A unique pattern of up- and down-regulation of chemokine receptor CXCR3 on inflammation-inducing Th1 cells
Abstract
Chemokine receptor CXCR3 and its CXC ligands play major roles in Th1 cell-induced inflammatory processes. Here, we examined the expression of CXCR3 by TCR-transgenic Th1 lymphocytes that induce ocular inflammation in mice expressing the target antigen in their lenses. The essential role of CXCR3 in this model was indicated by the observation that the ocular inflammation was significantly blocked by an antibody against this receptor. CXCR3 expression by Th1 cells was elevated during their initial activation in culture and further increased during the consecutive incubation with IL-2. However, CXCR3 expression declined dramatically during the ensuing antigenic reactivation, in parallel with down-regulation of its mRNA. Yet, reactivated Th1 cells exhibited the highest degree of pathogenicity when adoptively transferred into recipients. Transferred reactivated Th1 cells proliferated vigorously and re-expressed CXCR3 while residing in the spleen of recipient mice, reaching ∼85% positivity 4 days post cell transfer when their massive migration to the target eyes began. Importantly, infiltrating Th1 cells underwent profound phenotypic changes in the eye that closely resembled those seen during reactivation of Th1 cells in vitro and included down-regulation of CXCR3. These observations thus show that expression of CXCR3, a major participant in Th1-induced inflammation, fluctuates profoundly during cell activation and migration and is down-regulated upon re-exposure of these cells to the antigen, in vitro or in the target organ.
Abbreviations:
-
- ALPC:
-
Allophycocyanin
-
- CFSE:
-
Carboxy-fluorescein diacetate succinimidyl ester
-
- HEL:
-
Hen egg lysozyme
-
- RT:
-
Reverse transcription
-
- Tg:
-
Transgenic
-
- WT:
-
Wild type
1 Introduction
The crucial roles played by chemokines and their receptors in normal and pathological conditions have been well established 1–3. A major function of these molecules is to mediate and regulate migration of lymphoid cells to different tissues, inducing lymphocyte-mediated inflammation. This process has been studied extensively in experimental systems in which local inflammation is induced by adoptively transferred lymphocytes with specificity toward Ag located in the target tissue 4, 5. Only “activated/effector” lymphocytes are capable of this activity, and published studies provide information concerning the surface molecules that characterize these cells and, presumably, facilitate their tissue invasion 6. These molecules include mainly adhesion molecules, in particular integrin α4 (VLA-4) 7, activation markers like OX-40 and CD25 8, and certain chemokine receptors 9–11. Much attention has been focused on the chemokine receptor CXCR3 and its ligands, Mig/CXCL9, IP-10/CXCL10 and I-TAC/CXCL11, with regard to their pivotal participation in immune-mediated inflammation 10–13. These inflammatory processes are mostly Th1-mediated 14, 15, and CXCR3 has been found to be expressed mostly on Th1 cells, of both activated/effector and memory cell populations 11.
We have recently developed an experimental system in which inflammation is induced in eyes of transgenic (Tg) mice that express hen egg lysozyme (HEL) in their lenses, following adoptive transfer of HEL-specific TCR-Tg polarized Th1 or Th2 cells 16, 17. Recipient mouse eyes with Th1-induced inflammation exhibited strong up-regulation of both CXCR3 and its ligands, Mig/CXCL9 and IP-10/CXCL10 16.
The present study was aimed at analyzing the relationship between expression of CXCR3 and the disease-inducing capacity of Th1 cells. Initial activation in vitro of naive CD4 cells increased both their CXCR3 expression and the capacity to induce ocular inflammation. Reactivation of resting/effector Th1 cells, however, further augmented their inflammation-inducing capacity, but dramatically reduced their expression of CXCR3. The majority of adoptively transferred Th1 cells re-expressed CXCR3 in the spleen, reaching a peak by day 4 when the massive infiltration into the eye began. Importantly, down-regulation of CXCR3 was found in vivo as well, upon re-exposure of the migrating Th1 cells to the Ag in the affected eye.
2 Results
2.1 Anti-CXCR3 Ab blocked the development of Th1 cell-induced ocular inflammation
To learn about the role of CXCR3 in the pathogenic process of ocular inflammation induced by Th1 cells, we treated recipient mice with Ab against murine CXCR3. As seen in Fig. 1, the Ab significantly reduced the severity of pathological changes (Fig. 1A) and decreased the number of inflammatory cells that infiltrated the recipient eyes (Fig. 1B). The inflammation-inhibiting effect of the Ab is exhibited in Fig. 1C. These results thus indicate that CXCR3 plays an essential role in the induction of Th1-mediated ocular inflammation.
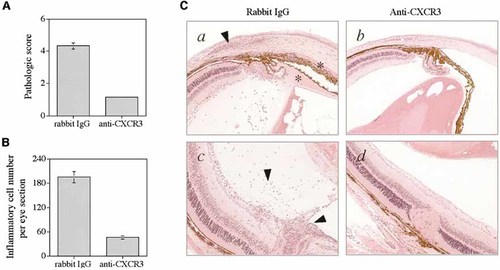
Inhibitory effects of anti-CXCR3 Ab on Th1 cell-induced ocular inflammation. Groups of three recipient mice were injected with reactivated Th1 cells, 0.3×106/mouse, and treated with anti-CXCR3 Ab (100 µg/mouse) or control rabbit IgG on days 1 and 3 thereafter. Eyes were collected on day 4, and disease severity (A) was scored as described 16, 17. Numbers of infiltrating cells (B) were counted throughout the anterior chamber and vitreous cavity in two sections of each eye. The data are the means of two experiments. (C) Histological sections showing the changes in the eyes of mice treated with the control rabbit IgG (panels a and c) or the Ab against CXCR3 (panels b and d). The changes in control eyes, characteristic for Th1-induced ocular inflammation in the anterior (a) and posterior (c) segments of the mouse eye 16, 17, include in particular infiltrates of inflammatory cells (arrowheads) and proteinaceous exudates (asterisks). Original magnification: ×10.
2.2 CXCR3 expression on Th1 cells varies profoundly at different stages of activation in vitro
T cells require activation to acquire pathogenicity 4. To examine the expression of CXCR3 on Th1 cells at different phases of activation in vitro, purified CD4 cell suspensions from 3A9 mice were analyzed by FCM, at five consecutive stages (Fig. 2A): (a) “naive” CD4 cells; (b) “activated” Th1 cells obtained after the initial activation/polarization with HEL, APC, and the Th1 polarization cocktail; (c) “resting/effector-I” Th1 cells collected following an incubation of the activated cells with IL-2; (d) “reactivated” Th1 cells prepared by a second cycle of activation/polarization; (e) “resting/effector-II” cells collected following a further incubation of reactivated cells with IL-2.
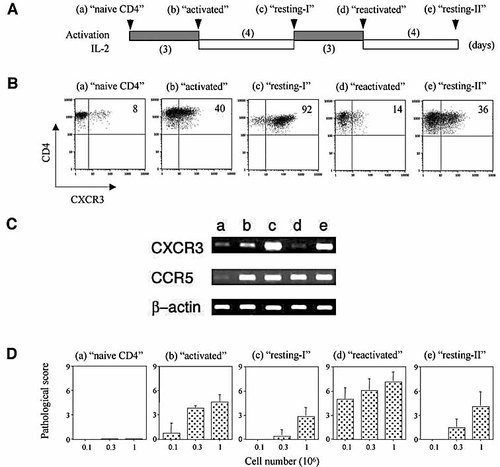
Characterization of Th1 cells from 3A9 donors at different stages of activation in vitro. (A) Schematic description of the culturing conditions applied for each stage. As detailed in Sect. 4, Th cells from 3A9 mice were incubated in the activation stages (gray bar) with HEL, APC and the polarization cocktail, or with IL-2 during the IL-2 stages (open bar). (B) Expression of CXCR3 on the cells at the end of each phase in culture, determined by FCM analysis. The recorded numbers denote the percentages of cells positive for CXCR3. Data of a typical experiment; very similar results were obtained in six additional experiments. (C) RT-PCR analysis of CXCR3 and CCR5 mRNA transcripts in naive CD4 cells (lane a), or Th1 cells collected at the activated (b), resting/effector-I (c), reactivated (d) and resting/effector-II (e) stages. Data of one experiment; the same pattern of responses was seen in two other experiments. (D) Immunopathogenicity of the T cells at the five different stages in culture. T cells collected at the different time points were injected at the indicated numbers into naive HEL-Tg recipients, and the development of ocular inflammation was determined 7 days later. The severity of inflammatory changes was scored as described 16, 17. Data recorded here are means ± SD of three repeated experiments.
The results of a representative FCM analysis of these five cell populations for CXCR3 expression are shown in Fig. 2B. A small proportion of CXCR3-positive cells was found among naive CD4 cells. Increased expression of CXCR3 was detected on activated cells, with 40% of the cells being positive for this marker in the experiment recorded here. Consecutive incubation with IL-2 substantially increased the expression of CXCR3, to 92% positivity of the resting/effector-I Th1 cells. Interestingly, a sharp decline in the proportion of CXCR3-positive cells was observed among reactivated Th1 cells, i.e. following a second antigenic reactivation; only 14% of these cells were positive. A partial recovery of CXCR3 was noted following an additional incubation with IL-2: 36% of the resting/effector-II cells were found positive.
2.3 Reactivation of Th1 cells down-regulates the production of CXCR3 mRNA transcript
To analyze the sharp decline in CXCR3 expression by reactivated Th1 cells, we determined by reverse transcription (RT)-PCR the levels of CXCR3 mRNA transcript in Th cells collected at all the different stages of activation in culture (Fig. 2C). A good correlation was found between the levels of CXCR3 mRNA transcript and surface expression of the protein, as monitored by FCM analysis (Fig. 2B). A weak reaction of CXCR3 mRNA transcript was detected in naive cells (lane a). A more intense band was seen with the activated Th1 cells (lane b), while a remarkably intense reaction was obtained with the sample collected from the resting/effector-I Th1 cells (lane c). In contrast, only a faint reaction was seen with the RNA sample of reactivated Th1 cells (lane d). A striking increase in the level of CXCR3 transcript was observed, however, in the resting/effector-II cells (lane e). The unique pattern of CXCR3 expression was emphasized by comparing it with the expression of CCR5, another selective marker for Th1 lymphocytes 10, 18. The CCR5 transcript was strongly up-regulated by the initial activation, but its levels did not vary much at the ensuing stages in culture (Fig. 2C).
2.4 Reactivation of Th1 cells augments their immunopathogenic capacity in vivo
To correlate between CXCR3 expression and the immunopathogenic capacity of Th1 cells, we injected cells at different stages of activation into HEL-Tg recipients and monitored the level of ocular inflammation 7 days later 16, 17 (Fig. 2D). Naive CD4 cells did not induce any detectable inflammation at the numbers tested here. Activated Th1 cells, with moderate levels of CXCR3, were highly pathogenic, whereas the resting/effector-I cells, expressing high levels of CXCR3, exhibited decreased pathogenicity. Strikingly, reactivation of these resting/effector cells, which drastically reduced their CXCR3 expression (Fig. 2B), profoundly enhanced the pathogenicity of these cells, to produce the highest inflammation scores. Re-culturing the reactivated Th1 cells with IL-2 (resting/effector-II cells), which elevated their CXCR3 expression (Fig. 2B), remarkably reduced their pathogenicity.
These results thus show that expression of CXCR3 on cultured Th1 cells does not correlate well with their pathogenicity, in particular following reactivation that enhances disease-inducing capacity, but reduces CXCR3 expression. The experiments recorded below analyzed this result by tracing the adoptively transferred cells in the recipient mice and by monitoring their proliferation, migration pattern and expression of CXCR3. In all these experiments, naive CD4 cells from 3A9 donors were tested concurrently with the reactivated Th1 cells.
2.5 Kinetics of expansion of transferred Th1 cells in recipient mice
To determine the number of transferred 3A9 donor cells in recipient HEL-Tg mice, we identified them with Ab against both CD4 and the clonotypic TCR (1G12). Counting data are shown in Fig. 3A. Very low numbers of the reactivated Th1 cells were found in the recipients’ spleens and blood on days 1 or 2 post transfer. A sharp increase in the number of these cells was observed, however, on day 4, followed by a decrease on day 7. On the other hand, only small numbers of naive cells were found in spleen and blood, with no clear variations throughout the testing period (Fig. 3A).
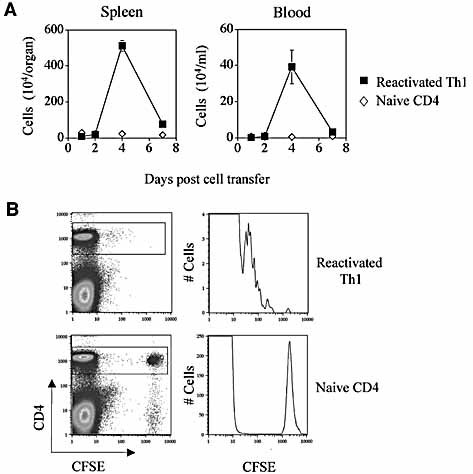
Migration and proliferation patterns of adoptively transferred T cells. (A) Recipient mice of 107 reactivated Th1 cells or naive CD4 cells were sacrificed at different time points, and the number of donor cells (1G12+) in the spleen and blood was determined by FCM. The recorded data are means ± SEM of four experiments. (B) Reactivated Th1 cells or naive CD4 cells from 3A9 donors were labeled with CFSE and injected i.v. into HEL-Tg recipients, 107 cells per recipient. Spleens of recipient mice were collected 2 days post cell injection, and their CD4 cell subpopulation was analyzed by FCM for CFSE dilution.
A similar pattern of changes in donor cell number was found when the cells were injected into wild-type (WT) recipients (data not shown), thus indicating that the expression of HEL in the HEL-Tg mice had little or no effect on the expansion of transferred cells.
To learn about the proliferation of the transferred cells in the recipient mice, we labeled them with carboxy-fluorescein diacetate succinimidyl ester (CFSE) before injecting them into recipient mice. The rate of cell division was evaluated by analysis of CFSE dilution. As seen in Fig. 3B, reactivated Th1 cells divided rapidly in the recipients’ spleens, with the majority of reactivated Th1 cells already substantially diluting their CFSE on day 2 post cell transfer. In contrast to the reactivated Th1 cells, essentially no divisions were noted among the control naive CD4 cells during the time period tested (Fig. 3B).
2.6 Re-expression of CXCR3 by the transferred Th1 cells
To learn about the function-related features of the transferred reactivated Th1 cells, we performed FCM analysis of the 1G12+ lymphocytes in the recipients’ spleens and blood, using Ab against a variety of key phenotypic markers (Fig. 4). Similar patterns of marker expression were seen in blood and spleen. Of particular importance are the observations concerning CXCR3 expression: only small proportions of the 1G12+ cells expressed CXCR3 on day 1, but the expression of this receptor increased rapidly on day 2 and reached its peak (∼85%) on day 4 post cell injection. Moderate levels of decrease in CXCR3 expression were seen on day 7.
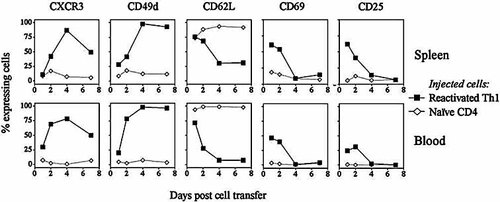
Expression of CXCR3 and other surface molecules on donor cells in spleen and blood of recipient mice. HEL-Tg recipient mice were injected with 107 reactivated Th1 cells or naive CD4 cells of 3A9 mice, and spleens and blood were collected at different time points and stained for the five cell markers. The data are expressed as percentage of cells stained for each of the tested markers at the indicated time points, and the recorded values were obtained with the gated subpopulations of CD4+1G12+ cells. These data were collected in a typical experiment; similar results were obtained in four repeated experiments.
Remarkable changes were also observed in the levels of most other cell surface markers on the donor cells (Fig. 4). Low proportions (20–25%) of the transferred cells expressed CD49d (VLA-4), a tissue-homing receptor 19, on day 1, followed by a steep increase, with almost all cells (>95%) expressing this adhesion molecule on days 4 and 7. In parallel with up-regulation of CD49d, a substantial down-regulation of CD62L (L-selectin), a lymph node-homing receptor 20, was found on the donor cells on day 4 or day 7 post transfer. On the other hand, the levels of the activation markers CD69 and CD25 gradually declined on donor cells during the 7-day period. Similar changes in surface markers were seen on donor cells when transferred into WT recipients (data not shown), thus indicating that the changes are not due to exposure to the Ag in the HEL-Tg mice.
Unlike the changes observed on donor cells in recipients of reactivated Th1 cells, essentially no fluctuations were seen in the levels of these surface molecules on the donor cells in recipients of the naive CD4 cells from 3A9 donors. Throughout the test period, these cells retained the profile of naive T lymphocytes (Fig. 4).
2.7 Kinetics of migration of T lymphocytes into the recipients’ eyes
To monitor migration of donor lymphocytes into the recipients’ eyes, we employed two methods: (i) tracing the CFSE-labeled cells in mounted retinas at the early stage of invasion, (ii) FCM analysis of cells collected from the affected eyes following treatment with collagenase and staining with Ab against CD4 and 1G12.
Small numbers, between 6 and 20, of CFSE-labeled cells were found in retina mounts of recipient mice at 3 and 24 h post cell injection (data not shown). FCM analysis of cells collected from recipient eyes showed that very low numbers of infiltrating lymphocytes were present in the eyes on day 2 post transfer. However, a sharp increase in the number of these cells was observed on day 4, and a further increase was seen on day 7 (Fig. 5). The data of a representative experiment summarized in Fig. 5 show that the eye infiltrate consisted mostly of the donor cells (1G12+) on day 4, while on day 7, the majority of cells were of host origin (1G12–).
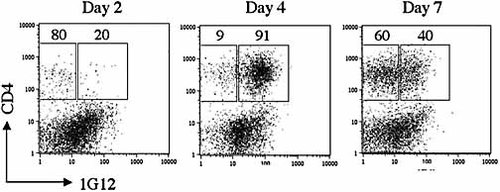
FCM analysis of the infiltrating lymphocytes in inflamed recipient eyes. HEL-Tg mice were injected with 107 reactivated Th1 cells of 3A9 donors, and their eyes were collected on days 2, 4 and 7. Infiltrating cells were investigated using 1G12 Ab to identify donor cells. Recorded numbers denote percent values of the gated subpopulations. Similar results were obtained in four repeated experiments.
Data recorded here thus show that a very small number of the transferred Th1 cells entered the recipients’ eyes immediately after transfer, while a massive migration started on day 4, with many host lymphocytes participating in the process.
2.8 Down-regulation of CXCR3 on the eye-infiltrating Th1 cells
FCM analysis of donor cells collected from eyes and spleen on day 4 revealed remarkable differences in their surface marker profiles (Fig. 6A). The profile of donor cells collected from the blood of these mice resembled closely that of the cells from the spleen (data not shown). Of particular interest is the difference in CXCR3 expression between the two populations of cells. Whereas this receptor was found on the majority (85–90%) of spleen donor cells, it was expressed by only a small proportion (10–15%) of the eye-infiltrating cells.
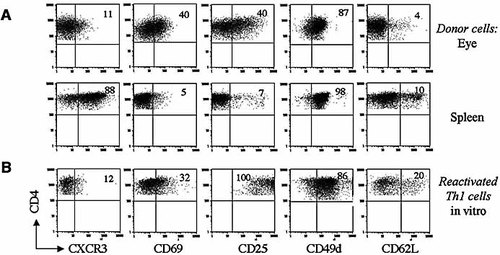
Expression of cell surface markers on the donor cells that infiltrate the eyes or reside in the spleen of recipient mice. (A) HEL-Tg mice were injected with 107 reactivated Th1 cells from 3A9 donors. After 4 days, eye-infiltrating cells or splenocytes were stained for CD4, 1G12 and the five cell surface markers. The panels show the staining profiles of the subpopulations of CD4+1G12+ for the indicated markers, as well as the percentage of cells positive for each marker. (B) Staining profile of reactivated Th1 cells in culture, for comparison with the staining profiles of the cells collected from the recipient organs. The recorded data of recipient mice were obtained in a representative experiment; similar observations were made in three additional experiments.
Another difference between the donor cells in the recipient spleen and eyes can be observed in the proportion of cells expressing the activation markers CD25 and CD69. These markers were found on less than 10% of cells in the spleen, but on approximately 40% of the cells in the eyes. The populations of donor cells in spleen and eyes were similar in expressing high levels of CD49d and low levels of CD62L.
These results thus show that Th1 cells underwent profound changes following infiltration into the recipient eyes. These changes included a sharp decline in CXCR3 expression, but up-regulation of the activation markers CD25 and CD69. The similarity in the profile of surface markers between the eye-infiltrating cells (Fig. 6A) and Th1 lymphocytes reactivated in vitro (Fig. 6B) suggests that the donor cells underwent reactivation upon re-exposure to the Ag in the recipient eyes.
3 Discussion
This study provides new information concerning the expression of CXCR3 by inflammation-inducing Th1 lymphocytes. The crucial role played by CXCR3 in the inflammatory process was indicated by the inhibitory effect of the CXCR3 Ab (Fig. 1). Our studies have demonstrated, however, a complex pattern of up- and down-regulation of CXCR3 on the inflammation-inducing Th1 cells at the different phases they undergo during activation in vitro, as well as during migration, proliferation, and activation in vivo.
Th1 cells polarized/activated in vitro demonstrated increased expression of CXCR3, but the highest levels of the receptor were observed on resting/effector cells (Fig. 2B). Interestingly, reactivation of the resting/effector cells with the specific Ag produced a drastic decline in the expression of CXCR3 (Fig. 2B). This steep decline could be attributed to two mechanisms, i.e. internalization of the receptor 21 and/or down-regulation of its production. Our data suggest, however, that internalization of CXCR3 plays a minor role, if any, in the decline of CXCR3 on reactivated Th1 cells in our in vitro system (unpublished data). On the other hand, reactivation of the resting/effector cells was found to drastically down-regulate the expression of CXCR3 mRNA transcript by these cells (Fig. 2C), suggesting that this is the major mechanism responsible for the decline of surface CXCR3 on reactivated Th1 cells. Our observation with mouse Th1 cells is in line with previous findings with human T cells 22–24. It is of particular interest that, unlike with CXCR3, reactivation of the Th1 cells had little effect on the expression of CCR5 (Fig. 2C), another chemokine receptor that characterizes Th1 cells 10, 18.
A major finding in the present study is the dissociation between CXCR3 expression on Th1 cells and their capacity to induce ocular inflammation when adoptively transferred into recipients expressing the target Ag (HEL) in their eyes. Most strikingly, the highest level of immunopathogenicity was found with reactivated cells in which the CXCR3 was drastically reduced (Fig. 2D and 2B, respectively). This observation may seem to be paradoxical, since CXCR3 was shown to play a major role in Th1-mediated inflammation, as indicated by the inhibitory effect of CXCR3 Ab (Fig. 1) 13, 25, and both this receptor and its ligands (CXCL9 and CXCL10) were found to be up-regulated in eyes in which inflammation was induced by reactivated Th1 cells 16. The dissociation between CXCR3 expression and high pathogenicity of reactivated Th1 cells could be explained, however, by the small number of the Ag-specific effector/activated Th1 cells that trafficked into the eye shortly after adoptive transfer to initiate the disease, whereas most Th1 cells migrated first to the spleen and other peripheral lymphoid organs 2, 5, 26, where they proliferated and re-expressed CXCR3, before their massive migration into the eye (Fig. 4, 5). These findings thus suggest that CXCR3 does play a major role in mediating the migration of tissue-specific Th1 cells into the target eye tissues, where high levels of CXCR3 ligands are produced 16. It is further proposed that the inhibitory effect of CXCR3 Ab in the present system was exerted mainly during the late massive migration of CXCR3-expressing Th1 cells into the recipient eyes.
Of particular interest are the observations concerning the migration pattern and phenotype changes of the transferred Th1 cells. The number of these cells in the recipient spleen and blood increased rapidly, reaching a peak on day 4 post cell injection (Fig. 3A). The mechanism responsible for this rapid expansion of donor cells is not known, but it is possible that an interaction between the donor and host lymphocytes plays an initiating role. During this period of 4 days post injection, the reactivated donor cells underwent remarkable changes in their surface molecules, including down-regulation of the activation markers CD25 and CD69 and of the lymph node-homing receptor CD62L. On the other hand, these cells showed up-regulation of the integrin CD49d and, important to this study, of chemokine receptor CXCR3 (Fig. 4). It is of note that the change in the marker profile of donor cells while residing in Tg recipients was similar to that observed when these cells were transferred into WT mice, indicating that the profile change could not be attributed to the presence of HEL in Tg mice. It is assumed that these phenotypic changes are required for the process of cell migration from secondary lymphoid organs into the target organ, the eye. The patterns of migration and phenotypic changes of donor cells observed in the present study resemble those reported by Flugel et al. 5 in a rat system of adoptively transferred EAE. However, it is of note that, unlike in the present study with mice, only minor changes were seen by Flugel et al. in the expression of CXCR3 on the donor cells in the recipient rat spleen 5. The cellular processes and changes we observed in donor cells prior to their migration to the target organ also resemble in part those described by Roman et al. 27 in murine recipients of naive CD4 cells specific to influenza, following infection with this virus. Expression of CXCR3 has not been tested, however, by Roman et al.
Another major observation of the present study is represented by the remarkable differences between surface markers on the donor cells in the eye and in the spleen or blood of the recipient mice. Of particular interest is the difference in the expression of CXCR3: unlike the majority of donor cells expressing CXCR3 in the spleen or blood, only ∼1/10 of donor lymphocytes in the eyes expressed this receptor (Fig. 6A). It is likely that the majority of Th1 cells infiltrating the eye do initially express CXCR3, as indicated by the inhibitory effect of the CXCR3 Ab. The decline in the receptor levels on the infiltrating lymphocytes could be attributed in part to internalization of the receptor 21 upon interaction with its ligands MIG/CXCL9 and IP-10/CXCL10, which are produced in large amounts in these recipient mouse eyes 16. We prefer, however, the alternative notion that CXCR3 is down-regulated on the invading Th1 cells following their re-encounter with the target Ag that is expressed in these eyes. The latter notion is in line with the suggestion of Sallusto et al. 24 that loss of responsiveness to inflammatory chemokines following antigenic stimulation may result in a migratory arrest for activated T cells. It is further conceivable that this arrest of Th1 cells promotes their accumulation at the inflammation site, allowing them to fully exert their functional activity, such as cytokine release 16. In addition to showing down-regulation of CXCR3, the eye-infiltrating cells exhibited a profile of activated T lymphocytes in expressing higher levels of the activation markers CD25 and CD69 than the cells in the spleen (Fig. 6A). The similarity in phenotype profile between the eye-infiltrating cells and Th1 cells reactivated in culture (Fig. 6B) supports the notion that donor T cells undergo reactivation in the eye upon exposure to their target Ag. Notably, a similar observation with increased expression of CD25 by tissue-infiltrating cells was made by Flugel et al. 5 and Kawakami et al. 28 with lymphocytes collected from the inflamed spinal cord in rats with adoptively transferred EAE, and by Roman et al. 27 with lymphocytes from the inflamed lungs of murine recipients of influenza-specific CD4 cells following infection with the virus.
In summary, this study describes for the first time the complex pattern of CXCR3 expression by inflammation-inducing Th1 cells in an adoptive transfer Tg mouse system. The fluctuations in CXCR3 expression observed on migratory Th1 cells in recipient mice closely resemble those on cells at different stages of activation in culture. The data are in line with the notion that CXCR3 is essential for inflammation induction by Th1 cells, but its expression is restricted in vivo to the period during which these lymphocytes reside in secondary lymphoid organs and undergo phenotype changes prior to their migration to the target organ. On the other hand, CXCR3 expression is down-regulated on Th1 cells during reactivation by the Ag, in culture or in the target tissue.
4 Materials and methods
4.1 Mice
HEL-Tg mice expressing HEL under control of the αA-crystallin promoter, on the FVB/N background, were generated as described 29. HEL-specific TCR-Tg mice, on the B10.BR background and designated 3A9 30, were a generous gift from Mark Davis (Stanford University, Stanford, CA). Tg mice from each of the two lines were mated to produce (FVB/N × B10.BR) F1 hybrids that have been used in all experiments of the present study. In all adoptive transfer experiments recorded here, the cells were from 3A9 donors, whereas recipients were HEL-Tg mice. All manipulations were performed in compliance with the NIH Resolution on the Use of Animals in Research.
4.2 Generation of HEL-specific Th1 lymphocytes
Th1 cells at different stages of activation were prepared as described in detail elsewhere 16, 17 and illustrated here in Fig. 2A. Briefly, CD4 cells from 3A9 mice were incubated with HEL for 3 days under Th1-polarizing conditions to yield activated preparations. Ensuing incubation with IL-2 for 4 days produced resting/effector-I cell samples, which were cultured again with HEL and the Th1 polarization cocktails to produce the reactivated cell preparations. In some experiments, as indicated, reactivated cells were expanded once more with IL-2 to obtain resting/effector-II cell preparations.
4.3 Induction of ocular inflammation
Different numbers of Th1 cells at the various stages of activation were injected via the tail vein into naive HEL-Tg mice to induce disease. Recipient mice were sacrificed 4 or 7 days following cell transfer, as indicated. Eye sections were stained by conventional hematoxylin and eosin procedure, and the severity of inflammation was scored as described 17.
4.4 Treatment with anti-CXCR3 Ab
Rabbit Ab against murine CXCR3 25 was given i.v. at 100 µg per mouse on days 1 and 3, and recipient mice were sacrificed on day 4 post adoptive transfer of cells. Normal rabbit IgG (Pierce, Rockford, IL) was used for control. Eye sections were prepared, and the pathological changes were evaluated as described above. The numbers of inflammatory cells were counted throughout the anterior chamber and vitreous cavity of eye sections.
4.5 FCM analysis
mAb against CD4 [allophycocyanin (ALPC)], CD49d (FITC), CD62L (PE), CD69 (FITC) and CD25 (PE) were purchased from PharMingen (San Diego, CA). Rabbit anti-CXCR3 Ab was from Zymed Laboratories (San Diego, CA), whereas goat anti-rabbit IgG Ab (PE) was from Southern Biotech (Birmingham, AL). A clonotypic mAb specific for the Tg TCR of the 3A9 mice, designated 1G12, a generous gift from Emil Unanue (Washington University, St. Louis, MO), was used conjugated with biotin. The secondary Ab conjugated with streptavidin-peridinin chlorophyll protein (SA-PerCP) for 1G12 detection was from Becton Dickinson (San Jose, CA). Isotype Ab (PharMingen) and rabbit IgG (Pierce, Rockford, IL) were used for control and anti-CD16/CD32 Ab (PharMingen) was used to block Fc receptors in all stainings.
Cultured cells were collected at different stages of activation and washed thoroughly before staining. Blood, spleens and eyes of recipient mice were collected at different time points following adoptive transfer of donor cells. Single-cell suspensions of blood and spleen cells were prepared by conventional methods. Eyes were dissected and then digested in RPMI 1640 medium containing 10% FCS and 1 mg/ml collagenase (Sigma-Aldrich, St. Louis, MO) for 2 h at 37°C. Collected cells were filtered through a cell strainer, followed by erythrocyte lysis with ammonium chloride potassium (ACK) buffer and washing in FACS buffer. FCM analysis was performed on a FACSCalibur (Becton Dickinson). For the cultured lymphocytes, 10,000 events in a live gate were acquired, and 106 events were acquired for cell samples collected from recipient mice.
4.6 CFSE dilution assay
Reactivated Th1 cells or naive CD4 cells (5×107 cells/ml) were labeled with CFSE (Molecular Probes, Eugene, OR) for adoptive transfer as described elsewhere 31. CFSE-labeled cells were injected i.v. into HEL-Tg mice (1×107 cells in 200 µl). Recipient mice were sacrificed at the indicated time points, and splenocytes were collected and stained with ALPC-conjugated anti-CD4 mAb (PharMingen). Cell suspensions were analyzed on a FACScan Cytometer (Becton Dickinson) and were gated for CD4+CFSE+ dye dilution peaks, with 106 events being acquired in a live gate.
4.7 RT-PCR
Total RNA was prepared from naive CD4 cells or Th1 cells at different stages of activation in culture, and cDNA was synthesized for determination of CXCR3 and CCR5 transcripts by RT-PCR, as detailed elsewhere 32. The primers used for RT-PCR were: CXCR3: 5′-GCTAGATGCCTCGGACTTTG-3′ and 5′-GCTGATCGTAGTTGGCTGATA-3′, amplifying a 557-bp product; CCR5: 5′-ACTTGGGTGGTGGCT-GTGTTT-3′ and 5′-TTGTCTTGCTGGAAAATTGAA-3′, amplifying a 539-bp product; β-actin: 5′-GTGGGCCGCTCTAGGCACCAA-3′ and 5′-TCGTTGCCAATAGTGATGACTTGGC-3′, amplifying a 650-bp product. The PCR products were analyzed on 1.2% agarose gels containing 0.5 mg/ml ethidium bromide.
Acknowledgements
We thank Drs. Joost J. Oppenheim and Philip M. Murphy for useful discussions and advice, and Drs. Mercedes M. Campos and W. Gerald Robison, Jr., for use of the Biological Imaging Care Facility, NEI.
- WILEY-VCH
- WILEY-VCH
- WILEY-VCH
- WILEY-VCH
- WILEY-VCH
- WILEY-VCH




