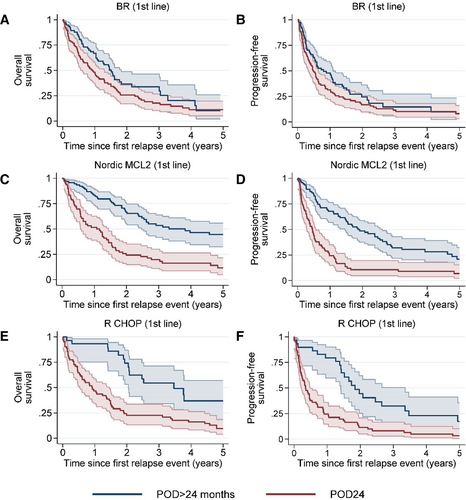
Outcome (5-y overall survival and progression-free survival) in patients with a first relapse/POD24 vs patients relapsing later than 24 months from treatment start (POD > 24) by first-line treatment regimen. Overall survival by time since POD is presented in the left column and progression-free survival by time since POD in the right column. (A–B) BR; (C–D) Nordic MCL2; and (E–F) R-CHOP. BR = rituximab-bendamustine; MCL = mantle cell lymphoma; POD24 = disease progression within 24 months from start of treatment; R-CHOP = rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone.
DISCUSSION
In this study, we present truly population-based benchmark results for MCL patients with several chemoimmunotherapy-based treatment lines, based on the SLR and enriched by a detailed review of medical charts to ensure complete data. We have previously reported OS outcomes following frontline regimens in MCL in Sweden for 2 different time periods, 2000–201113. and 2007–201714. and specifically on ASCT eligible patients over different calendar periods.14.,15. Here, we are also able to present data on PFS and have included all further treatment lines. We note that close to half of the patients experience progression/relapse after first-line treatment, and that outcomes among these patients are poor overall (PFS <12 months), especially among patients with POD24 (PFS <6 months). At the same time, 15% could be managed initially with a watch-and-wait approach, a marked increase from 2% in our previous study, describing patients diagnosed 2000–2011.13. This probably reflects increased awareness of this approach in the lymphoma community, in line with published guidelines.16. The outcome of this group of patients was favorable, with a median OS of >10 years, indicating proper selection of patients for this approach. Patients with limited stage MCL are rare, and standard management is controversial. In this cohort, single modality radiotherapy was given to 27% of patients with stage I–II disease, with favorable results, a median PFS 2.8 years and median OS 11.1 years, indicating that this may be a feasible approach for this population and an approach that may delay the need of systemic treatment.
During recent years, several datasets on real-world data on MCL have been reported, from different populations, including data from the US,17. Belgium,18. and Japan.19. As in the Japanese cohort, BR was the most frequently used regimen in our dataset in first, second, and third-line, reflecting that the majority of MCL patients belong to the elderly population. However, one-fourth of the patients in our cohort received ASCT upfront, clearly higher than the 8% in the Japanese and US cohorts but lower compared with a Belgian study where 44% received ASCT, which may be explained by differences in treatment guidelines, differences in the cohorts, and varying national reimbursement policies. The median PFS in the present cohort was 40 months, compared with a median of 24 months time to next treatment in the US study and in the Japanese cohort. The US cohort was mainly derived from community-based practices and may not reflect the nation as a whole. Another single-center study from a US academic center reported a median PFS of 4 years.20. Median OS, a more solid end point, was 5½ years for initially treated patients in our series, markedly shorter than the >10 years in the US single-center study, a difference likely to reflect the outcome of a nonselected nationwide cohort.
Early progression after frontline therapy, POD24, is associated with poor outcome.10. In our cohort, we could confirm these results on a population level, in patients initially receiving cytarabine-based intensive chemoimmunotherapy or R-CHOP.
In patients treated with BR, however, there was no clear difference between early or late POD. Possibly, this population had less available effective treatment options at relapse, as BR was the most common second-line therapy post other regimens, but may also reflect different distributions of patients in terms of age and biological high-risk features. Furthermore, and perhaps not unexpectedly, patients with early progression after second-line treatment (early POD2) had inferior survival. The minority (15%) with late POD2 had more often received radiotherapy either as first-line or second-line treatment. This may partly explain the favorable outcome, as the first group received their first systemic treatment at relapse, and the second group most likely relapsed with predominantly localized disease.
Visco et al10. were able to show that use of ibrutinib in second line was associated with superior outcome compared with chemoimmunotherapy in early POD24. Due to a low number of patients receiving this agent, this could not be confirmed in our data. In general, the outcome even after first relapse was poor, with a median PFS of <1.5 year, highlighting the need for improved therapies also in relapsed MCL.
Allogeneic transplantation has been suggested to induce long lasting remissions and potential cure in a proportion of relapsed/refractory MCL, but its use is limited to the younger population, due to a substantial risk of nonrelapse mortality (NRM) and severe morbidity. Its role is also less clear when CAR-T cell therapies have entered the treatment algorithm. In an EBMT series, NRM was 24% at 1 year, and 4 year PFS and OS was 31 and 40%, respectively.21. There was only a small population (n = 20) undergoing allo-SCT in the present study. The median PFS was <2 years, indicating that the selection of patients may have been suboptimal.
Rituximab maintenance has been associated with improved OS in MCL, after R-CHOP and after cytarabine-based induction followed by ASCT. In the present series, only a minority of patients received rituximab maintenance, but unlike in previous randomized trials and real-world studies, there was no clear benefit of rituximab, possibly due to large heterogeneity in the various regimens used.
Our study has several limitations. No central review of pathology has been performed, and pathology reports in most cases did not include information on histological subtype and Ki-67 expression. Among the strongest prognostic factors in MCL is the presence of a TP53 mutation, but this analysis was not part of standard diagnostic work-up of MCL during the study period. Another factor limiting the generalizability of our results is that ibrutinib and other BTK-inhibitors (BTKi) were not reimbursed in Sweden during the study period and were used to a lesser extent than in the United States and in other countries. In our cohort, most patients (54%) did not experience a first relapse. However, this may change with prolonged follow-up, which is still relatively short, with a median of 6.8 years.
It is important to point out that the management of patients with MCL is currently undergoing radical changes. Results from the recently presented TRIANGLE study support the frontline use of BTKi, and question the utility of ASCT.22. In patients progressing on BTKi, we now have access to CAR-T cell therapy and noncovalent BTKi:s, and we also expect that bispecific antibodies will enter our treatment algorithms. This means that the present series represent a bygone treatment era, but may serve as a benchmark from which we can measure the expected improvement in outcome.
The MCLcomplete project is still not complete. As a next step, our data will be linked to national health care databases including data on comorbidity, medications, socioeconomic factors, other malignancies and data on sick leave and disability pension. The aim is to depict a nationwide description of MCL at an even higher level of granularity.
We conclude from this population-based cohort that outcomes of MCL patients diagnosed and treated in clinical routine has become more variable with an increasing proportion that can be successfully managed with initial watch-and-wait. Still, in spite of standard intensive first-line chemoimmuntherapy regimens and existing consolidative strategies, many patients experience progression or relapse with generally poor outcome regardless of age. It will be important within the next few years to understand how novel targeted and cellular treatments should be coupled with molecular subtyping to maximize benefit for all patients with MCL.
AUTHOR CONTRIBUTIONS
MJ, KES, IG, and AAL designed the study. SE performed the statistical analysis. MJ and SE wrote the article. All authors collected data, gave input to the article, and approved of the final version.
DISCLOSURES
MJ: Honoraria: Kite/Gilead, Janssen, Abbvie, AstraZeneca, Pierre Fabre, Roche, Genmab, MSD. Research support: Roche, Abbvie, AstraZeneca, BMS, Janssen. IG: Honoraria: Janssen, Takeda. Research support: Lokon Pharma. All the other authors have no conflicts of interest to disclose.
SOURCES OF FUNDING
This work was partly supported by a public-private real-world evidence collaboration between Karolinska Institutet and Janssen Pharmaceuticals NV (contract: 5-63/2015).